 JAS 2000 Quarterly Rpt. sidebar
JAS 2000 Quarterly Rpt. sidebar
|

Survival and
Straying of Pink Salmon
Using Recoveries of Coded-Wire Tags and
Thermally-Induced Otolith Marks
(Quarterly
Report for July-Aug-Sept 2000)
by
Donald Mortensen, Alex Wertheimer, Jacek Maselko,
and Sidney Taylor

Figure 1. Thermally induced otolith marks is an
alternative
method of marking pink salmon.
Anadromous salmonids are known
for their ability to home to their natal streams to
spawn. The ability to home is thought to be due to
olfactory memory of trace chemicals in the natal
stream and perhaps, also, population-specific odors
(pheromones). Although most salmon home to natal
streams, some stray to others. The degree of
straying varies among years, species, stocks, ages,
and genders and can change with the number of fish
returning. Stream type, water source, and physical
location may also play an important part in the
extent of straying. Natural straying may be
essential for colonizing new habitat, recolonizing
degraded spawning habitats and also may reduce
inbreeding in small populations.

Figure 2. Typical half-length (0.5 mm) coded-wire
tag, a device
commonly used in marking pink salmon.
Pink salmon, Oncorhynchus
gorbuscha, have a particular reputation as
strayers. Studies conducted in Prince William Sound,
Alaska, (Sharr et al., 1995 and Habicht et
al., 1998) indicate that coded-wire tags (CWT)
(Figure 2 above) inserted in juvenile pink salmon
snouts may exacerbate straying of those surviving to
maturity. As olfaction is thought to play an
important part in the ability of the salmon to find
its home stream, the injection of a CWT into
sensitive olfactory tissue in the fish’s
nasal area could negatively influence homing
ability. Studies conducted at Auke Creek,
Alaska, in the 1980s noted that return rates of
unmarked adults have been as high as seven times
that of fry tagged with CWTs (tagged fish)
concurrently, suggesting a higher mortality or
straying rate for tagged fish. To provide an
external cue of the CWT, the adipose fin is usually
clipped. It has also been shown that
fin-clipping of wild pink salmon fry substantially
reduces return rates. The apparent handicap of
tagged fry may be due in part to the trauma of
fin-clipping as well as tag injection, which affects
survival or homing. Greater mortality rate of tagged
fish in relation to that of untagged fish could
result in errors in the estimation of fishery
contributions, survival rates, and straying rates of
hatchery or wild fish that the tagged fish are
assumed to represent.
The relatively new method of thermally inducing
marks on the otoliths of fish (Figure
1 at top) is considered to be a noninvasive
way of marking pink salmon fry and can be used to
measure survival, straying, and the effect of
applying CWTs on survival and straying. Thermal
marking is not known to cause trauma to olfactory or
other nerve tissue, and an entire hatchery
production can be marked, greatly increasing
sampling resolution. About 14 km away from Auke
Creek, the Gastineau Hatchery, operated by Douglas
Island Pink and Chum Corporation, has routinely
thermally marked portions or all of their pink and
chum salmon production since 1990 with no
apparent effect on survival. In 1994, the
hatchery thermally marked about 33% of their 1994
brood (1996 adult ) pink salmon.
Our study objectives were to compare the survival
and straying rates of thermally marked and
coded-wire-tagged Auke Creek pink salmon and
thermally marked Gastineau Hatchery pink salmon.
Methods
Auke Creek is a small, lake-fed
stream, which empties into Auke Bay in northern
Southeast Alaska (Figure
3). The National Marine Fisheries
Service maintains a permanent two-way counting weir
and small research hatchery at the confluence of
Auke Creek with Auke Bay. We spawned a portion
of the early- and late-run pink salmon returning to
Auke Creek in 1994 to provide eggs for thermal
marking and seeded them into gravel incubators
plumbed with water from Auke Creek. A small
water heating system at the Auke Creek hatchery
provided warm water, which allowed us to thermally
induce unique five-band patterns in the otoliths of
prehatch early- and late-run pink salmon embryos.
To induce a thermal mark on the otoliths of
salmon, the ambient water temperature surrounding
the incubating salmon eggs or alevins is raised or
lowered by at least 3 degrees for 16 to 24 hours.
