| |
| Handling
Inappropriate Vaccine Storage Conditions (Light and Temperature) |
 Immediate action
must be taken to correct improper vaccine storage conditions, including
inappropriate exposure to light and inappropriate exposure to storage
temperatures outside the recommended ranges. Immediate action
must be taken to correct improper vaccine storage conditions, including
inappropriate exposure to light and inappropriate exposure to storage
temperatures outside the recommended ranges.

 Immediate action must be taken to correct improper vaccine storage
conditions. Immediate action must be taken to correct improper vaccine storage
conditions. |
Furthermore, this action should
be documented. In your documentation, state what the problem is, what
has been done to protect the vaccines, what has been done to correct the
problem, and whether or not the problem has been corrected. You may use
the back of the temperature log to record this information. If you become
aware of inappropriate vaccine storage conditions, the following steps
should be taken:
|
1. |
Notify
the primary or backup vaccine coordinator immediately of any vaccine
storage unit temperatures that are outside the recommended range.
If the primary coordinator or the backup
person is not available, report the problem to an immediate supervisor. |

Notify the primary or backup vaccine coordinator immediately
of any vaccine storage unit temperatures that are outside the
recommended range.
|
2. |
Record the
room temperature and the temperature inside the refrigerator and freezer
at the time the problem is discovered. Also note the minimum and maximum temperature
readings if you have minimum/maximum thermometers in the refrigerator
and freezer. Record the length of time the vaccine may have been exposed
to inappropriate storage temperatures. |
| 3. |
Conduct an
inventory of the vaccines affected by this event and record the actions
taken. Also note if water bottles were in the refrigerator and frozen
packs in the freezer at the time of this event. You may use the Emergency
Response Worksheet in the Resources section of this toolkit to help
organize your response. Consult your agency, local health department,
or state health department immunization program, as appropriate for
your situation, for any special instructions or forms. |
| 4. |
Isolate the
affected vaccine vials or packages and mark them as "DO NOT USE."
This will reduce risk of inadvertently using vaccine that may have
lost its potency because it was stored under inappropriate conditions. |

Isolate the affected vaccine vials or packages and mark
them as "DO NOT USE."
|
| 5. |
Store the potentially
compromised vaccines under
appropriate conditions in a properly functioning vaccine storage unit
until the integrity of the vaccine is determined. If your vaccine
storage unit is not maintaining the appropriate storage conditions,
activate the Emergency Vaccine Retrieval and Storage Plan (see section on Storage and Handling Plans). |
6. |
Contact the
vaccine manufacturer and the state health department immunization
program for further guidance. Do not assume that vaccine inappropriately
exposed to light or to excessive temperatures cannot be salvaged. |
7. |
Finally, if HPV, MMR,
MMRV, rotavirus, varicella, and/or zoster vaccines have been exposed
to light, return the vaccine to a dark environment at the appropriate
storage temperature and record the length of time the vaccine may
have been exposed. Again, contact the vaccine manufacturer and the
state health department immunization program for further guidance. |
If vaccines have been exposed
to inappropriate storage temperatures because of a fault in the refrigerator
or freezer, follow the directions above and see Handling Malfunctioning
Vaccine Storage Units in this section for further details.
| Handling
Malfunctioning Vaccine Storage Units |
General
Instructions
The vaccine storage unit is
not working properly if any of the following situations occur:
| The most important step to take if the vaccine storage unit is not working properly is to protect the vaccine supply. |
The most important step to
take if the vaccine storage unit is not working properly is to protect
the vaccine supply. Do not allow the vaccine to remain in a nonfunctioning
unit for an extended period of time while you
attempt to correct the problem. If at any time you are unsure how
long the storage unit will not be functioning properly or you determine
that the problem cannot be corrected in time to maintain the internal temperature within the recommended range, activate the Emergency Vaccine Retrieval and Storage Plan (see section on Storage and Handling Plans).
The problem-solving flow charts
provided in this section may be used to
identify and correct vaccine storage unit problems. Follow these instructions
when using the problem-solving flow charts:
| 1. |
Record the
room temperature and the temperatures inside the refrigerator and
freezer when the problem is discovered. Also conduct an inventory
of all vaccines affected by the event (see Handling Inappropriate
Vaccine Storage Conditions [Light and Temperature] in this section). |
| 2. |
Always start
with the first problem shown in the problem-solving flow chart. |
| 3. |
Make sure
that a problem does not exist before moving on to the next step. |
| 4. |
If the storage
unit is still not working properly after completing all the steps
in the flow chart, |
| |
a. |
call a technician
to examine the faulty equipment; and |
| |
b. |
if you have not yet done
so, transfer the vaccine into another functioning storage unit that
has enough space to store the vaccines properly (see Emergency Vaccine Retrieval and Storage Plan in the Storage and Handling Plans section). |
| 5. |
Record in
the vaccine storage unit logbook all the checks you made and the actions
taken (see Equipment
Logbooks in the Vaccine Storage Equipment section for details).
