The Office of Basic Energy Sciences
(BES) has been at the
forefront of scientific discovery for several decades. BES staff and
colleagues in March 2001 identified 28 of the more important discoveries
supported by the program. Short descriptions of these
major accomplishments are provided below. See the Office of
Science (SC) Decades
of Discovery for 73 descriptions of major
achievements from other SC programs.
|
  |
|
|
  |
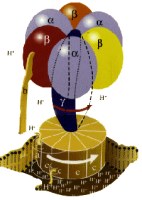
Adenosine Triphosphate: The Energy Currency of Life
|
 |
|
Enzymatic
mechanism
of ATP synthesis
|
The energy cycle of
all living organisms involves the molecule adenosine
triphosphate (ATP), which captures the chemical
energy released by the metabolism of nutrients and
makes it available for cellular functions such as
muscle contraction and transmission of nerve
messages. A hard-working human adult can convert
almost a ton of ATP daily. From the early 1960s
through 1994, the Office of Science supported Paul
D. Boyer's research at the University of California
at Los Angeles on ATP synthase, the enzyme
responsible for synthesizing ATP. His research
examined the detailed chemical reactions involved in
ATP synthesis and how the enzyme uses energy to
create new ATP. Boyer theorized that this
"molecular machine" with rotating parts
functions in a surprising way for enzymes, a
mechanism later supported by the work of John E.
Walker of the United Kingdom. Among other things,
Boyer discovered that energy input was not used
primarily to form the ATP molecule, but rather to
promote the release of an already formed and tightly
bound ATP. Boyer and Walker shared half of the 1997
Nobel Prize in Chemistry for these achievements.
Scientific
Impact: This work uncovered new concepts in
enzymology and advanced understanding of how living
cells function at the molecular level. Determination
of how cells store and transfer energy has been
among the most important advances in molecular and
cell biology, enabling an entire generation of work
at the cellular level in animal and plant research.
Social
Impact: Research in cell biology has led to
tremendous advances in medicine and physiology, such
as clues to the genesis and treatment of cancer.
Reference:
Boyer, P.D., "The ATP synthase—a splendid
molecular machine," Ann Rev Biochem
997;66:717-49
|
|
 |
Making Better Catalysts
|
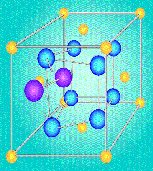 |
| Model
for Ceria Oxide System. Ceria atoms are
yellow, oxygen atoms are blue, and the
active oxygen at the defects are purple. |
Catalysts, which
accelerate chemical reactions, are valuable in many
industries, from fuels to pharmaceuticals. Long-term
research by government and academic scientists
supported by the Office of Science has led to new
understanding of catalytic phenomena, in particular
the relationship between chemical structure and
reactivity. For example, early work established two
classes of heterogenous catalysts (which function by
adsorbing molecules), based on whether chemical
reactivity is, or is not, sensitive to surface
structure. These studies showed that catalytic
reactions once thought to be structurally
insensitive actually took place on a dynamic
surface. Research on the reactivity of hydrogen with
catalysts—an issue in the world's largest-scale
industrial processes, such as sulfur removal from
crude oil-disproved the widely held belief that
hydrogen molecules must dissociate into two atoms
before undergoing reactions, and challenged the
accepted notion that surface-bound (as opposed to
embedded) hydrogen was the only reactive form. Other
discoveries concerned the chemical behavior of
organometallic complexes (combinations of organic
and metallic species) that are used, for example, in
plastics manufacturing.
Scientific
Impact: Research on structure-reactivity
relationships has increased understanding of both
natural and synthetic processes. The discovery of
nonclassical binding of molecular hydrogen created a
new field of study that may overcome some of
chemistry's greatest challenges, such as conversion
of natural gas to more usable liquid fuels (methanol
or gasoline).
Social
Impact: A modern society's standard of
living can be measured by its accomplishments in
catalysis, because every manufacturing process and
energy-generating technique starts with catalysis.