The water temperature is then quickly returned
to ambient which results in a dark increment or
stress check being entrained in the otolith of the
incubating salmon. This procedure is repeated
several times to put a series of checks (much like a
bar-code) on the otolith. As these marked pink
salmon fry emerged from the gravel incubators in
spring 1995, we adipose-clipped and injected CWTs
into representative samples. We also
adipose-clipped and injected CWTs into wild Auke
Creek fry throughout their emergence (Table
1).
Upon their return to Auke Creek and Gastineau
Hatchery in 1996, adult pink salmon were examined
for adipose clips (denoting CWTs) daily. The
adipose clipped fish were killed and counted and the
heads removed in order to recover the CWTs and
otoliths. We also conducted foot surveys twice
a week at Waydelich, Fish, and Auke Nu Creeks.
At Salmon Creek, the water source for
Gastineau Hatchery, we conducted foot surveys once a
week. During the foot surveys otoliths were removed
from spawned-out pink salmon carcasses and the
carcasses were also examined for adipose clips
(denoting stray, coded-wire tagged Auke Creek pink
salmon).
When an adipose clipped carcass was found, the head
was taken back to the laboratory where the CWT was
removed and decoded. We removed the otoliths
of all carcasses found without adipose clips to
provide estimates of thermally marked pink salmon
strays from Auke Creek and Gastineau Hatchery. At
the laboratory, CWTs were extracted from the heads
of adipose-clipped fish and decoded using a
dissection microscope. The sagittal otoliths
from the carcasses were glued to microscope slides
and ground to the central lateral plane using
1,000-grit sandpaper. The ground otoliths were
then examined for thermal marks under a compound
microscope at magnifications between 200 and 400 X.
Adult survival of tagged (CWT) fish was computed as
the proportion of fry released from a tagged group
that returned to the Auke Creek weir. We
computed the survival of thermally marked fish
returning to Auke Creek as the expanded number of
marks divided by the estimated number of thermally
marked only fry. The run composition of Auke
Creek fish was then estimated by allocating unmarked
fish based on representative tagged or marked
groups. The estimates of unmarked pink salmon from
Gastineau Hatchery and the wild Auke Creek
component were subtracted from the total estimate of
unmarked fish which spawned in Auke Creek. The
remainder of the unmarked fish in Auke Creek were
assumed to be strays from other streams or from
intertidal spawning below the Auke Creek weir.
Our estimate of the number of unmarked, wild Auke
Creek adults was derived from CWT wild fry survival
and a “handicap ratio,” defined as the ratio of
the survival of marked only fry to tagged and marked
fry. The corrected survival rate was then
applied to the number of unmarked fry released at
the weir.
Spawning populations in the three streams within 10
km of Auke Creek (Figure
3) were estimated using a mark-recapture
method. We estimated the proportion of carcasses in
the stream and the number of strays in each creek by
dividing the number of otolith marks or CWTs found
in the sampling fraction. We also examined
pink salmon carcasses in Salmon Creek, which is
approximately14 km from Auke Creek, for adipose-fin
clips and thermal marks but did not make a
population estimate. All pink salmon adults
returning to Gastineau Hatchery were examined for
the presence of adipose fin clips (CWT), and about
12% were sampled for thermal marks.
We computed the straying rate of marked Auke Creek
fish to the index creeks by summing the estimates of
the strays to each site, and dividing by the number
of fish that returned to the sample area.
Results and Discussion
Tagging small pink salmon with CWTs and removing
the adipose fin reduced their rate of survival; fry
marked only with thermal marks had consistently
greater survival rates. Survival rates for the three
CWT groups, 0.20%, 0.17% and 0.16% for the early-,
late- and wild run pink salmon, respectively (Table
1), were not significantly different (P >
0.5). Survival rates for the groups marked
only with thermal marks were 0.41% and 0.47% for the
early- and late-run pink salmon, respectively;
these rates were also not significantly different (P
>0.4). Estimated survival of fry marked
only with thermal marks was 2.1 times greater for
early-run and 2.8 times greater for late-run than
for the respective CWT early and late run groups.
We took the average of these ratios (2.4) to
provided a mean handicap to estimate a survival rate
for unmarked wild pink salmon fry (0.39%).