This will help the technician identify the fault with the storage
unit. |
Vaccine
Storage Unit is Too Warm
Warning! Do not allow the vaccine
to remain in a nonfunctioning unit for an extended period of time while
you attempt to correct the problem. If at any time you are unsure how
long the storage unit will not be functioning properly or you determine
that the problem cannot be corrected in time to maintain the internal
temperature within the recommended range, activate the Emergency Vaccine Retrieval and Storage Plan (see section on Storage and Handling Plans).
Vaccine
Storage Unit is Too Cold
Warning! Do not allow the vaccine
to remain in a nonfunctioning unit for an extended period of time while
you attempt to correct the problem. If at any time you are unsure how
long the storage unit will not be functioning properly or you determine
that the problem cannot be corrected in time to maintain the internal
temperature within the recommended range, activate the Emergency Vaccine Retrieval and Storage Plan (see section on Storage and Handling Plans).
Vaccine
Storage Unit is Too Noisy
Warning! Do not allow the vaccine
to remain in a nonfunctioning unit for an extended period of time while
you attempt to correct the problem. If at any time you are unsure how
long the storage unit will not be functioning properly or you determine
that the problem cannot be corrected in time to maintain the internal
temperature within the recommended range, activate the Emergency Vaccine Retrieval and Storage Plan (see section on Storage and Handling Plans).
Vaccine
Storage Unit Has Stopped
Warning! Do not allow the vaccine
to remain in a nonfunctioning unit for an extended period of time while
you attempt to correct the problem. If at any time you are unsure how
long the storage unit will not be functioning properly or you determine
that the problem cannot be corrected in time to maintain the internal
temperature within the recommended range, activate the Emergency Vaccine Retrieval and Storage Plan (see section on Storage and Handling Plans).
| Refrigerator
and Freezer Door Problems |
Checking
the Door Seal
To check that the vaccine storage
unit door is sealing properly:
| 1. |
Place a thin
paper strip against the cabinet front (see Illustration 1). |
| 2. |
Close the door. |
| 3. |
Pull the paper strip.
If it moves easily or falls away by itself, the door and the rubber-like
seal need to be adjusted. |
| 4. |
Check all the way around
the door. Pay particular attention to the corners. |

Illustration 1—Checking the door seal.
(Adapted from the User’s
Handbook for Compression Refrigerators WHO/EPI/LOG/84/15)
|
Adjusting
the Door Seal
If you have checked the rubber-like
door seal and determined that the door is
not closing properly:
| 1. |
Loosen the
hinge screws on the door slightly and check if the door seals better
after the hinges are adjusted (see Illustration 2). |
| |
a. |
If the seal
on the handle side of the door is loose, move the hinges slightly
outward. |
| |
b. |
If the seal on the hinge
side of the door is loose, move the hinges slightly inward. |
| 2. |
If the door
seal is loose at the corner, it will need to be replaced. Call a trained
technician. |
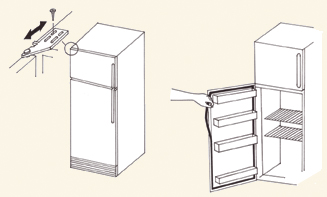
Illustration 2—Adjusting the door seal.
(Adapted from the User’s Handbook for Compression Refrigerators WHO/EPI/LOG/84/15) |
Adjusting
Dropped Doors
If the vaccine storage unit
door is not level when closed or if it is touching the toe kick plate
or another door on the unit, it requires adjustment. Dropped doors can
compromise the door seal. To adjust a dropped door:
| 1. |
Loosen the
upper hinge screws. |
| 2. |
Make sure
the edge of the door is in line with the side of the refrigerator. |
| 3. |
Hold the door
in position and tighten the screws. |
| |
a. |
If the lower
hinge is worn, the door can be adjusted upward by putting some washers
on the hinge. (see Illustration 3) |
| 4. |
Check the
door seal to be sure it is closing properly and tightly. |
| 5. |
If a dropped
door cannot be adjusted, call a trained technician. |
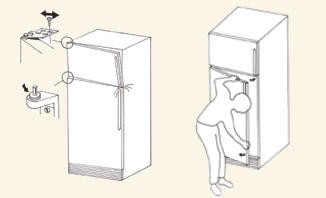
Illustration 3—Adjusting dropped doors.
