

| Agenda | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Click here to download a PDF version of the Agenda for this meeting. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *This file is in PDF format, which requires the free Adobe Acrobat Reader for viewing. Attention visually impaired visitors: To use common screen reading programs with PDF documents, please visit http://access.adobe.com. This site provides free tools that convert PDF documents to simple HTML or ASCII text. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
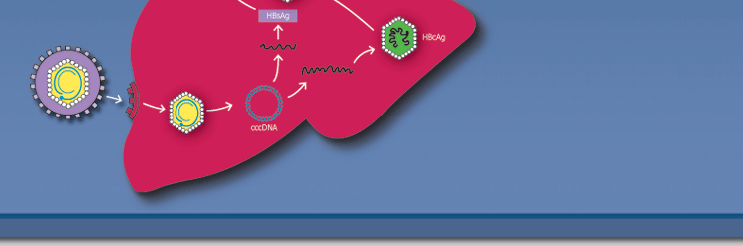
| Aims of the meeting: To assess current
understanding of hepatitis B virus (HBV), the disease that
it causes, and its optimal management; and to make
recommendations for directions for future research, both
basic and clinical. Topics will include HBV replication and
mode of inducing chronicity, the immunology of hepatitis B,
the virus-host interactions, the natural history of chronic
infection, and up-to-date results of trials of antiviral
therapies, including peginterferon, la.m.ivudine, adefovir
dipivoxil, tenofovir, entecavir, and LdT alone and in
combination. Histological, clinical, and virological
endpoints of therapy and the needs for future research will
be discussed. The meeting will be sponsored by the Liver
Disease Research Branch of the Division of Digestive
Diseases and Nutrition of NIDDK in collaboration with the
A.m.erican Association for the Study of Liver Disease (AASLD),
the A.m.erican Liver Foundation, the Hepatitis B Foundation
and Hepatitis Foundation International. A summary of the
meeting and these recommendations will be submitted for
publication. In preparation for the meeting, the following documents will be prepared: 1. Standardized definitions of response and endpoints of therapy. 2. Guidelines for the format of presentation of results of clinical trials. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Return to NIDDK homepage. This page last updated: 4/25/2006 |
 |