Catalysts first introduced by investigators
supported by the Office of Science revolutionized a
process used to make about 100 billion pounds of
plastics per year worldwide; this work is leading to
catalysts that produce superior plastics with new
properties.
Reference:
C.E. Tripa and J.T. Yates, Jr. Nature, 398 (1999)
591.
|
|
 |
Understanding Chemical Reactions
|
 |
|
The molecular
features that influence the rate of chemical
reactions were poorly understood until the
mid-1960s, when Dudley Herschbach and his
postdoctoral student Yuan T. Lee began a series of
experiments at Harvard University. With funding from
the Office of Science and predecessor agencies, they
explained in detail how chemical reactions take
place, and solved the problem of how to observe the
random directions and velocities of molecules in a
gas or a liquid. They developed an apparatus in
which crossed molecular beams were used to vary the
velocities and approach angles of reacting
molecules. With this tool, the scientists discovered
and studied long-lived complexes of reactants formed
before a reaction was completed, and described
theoretically their formation and decay. They also
examined the velocities of products, rotational
energies, and internal vibration energies. In this
way, they could map out all the details of a
chemical reaction and explain the effects of
temperature and pressure. Initially, these studies
focused on reactions between alkali atoms and other
molecules; Lee later adapted the crossed molecular
beam method for general reactions. The 1986 Nobel
Prize in Chemistry was awarded jointly to
Herschbach, Lee, and a third scientist.
Scientific
Impact: This work contributed significantly
to the modern knowledge base for atmospheric and
combustion chemistry. These scientists helped
establish reaction dynamics as a discrete field of
research, and their crossed molecular beam approach
is among the most important contributions to this
field.
Social
Impact: These studies contributed to
improvements in industrial production efficiency and
assisted in the design of new products to be more
useful, durable, and conserving of natural
resources. This work also contributed to the
development of predictive theories and models used
to design and manufacture new products ranging from
plastics to pharmaceuticals.
Reference:
http://nobelprize.org/nobel_prizes/chemistry/laureates/1986/index.html
|
|
 |
New Types of Superconductors
|
 |
| Organic
superconductor with the highest Tc
k-(ET)2Cu[N(CN)2]Cl |
Superconductors
conduct electricity with little or no resistance.
Organic superconductors contain carbon and are less
dense than their ceramic or metallic counterparts;
they also offer unusual potential for fine-tuning of
electrical properties. Argonne National Laboratory
long has carried out the major U.S. effort to
synthesize and identify organic superconductors.
Nearly 100 new superconductors of this type have
been produced, with critical temperatures (at which
a superconductor loses all electrical resistance) as
high as -260 degrees C, or -434 degrees F. Recently,
the first superconductor composed entirely of
organic components (with no metal atoms) was
synthesized, with a transition temperature in this
range. Although this remains far lower than the
highest known transition temperature for ceramics,
scientists still expect that a high-temperature
organic superconductor may be possible, such that
liquid nitrogen (at -196 degrees C, or -321 degrees
F) could be used as the coolant instead of the more
costly liquid helium, thus making practical
applications more feasible. The new compound has a
two-dimensional, layered structure, which may
provide significant insight into the nature of
superconductivity.
Scientific
Impact: These advances will help scientists
develop a theory of how organic superconductors work
and contribute to the design of new materials with
higher transition temperatures. The all-organic
material is ideal for studies of magnetic and charge
transport properties because there is no possibility
of contamination from metallic impurities.
Social
Impact: Superconductivity already has
important applications, such as medical diagnostic
equipment, and many more uses are possible if
transition temperatures are high enough. The
availability of purely organic superconductors
greatly expands the possibilities, especially for
applications in which weight is a factor.