(Adapted from the User’s Handbook for Compression Refrigerators WHO/EPI/LOG/84/15) |
Checking
Thermometer Placement
If the thermometer indicates
a temperature outside the recommended range, check that the thermometer
is appropriately situated in the center of the storage unit compartment,
adjacent to the vaccine. If the thermometer is placed near the coils,
walls, floor, or fan, it may indicate colder or warmer temperatures than
a thermometer appropriately placed in the center of the compartment where
the vaccines should be kept.
Checking
If the Thermometer Works
A slight variation in temperature
is often seen from one thermometer reading to another, even when the vaccine
storage unit thermostat is set at a particular temperature. This is normal.
If the thermometer reading does not fluctuate at all over several readings,
temporarily remove the thermometer from the storage unit and place it
outside the unit at room temperature. Check whether the temperature reading
rises. If no change in the temperature reading occurs, the thermometer
is faulty and needs to be replaced.
Checking
If the Thermometer Is Accurate
If the thermometer appears
to be working but there is concern regarding the accuracy of the reading,
the standard method of testing the thermometer is to place another certified
calibrated thermometer inside the storage unit along with the original
one and check the readings on both thermometers.
Advance
Preparations
| When state officials, local officials, or providers have reasonable cause to believe that a power outage may occur, emergency procedures should be implemented in advance of the event. |
When state officials, local officials, or providers have reasonable cause to believe that a power outage may occur (e.g., adverse weather conditions,
natural disasters, or other mergencies that might disrupt power to
any office where vaccine is stored) emergency procedures should be
implemented in advance of the event.
Temperature
Considerations
Most refrigerated vaccines
will remain stable at elevated temperatures for limited periods of time.
The vaccines of most concern are MMR, MMRV, varicella, and zoster vaccines,
which are more sensitive to elevated temperatures. Whenever a question
arises about the integrity of a vaccine, contact the vaccine manufacturer
and the state health department immunization program for guidance (see
Handling Inappropriate Vaccine Storage Conditions
[Light and Temperature] in this section for details).
Power
Outage Procedures
The information below is provided
as a guideline. You may use the Emergency Response Worksheet in the Resources
section of this toolkit to help organize your response. Consult your agency,
local health department, or state health department immunization program,
as appropriate for your situation, for any special instructions or forms. If there is an ongoing
power outage, take the following steps:
| 1. |
Do not allow
the vaccine to remain in a nonfunctioning unit for an extended period
of time. If at any time you are unsure how long the power to the vaccine
storage unit will be interrupted or you determine that the power will
not be restored in time to maintain the internal temperature within
the recommended range, activate the Emergency Vaccine Retrieval and Storage Plan (see section on Storage and Handling Plans) and disregard the
following steps. |
| 2. |
If you are
certain the power will be restored before the vaccine storage unit
temperature rises above the recommended range, take the following
steps: |
| |
a. |
Do not open
the refrigerator or freezer door until the power is restored. |
| |
b. |
Continue to
monitor the temperatures inside the vaccine storage unit. |
| |
|
i. |
Some thermometers
allow temperature monitoring without opening the storage unit doors.
In this case, record the room temperature and the temperature(s) inside
the unit(s) at the time the problem is discovered, as well as the
minimum and maximum temperatures reached inside the unit(s) during
the power outage. |
| |
|
ii. |
If these types of thermometers
are not being used, do not open the door(s) to check the temperature(s)
during the power outage. Instead, record the room temperature, the
duration of the power outage, and the temperature(s) inside the unit(s)
as soon as possible after the power is restored. This will provide
data on the maximum temperature and
maximum duration of exposure to elevated temperatures. |
| |
c. |
Record the
room temperature and the temperatures inside the vaccine storage units
as soon as possible after power has been restored.
Record the length of time the power has been off and the maximum temperature
observed. |
| |
d. |
If the temperature
inside the refrigerator has exceeded the recommended range of 35°
to 46°F (2° to 8°C) or if the temperature inside the freezer
has risen above 5°F (-15°C), record the duration of inappropriate
temperature exposure and follow the procedures for Handling Inappropriate
Vaccine Storage Conditions [Light and Temperature] in this section. |
The Center for Biologics Evaluation
and Research (CBER) at the Food and Drug Administration (FDA) offers general
guidance concerning the storage and use of temperature-sensitive biological
products that have been involved in a temporary electrical power failure
or flood conditions (see Emergency
Management Internet Resources in the Resources section of this toolkit).
| Other
Imminent Emergencies |
When state officials, local
officials, or providers have reasonable cause to believe that weather
conditions, natural disasters, or other imminent emergencies might disrupt
power or flood any office where vaccine is stored, emergency procedures
should be implemented in advance of the event (see Emergency Vaccine Retrieval and Storage Plan in the Storage and Handling Plans section).
|
