In its efforts to provide public health guidance to state and local health departments, other federal agencies, health professionals, and the public on the health effects of environmental pollutants, the Agency for Toxic Substances and Disease Registry (ATSDR) relies on the latest advances in computational toxicology. Housed within the Division of Toxicology and Environmental Medicine, the Computational Toxicology Laboratory (CompTox Lab) supports the agency mission by developing and applying state-of-the-art computational toxicology methods such as physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) modeling, quantitative structure-activity relationship (QSAR) methods, and benchmark dose (BMD) models. The developed PBPK, QSAR and BMD models are then implemented to determine and characterize risks from exposure to hazardous substances.
PBPK models mathematically describe
absorption, distribution, storage, metabolism, and excretion of
chemicals as they are
 introduced
into the body. The models help predict human toxicity following
exposure to hazardous substances. The most common modeling applications
involve predictions based on information derived from animals (across
species extrapolations) or exposure routes (across route extrapolations)
other than those being studied.
introduced
into the body. The models help predict human toxicity following
exposure to hazardous substances. The most common modeling applications
involve predictions based on information derived from animals (across
species extrapolations) or exposure routes (across route extrapolations)
other than those being studied.
The current CompTox Lab library of PBPK models includes the following chemicals:
- Trichloroethylene
- Carbon tetrachloride
- Dichloroethylene
- Methylene chloride
- Benzene
- Dioxin
- PCBs
- Lead
- Methyl mercury
- Chlorpyrifos
- Parathion
- Tetrachloroethylene
- Chloroform
- Vinyl chloride
- Acetone
QSAR techniques are used to estimate
the toxicity of poorly characterized substances based on comparisons to well-studied substances having similar chemical structures. TOPKAT, a commercially
available software, is used to predict toxicity endpoints based on chemical structure. The software predicts carcinogenicity in
male and female mice and rats, developmental toxicity, mutagenicity, log octanol/water partition coefficients, oral LD50s, and oral chronic
LOAELs in rats.
techniques are used to estimate
the toxicity of poorly characterized substances based on comparisons to well-studied substances having similar chemical structures. TOPKAT, a commercially
available software, is used to predict toxicity endpoints based on chemical structure. The software predicts carcinogenicity in
male and female mice and rats, developmental toxicity, mutagenicity, log octanol/water partition coefficients, oral LD50s, and oral chronic
LOAELs in rats.
Benchmark dose (BMD)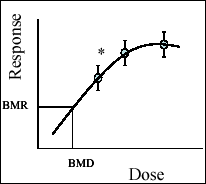 modeling is an
approach in which the dose response is modeled and the lower confidence
bound for a dose at a specified response level is calculated. BMD
is used to derive health guidance values by estimating the dose of a toxic substance without appreciable
risk or adverse health outcome. This is an alternative method to
the traditional threshold model approach (NOAEL/LOAEL) that is commonly
used for non-carcinogenic risk assessment of hazardous substances.
modeling is an
approach in which the dose response is modeled and the lower confidence
bound for a dose at a specified response level is calculated. BMD
is used to derive health guidance values by estimating the dose of a toxic substance without appreciable
risk or adverse health outcome. This is an alternative method to
the traditional threshold model approach (NOAEL/LOAEL) that is commonly
used for non-carcinogenic risk assessment of hazardous substances.
Emergency response and Bioterrorism
- QSAR is currently being conducted on a subset of chemicals from the CDC/ATSDR Chemical Terrorism List. Preliminary results suggest QSAR may be useful for providing limited data for use in acute exposure scenarios for chemical terrorism substances without available toxicity data. We are investigating the potential use of this data for establishing reoccupancy levels.
- In response to an urgent congressional request for information on the toxicity of 2-chloro-6-fluorophenol (one of the chemicals spilled at a chemical factory in New York), QSAR analysis conducted in the CompTox Lab predicted the chemical to be a mild skin sensitizer but noncarcinogenic and not a developmental toxicant. These results were provided to the New York State Department of Health (DOH), Bureau of Toxic Substance Assessment and follow-up consultations ensued.
- Based on a QSAR analysis performed by CompTox Lab scientists, the National Toxicology Program (NTP) initiated a chronic carcinogenicity study of a styrene-acrylonitrile trimer, a drinking water contaminant in the Tom’s River area.
- International collaborations with TNO Nutrition and Food Research,Netherlands continue using PBPK/PD models developed in the lab to investigate the mechanisms of interaction between trichloroethylene, benzene, lead, and methyl mercury.
- A PBPK/PD model was developed to predict the joint toxicity between chlorpyrifos and parathion in the rat. The model simulations indicated that additivity is obtained at oral levels below 0.08 mg/kg of each chemical.
- CompTox Lab scientists are collaborating with researchers at University of Georgia to develop mechanistically based PBPK models to investigate the interaction between perchlorate and PCBs.
- CompTox Lab faculty developed PBPK models for
 36
PCB congeners. Models were used to assess the impact of contaminated
fish consumption on PCB blood levels in Anniston, Alabama. Results
show that the contribution of consumption of contaminated fish
to PCB blood levels is congener specific.
36
PCB congeners. Models were used to assess the impact of contaminated
fish consumption on PCB blood levels in Anniston, Alabama. Results
show that the contribution of consumption of contaminated fish
to PCB blood levels is congener specific.
- Child-based PBPK models for methylene chloride and tetrachloroethylene were developed to determine if age-specific groups are more sensitive to chemical exposures than adults. Thus far, results of the modeling efforts show that neonates are 3 to 10-fold more susceptible to chemical toxicity via inhalation and oral routes than adults exposed to identical environmental conditions. Model validation data is needed to complete these models.
- Support ATSDR’s mission by using state-of-the-art decision-support tools for toxicologic evaluation of chemicals and their mixtures.
- Provide a scientifically credible, timely, and cost-effective alternative to traditional toxicology testing.
For more information, contact
Agency for Toxic Substances and Disease Registry
Division of Toxicology and Environmental Medicine
1600 Clifton Road NE, Mailstop F-32
Atlanta, GA 30333
Phone: 1-800-CDC-INFO • 888-232-6348 (TTY)
Email: cdcinfo@cdc.gov