Reference:
Ambient-Pressure Superconductivity at 2.7 K and
Higher Temperatures in Derivatives of beta(ET)2IBr2:
Synthesis, Structure, and Detection of
Superconductivity. Williams, J. M.; Wang, H. H.;
Beno, M. A.; Emge, T. J.; Sowa, L. M.; Copps, P. T.;
Behroozi, F.; Hall, L. N.; Carlson, K. D.; Crabtree,
G. W. Inorg. Chem. 1984, 23, 3839-3841.
A New Ambient-Pressure Organic Superconductor, kappa
(ET)2Cu[N(CN)2Br, with the
Highest Transition Temperature Yet Observed (
Inductive Onset Tc=11.6 K, Resistive Onset=12.5 K)
Kini, A. M.; Geiser, U.; Wang, H. H.; Carlson, K.
D.; Williams, J. M.; Kwok, W. K.; Vandervoort, K.
G.; Thompson, J. E.; Stupka, D. L.; Jung, D.;
Whangbo, M.-H. Inorg. Chem. 1990, 29,
2555-2557.
From Semiconductor-Semiconductor Transition (42 K)
to the Highest-Tc Organic Superconductor,
kappa (ET)2Cu[N(CN)2Cl (Tc=12.5
K) Williams, J. M.; Kini, A. M.; Wang, H. H.;
Carlson, K. D.; Geiser, U.; Montgomery, L. K.;
Pyrka, G. J.; Watkins, D. M.; Kommers, J. M.;
Boryschuk, S. J.; Strieby Crouch, A. V.; Kwok, W.
K.; Schirber, J. E.; Overmyer, D. L.; Jung, D.;
Whangbo, M.-H. Inorg. Chem. 1990, 29,
3272-3274.
The First Organic Cation-radical Salt Superconductor
(Tc=4 K) with an Organometallic Anion:
Superconductivity, Synthesis and Structure of kappa
(ET)2M(CF3)4(C2H3X3).
Schlueter, J. A.; Geiser, U.; Williams, J. M.; Wang,
H. H.; Kwok, W. K.;Fendrich, J. A.; Carlson, K. D.;
Achenbach, C. A.; Dudek, J. D.; Naumann, D.; Roy,
T.; Schirber, J. E.; Bayless, W. R. J. Chem.
Soc., Chem. Commun. 1994, 1599-1600.
Superconductivity at 5.2 K in an Electron Donor
Radical Salt of Bis (ethylenedithio)
tetrathiafulvalene (BEDT-TTF) with the Novel
Polyfluorinated Organic Anion beta (ET)2SF5CH2CF2SO3)
Geiser, U.; Schlueter, J. A.; Wang, H. H.; Kini, A.
M.; Williams, J. M.; Sche, P. P.; Zakowicz, H. I.;
VanZile, M. L.; Dudek, J. D.; Nixon, P. G.; Winter,
R.W.; Gard, G. L.; Ren, J.; Whangbo, M.-H. J.
Am. Chem. Soc. 1996, 118, 9996-9997.
|
 |
Development of
Neutron Scattering Facilities
|
 |
| Powder
Diffractometer High Flux Isotope Reactor
(ORNL) |
Neutron scattering
provides key information on the positions, motions,
and magnetic properties of solids. When neutrons
flowing from a nuclear reactor bounce off atoms in a
sample, the neutrons scatter in directions that
depend on the atoms' relative positions in the
sample structure. Changes in the neutrons' velocity
provide information on the atoms' oscillations, or
dynamics. Since the late 1940s, the Office of
Science and predecessors have been major supporters
of neuron science, including work by Clifford Shull
and Bertram Brockhouse, who shared the 1994 Nobel
Prize in Physics for their development of neutron
scattering techniques for studies of condensed
matter. Researchers at Oak Ridge, Brookhaven, and
Argonne national laboratories developed neutron
sources for spectroscopy, scattering, and imaging
experiments and helped pioneer most of the
associated instruments and techniques. The Office of
Science currently supports three neutron
sources—the High-Flux Isotope Reactor at Oak Ridge
National Laboratory, Intense Pulsed Neutron Source
at Argonne National Laboratory, and Manuel Lujan Jr.