Thermally inducing marks on salmon otoliths is
commonly assumed to have no effect on survival, but
there has been little or no explicit testing of this
assumption. Our results showed no
significant difference in survival between
thermally-marked fry from the early- and late-run
groups that were also tagged with CWTs and wild fry
tagged with CWTs but with no thermal marks, leading
to the conclusion that, indeed, thermal marking had
no effect on survival.
Pink salmon returning to Auke Creek originated from
the various groups of Auke Creek pink salmon and
from strays from other streams and nearby
Gastineau Hatchery (Figure
3). Auke Creek coded-wire-tagged and
thermally-marked pink salmon accounted for 3,516 of
the total return. The unmarked portion of the
return from Auke Creek wild fry was estimated at
175. We calculated this by multiplying the
survival rate for unmarked wild fry (0.39%) by the
number of unmarked wild fry released (43,967).
Expanding for sampling fraction and marking
fraction, the number of Gastineau Hatchery strays in
Auke Creek was 462 fish. This left about 605
of the Auke Creek run, which was composed of strays
from other streams or from intertidal spawning below
the weir.
We recovered strays from five of the six Auke Creek
marked groups in four streams and the Gastineau
Hatchery (Table
3). Most of these fish were from
thermally-marked groups: 49 Auke Creek thermal
marks were observed in the carcasses sampled, while
only three CWTs were recovered. The frequency
of Auke Creek strays in the escapement varied
substantially among the streams that were sampled.
Salmon and Waydelich Creeks had the greatest
proportions of Auke Creek strays, 7.6% and 6.1%,
respectively, while Auke Nu Creek and Fish Creek
were smaller, at 2.1% and 2.6%, respectively. Gastineau
Hatchery had the lowest frequency of Auke Creek
strays (0.3%), significantly less than the
proportion in Salmon Creek, the watershed for the
Gastineau Hatchery. Crowding in the hatchery
ladder may have influenced more of the Auke Creek
strays to enter the less crowded stream bed at
nearby Salmon Creek.
For coded-wire-tagged and thermally-marked groups,
more late-run strays were observed than early-run
strays (Table
4). We recovered two late-run tagged
strays compared to no early-run tagged strays and 31
late-run thermally-marked strays vs. 18 early-run
thermally-marked strays. There were not enough
CWT recoveries to test for statistical differences,
but we did find that thermally-marked early-run fish
strayed significantly less than the thermally-marked
late-run fish. This may be the result of the
early return timing and stream characteristics.
These early-run fish may experience unique
stream characteristics such as odor, water
temperatures, and flows which would result in
greater homing specificity. Additionally, by
virtue of its early return, the early-run fish
are not as susceptible to attraction by aggregations
of pink salmon near other streams as are the
late-run fish.
We estimated straying rates for Auke Creek pink
salmon to streams within 10-km of Auke Creek (Auke
Nu, Waydelich and Fish Creek); and within 14-km of
Auke Creek (Salmon Creek and Gastineau Hatchery).
Within 10-km the straying rate for fish
tagged with CWTs was based on just one recovery;
estimated rates were zero for early- and wild-run
fish and 5.6% for the late-run fish (Table
4). Estimated straying rates increased
to 1.4% and 6.6% for wild and late-run
coded-wire-tagged fish respectively, when strays
to14 km are included. Within 10 km of Auke
Creek we estimated straying rates for
thermally-marked Auke Creek pink salmon of 2.5 % and
3.2 % for the early- and late-run fish, respectively.
Estimates of straying rates increased to 4.4%
and 6.7% for the early- and late-run respectively
with the addition of strays to 14 km. These
estimates are biased low because there was no
expansion of strays due to sampling fraction in
Salmon Creek.
To increase the precision of the straying rate
estimates, the data for coded-wire-tagged and
thermally marked pink salmon were pooled across run
types. Pooling was justified because both
treatments marked fish over the entire emigration of
the Auke Creek population. We calculated
straying rates between tagged and marked pinks as a
group by estimating the total stray rate for each
group and observing the overlap of the confidence
intervals (Table
4). For streams within 10 km and 14 km
of Auke Creek, the point estimate of straying for
the coded-wire-tagged fish was 1.9% and 2.4%,
respectively. The point estimates of straying
of the thermally marked fish were higher: 2.8% and
5.7%, respectively, for streams within 10 km and 14
km of Auke Creek (Table
4). The extensive overlap of the
confidence intervals from each group indicates that
adipose fin clipping and marking with CWTs did not
affect straying rates in this study. Although
tagging with CWTs did not increase the straying rate
of pink salmon in our study, other researchers
(Habicht et al.,1998 and Thedinga et al., 1999) have
shown circumstantial and direct evidence that CWTs
can affect homing and straying in pink salmon.