Neutron Scattering Facility at Los Alamos National
Laboratory—used by hundreds of researchers
annually. Under construction is a spallation neutron
source at Oak Ridge that will be about an order of
magnitude more powerful than any existing pulsed
neutron source. Spallation produces neutrons with
little heat; pulsed operation provides very high
peak intensities.
Scientific
Impact: Neutrons' unique properties, such
as sensitivity to light elements, make them
invaluable tools for polymer, biological, and
pharmaceutical sciences. Studies made possible by
neutron sources and the associated techniques
contribute to the development of new materials, such
as ceramic superconductors.
Social
Impact: Neutron studies lead to new and
improved products, such as powerful magnets for
highly efficient electric motors. Also, because
their high penetrating power allows nondestructive
property measurements deep within a specimen,
neutrons have been used to examine automotive gears
and brake discs, and defects in aircraft wings,
engines, and turbine blades.
Reference:
Scientific Research Facilities: A National Resource,
Office of Basic Energy Sciences, http://www.sc.doe.gov/production/bes/Brochures.htm
URL:
http://www.sc.doe.gov/bes/BESfacilities.htm#Neu
|
|
 |
Development
of Synchrotron Radiation Light Sources
|
 |
| Example
shown is Si 111 (top) and Si 100 (bottom) |
Synchrotrons produce
a unique type of radiation—continuous across the
spectrum and tunable to the desired
wavelength—emitted by electrons accelerated in a
magnetic field. For two decades, the Office of
Science has been the major supporter of U.S.
synchrotron light sources. It currently operates
four, each with unique capabilities, used by a total
of more than 6,000 researchers annually from
academia, government, and industry. The four are the
Advanced Light Source at Lawrence Berkeley National
Laboratory, Advanced Photon Source at Argonne
National Laboratory, National Synchrotron Light
Source at Brookhaven National Laboratory, and
Stanford Synchrotron Radiation Laboratory at
Stanford Linear Accelerator Center. Scientists at
these sites helped pioneer many synchrotron
innovations that are widely used today, including a
lattice of magnets that increased brightness (photon
density) by two orders of magnitude;
"insertion" devices (linear arrays of
magnets called wigglers and undulators) that
oscillate the path of the electron beam to generate
X-ray and ultraviolet light that is high in flux
(number of photons) and collimation (parallel
alignment of photons); and powerful experimental
techniques such as X-ray scattering and X-ray
microscopy.
Scientific
Impact: These innovations made new science
possible and paved the way for significant
extensions of light source performance that have had
a broad and deep impact on the understanding of
matter. Synchrotrons are used for cutting-edge
research in materials science, physical and chemical
science, geosciences, environmental science,
bioscience, and medical and pharmaceutical science.
Social
Impact: Synchrotron research affects
society in areas such as information and energy
technologies. For example, recent high-resolution
imaging of thin films of copper may assist in the
development of ultrahigh-density computer hard
drives, and imaging of contaminants in solar cells
and their removal by heat treatment may lead to more
efficient and less costly solar energy.
Reference:
Scientific Research Facilities: A National Resource,
Office of Basic Energy Sciences, http://www.sc.doe.gov/production/bes/Brochures.htm
P. A. Montano and H. Oyanagi, "In Situ
Synchrotron Radiation research in Materials
Science," MRS Bulletin, (24) 13-20
(January 1999).
W.Yun et al., "S-ray Imaging and
Microspectroscopy of Plants and Fungi," J.
Synchrotron Rad., (5) 1390-1395 (1998).
URL:
http://www.sc.doe.gov/bes/BESfacilities.htm#Sync
|
|
 |
Development
of Lithium Batteries
|
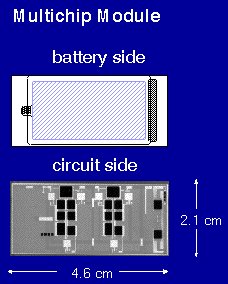 |
| A
thin-film Li-LiCoO2 battery was fabricated
onto the back side of the ceramic package of
a multichip module. Contact with the circuit
on the front side was made by depositing the
cathode and anode current collectors over
gold-plated through-holes. The battery was
designed to supply 150 µAh between 4.2 and
3.8 V at a low current. |
Lithium batteries,
which offer both high energy-storage capacity and an
environmentally benign alternative to the harmful
lead used in conventional batteries, are based on
research supported by the Office of Science and its
predecessors. An early innovation was the
development of organic solid
electrolytes—essential because traditional
water-based electrolytes could react with metals
such as lithium to cause an explosion. (A battery
consists of positive and negative electrodes
separated by an electrolyte, through which ions, or
charged atoms, flow.) Charles Tobias at Lawrence
Berkeley National Laboratory led the search for
nonaqueous solutions from which reactive metals,
such as lithium (then used in fusion-type nuclear
weapons), could be electrolytically deposited. He
focused on cylic esters, including propylene
carbonate, which today is used extensively in
battery technology. The pioneering research included
the purification of solvents to dissolve the
electrolyte, solubility and conductivity
measurements, and decomposition and
electrodeposition tests. The Office of Science
currently supports research on ion transport in
solid polymer and glassy electrolyte systems,
helping to lay the groundwork for the next
generation of highly efficient and environmentally
friendly batteries and fuel cells.
Scientific
Impact: Tobias is widely regarded as the
father of electrochemical engineering because he
introduced scientific methods into a field formerly
characterized by trial and error. His initial
characterization of nonaqueous electrolytes, and
demonstration that reactive metals could be
electrodeposited from them, spawned a new field of
battery research.
Social
Impact: Lithium batteries are widely used
in both consumer and defense applications, such as
cellular telephones and notebook computers, but such
batteries remain expensive. DOE applied research
programs are developing new and less costly versions
of rechargeable lithium batteries for use in
electric and hybrid vehicles.
Reference:
Dudney NJ, Bates JB, Lubben D, "Thin-Film
Rechargeable Lithium Batteries," in Role of
Ceramics in Advanced Electrochemical Systems.
American Ceramic Society, 1996 p. 113.
|
|
 |
A New Class of
Carbon Structures
|
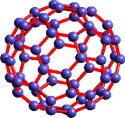 |
| A
new allotrope of carbon that consists of 60
carbon atoms, shown above, in the shape of a
soccer ball. |
Several lines of
research—in spectroscopy, astronomy, and metallic
clusters—converged in 1985 to lead to the
discovery of an unusual molecule. This cluster of 60
carbon atoms was especially stable because of its
hollow, icosahedral structure in which the bonds
between the atoms resembled the patterns on a soccer
ball. The molecule was named Buckminsterfullerene
after the geodesic domes designed by architect
Buckminster Fuller. The identification of this form
of carbon (also called buckyballs) sparked broad
interest in the chemistry of an entire class of
hollow carbon structures, referred to collectively
as fullerenes. Formed when vaporized carbon
condenses in an atmosphere of inert gas, fullerenes
include a wide range of shapes and sizes, including
nanotubes of interest in electronics and hydrogen
storage. The initial discovery was recognized by the
1996 Nobel Prize in Chemistry, awarded to Richard E.
Smalley and Robert F. Curl, both supported by the
Office of Science, and Curl's colleague Sir Harold
W. Kroto of Great Britain. More recently, scientists
at Lawrence Berkeley National Laboratory reported a
new synthetic method for producing, extracting, and
purifying a cluster of 36 carbon atoms in quantities
useful for research purposes; they also confirmed
the high reactivity and other unusual electrical and
chemical properties of this material.
Scientific
Impact:
The discovery of fullerenes launched a new branch of
chemistry, and related studies have contributed to
growing interest in nanostructures in general and
the principles of self-assembly. Fullerenes also
have influenced the conception of diverse scientific
problems such as the galactic carbon cycle and
classical aromaticity, a keystone of theoretical
chemistry.