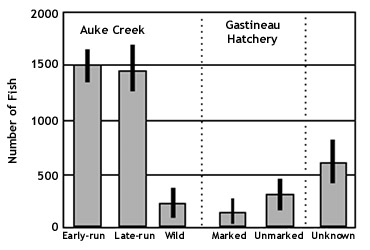
Figure 4. Origin of pink salmon returning to
Auke Creek, expanded for marking and sampling
fraction. Vertical bars denote the standard
deviation around the estimate.
Only 19% of the pink salmon
sampled for thermal marks at Gastineau Hatchery had
the hatchery marks (Figure 4 above). This
proportion is significantly less than the 33%
expected based on marking rate. Assuming
there was no mortality or differential straying due
to thermal marking, then large numbers of unmarked
salmon must have strayed to the hatchery to reduce
the mark rate. Movement of fish from other streams
into the hatchery is demonstrated by the presence of
Auke Creek thermal marks (2%) in the return. At
Salmon Creek, 15% of the carcasses sampled had
Gastineau Hatchery thermal marks. This
frequency was not significantly different than that
at the hatchery itself, indicating that Salmon Creek
pink salmon and the Gastineau Hatchery pink salmon
were returning in similar proportions to the two
sites. The suspected decrease in the proportion of
sampled fish with the Gastineau Hatchery mark at the
hatchery may have been largely due to Salmon Creek
fish returning to the hatchery. Because the
hatchery and Salmon Creek are adjacent and share the
same water shed, we considered the hatchery fish
returning to either the hatchery or Salmon Creek to
have homed.
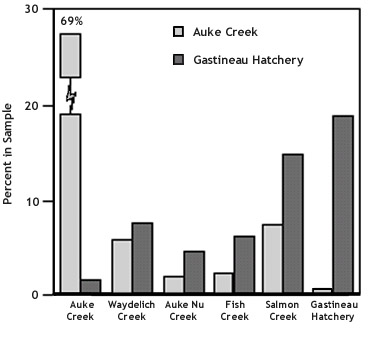
Figure 5. Percent of Auke Creek and Gastineau
Hatchery pink salmon found in samples of carcasses
taken from local creeks and the Gastineau hatchery.
Gastineau Hatchery fish also
strayed to the other four streams sampled. The
percentage of thermal marks in carcasses
sampled in these streams ranged from 4% in Auke
Creek to 8% in Waydelich Creek (Figure 4 above).
The estimated straying rate of Gastineau
Hatchery pink salmon was 6.9% (Table
4), if Gastineau Hatchery marks recovered in
Salmon Creek and at Gastineau Hatchery are assumed
to have homed. This estimate is biased high
because we did not estimate the population of Salmon
Creek and were unable to adjust the number of homing
fish in the sampling fraction.
Our results indicate substantial straying among wild
and hatchery pink salmon populations in the Auke
Creek area. This is consistent with the
concept of spatial or geographic structure within
populations of pink salmon, where the lack of
multiyear age structure provides no buffering to
compensate for poor survival years. The genetic
homogeneity of pink salmon found in Southeast Alaska
also indicates a relatively high degree of straying
especially between local systems. An earlier
study using a genetic marker in Auke Creek pink
salmon (Gharrett, 1985) showed low gene flow to
Waydelich Creek. Thus while we did find strays from
all Auke Creek population components in local
creeks, they may have enjoyed limited reproductive
success. The large degree of mixing does
however provide the mechanism for gene flow and
colonization.
That local adaptations and differences persist among
pink salmon populations exposed to large potential
introgression underscores the importance of
environmental selection for fitness traits, such as
migration timing or developmental rates, in the
adaptive strategy of pink salmon. Further
straying and survival studies on Auke Creek pink
salmon are being considered, perhaps combining
genetic, thermal, and CWT marking to directly
address the question of straying and resultant gene
flow to other pink salmon populations in the
vicinity of Auke Creek. A higher level of CWT-
and thermal-mark recovery will provide better
estimates of pink salmon populations in the streams
as well as better estimates of straying and survival
Up
|