Social
Impact:
Fullerenes are highly versatile (there are literally
thousands of variations) and thus have many
potential applications. For example, fullerene
structures can be manipulated to produce
superconducting salts, new three-dimensional
polymers, new catalysts, and biologically active
compounds.
Reference:
"C60: Buckminsterfullerene," H.W. Kroto,
J.R. Heath, S.C. O'Brien, R.F. Curl, and R.E.
Smalley, Nature 318, 162, November 14,
1985"
|
|
 |
Designing
Organisms to Make Valuable Biomaterials
|
 |
|
Plants and microbes
are natural biochemical factories, producing
important chemicals and materials. (Petroleum
deposits are the altered remains of prehistoric
plants and microbes.) The Office of Science long has
supported basic studies on biochemistry and genetics
that are providing insights into how plants and
microbes can be modified to make more products with
economic value. Christopher Somerville, while at
DOE's Plant Research Laboratory at Michigan State
University, demonstrated the capability to transfer
an alignment of genes from bacteria to higher plants
that confer the ability to synthesize biodegradable
plastic components. He also studied the biosynthetic
pathways for plant oils to learn what genetic
changes would produce a different and more desirable
type of oil. Research by Lonnie Ingram at the
University of Florida focused on the regulation of
genes that play critical roles in a bacterium's
natural production of ethanol. He engineered DNA
with genes for making two key enzymes; not only did
this DNA alter the production pathway, but it also
was incorporated into the genetic material of
numerous other bacteria that did not normally form
ethanol—and they started to make it.
Scientific
Impact: Somerville's work represents an
early breakthrough in enhancing the use of plants as
biosynthesizers of precursors for biodegradable
plastics, which could replace products now derived
from petroleum. Ingram's research suggests the
potential for altering many bacteria, with many
potential growth substrates, to produce ethanol.
Social
Impact: Biosynthesis of compounds that can
replace petroleum-derived products could reduce U.S.
reliance on foreign oil. The University of Florida
patented an ethanol-producing organism capable of
growing on certain sugars, and an ethanol plant in
Louisiana is demonstrating the commercial potential
of a process based on this research.
Reference:
Buchanan, B.B., W.Gruissem, and R.L. Jones, Biochemistry
& Molecular Biology of Plants, American
Society of Plant Physiologists (aspp@aspp.org),
2001.
|
|
 |
Heavy
Element Chemistry
|
 |
|
For more than 50
years, the Office of Science and predecessor
agencies have supported the discovery and study of
the actinide elements, in particular the
transuranium elements-atoms that are heavier than
uranium. Glenn Seaborg and Ed McMillan of the
Lawrence Berkeley National Laboratory, 1951 Nobel
Laureates in Chemistry for the discovery of
plutonium and other actinide elements, began this
quest. Today, the Heavy Element Chemistry program
continues the pursuit for a fundamental
understanding of actinide and fission product
chemistry. The discovery and the exploration of the
properties of the transactinides, elements heavier
than the actinides, is also being undertaken and
presents significant challenges since these elements
decay to the lighter elements in minutes, seconds,
or milliseconds. One of the leading researchers in
this area is Darleane Hoffman of Lawrence Berkeley,
whose work earned her the National Medal of Science
in 1997 (the nation's highest scientific honor) and
the Priestly Medal of the American Chemical Society
in 2000. Hoffman contributed to the development of
"atom at a time" chemistry which makes
possible the study of heavy elements with half-lives
of a minute or less. She was among the researchers
to confirm the existence of the element seaborgium,
named after Seaborg. Hoffman now is involved in an
international collaboration to study the chemistry
of the transactinides, work inspired by predictions
of unexpected chemical properties caused by
relativistic effects.
Scientific
Impact: Research on the heavy elements
yields the basic knowledge that can be used to
develop new technologies and processes for the safe
handling and disposition of these radioactive
materials. For the transactinides, new "atom at
a time" chemical techniques are being used to
determine and compare their chemical properties to
other known elements. The Office of Science heavy
element chemistry program is the nation's sole
effort addressing the fundamental science of the
transuranium elements.
Social
Impact: This research helps DOE carry out
what is perhaps its most important and difficult
responsibility—stewardship of the nation's nuclear
science and technology. Studies of these elements
and their fission products are needed to address the
environmental consequences of the weapons programs
and possible accidental release of nuclear
materials.
Reference:
http://www.nobel.se/chemistry/laureates/1951/seaborg-bio.html
|
|
 |
Improving
Intermetallic Compounds
|
 |
|
Intermetallic
compounds (metallic materials composed of definite
proportions of two or more elemental metals) resist
oxidation and remain strong at high temperatures,
making them useful for energy technologies. But
until recently, these compounds were too brittle to
be fabricated into conventional shapes using
traditional methods. In 1981, Oak Ridge National
Laboratory started a program to increase
understanding of intermetallic compounds and develop
scientific principles for improving their
properties. Following a Japanese report suggesting
that small amounts of boron made a nickel aluminide
compound more ductile, Oak Ridge researchers led by
Chain T. Liu determined the mechanism behind the
change. They also showed that iron aluminides are
intrinsically ductile at ambient temperatures and
that brittleness is caused by moisture in the air.
Quantum mechanical calculations demonstrated a
mechanism that reduced the cohesive strength of
atomic layers in these alloys by 70 percent, a
discovery that led to new and improved alloy
designs. Liu was awarded the 2001 Acta
Metallurgica Gold Metal for his outstanding
leadership and achievements in this research. The
Office of Science then worked with DOE offices of
Energy Efficiency and Fossil Energy to fund a
research program on intermetallic compounds, an
effort that has won three R&D 100 awards from R&D
Magazine recognizing significant new
technologies, and has resulted in more than 16
patents and 12 licenses.
Scientific
Impact: Materials and processing research
at Oak Ridge has increased scientific understanding
of intermetallic compounds. This work overcame the
brittleness problem and improved manufacturability,
thus making it practical to use nickel and iron
aluminides for high temperature engineering
applications.
Social
Impact: This research has helped to improve
product quality and reduce costs. For instance, the
use of nickel-aluminide dies for the hot forging
process improves the quality of steel parts in
automobiles, and iron-aluminide filters used to
remove ash particles during coal gasification reduce
costs and resist the corrosiveness of hydrogen
sulfide in the gas stream.
Reference:
Pope, D. P., C, T. Liu, S. H. Whang, and M.
Yamaguchi, eds., High Temperature Intermetallics,
Elsevier, New York (1997).
|
|
 |
Ion Beam
Techniques Enhance Materials Science
|
 |
|
Since the 1940s, Oak
Ridge National Laboratory has played a leading role
in the development of ion beam technology and its
application in materials processing and
characterization. A key advance was made in the
early 1960s when, in one of the first applications
of computers in materials science, researchers
predicted that positive ions (charged atoms) moving
through a crystal would follow channels between the
rows of atoms, thereby penetrating well into the
crystal structure. The "ion channeling"
effect became the basis for valuable scientific and
commercial processes used to force ions into
materials as a means of tailoring or altering their
properties. One such process is ion implantation,
now developed into a fine art that relies on
accelerators to drive selected ions into materials
at precise distances. Many materials so modified are
now in routine use. Today, Oak Ridge operates a
facility where the broader scientific community
carries out fundamental research on various ion beam
techniques to selectively design the near-surface
properties of materials.
Scientific
Impact: Ion beam techniques are widely used
for research on topics such as superconductivity,
thin-film electrolytes, quasicrystals, and surface
structure and chemistry. The science continues to
evolve; new approaches to controlling the morphology
and properties of ion-implanted materials and layers
now are being developed based on defect physics.
Social
Impact: Ion implantation is used
extensively in the electronics industry to
"dope" semiconductors with special
properties, both chemically and spatially. The
process is also used to improve the wear resistance
of titanium alloys in artificial prostheses for hip
and knee replacements. By eliminating the need to
rework failed replacement joints, this technology
spares individuals from additional surgeries and
saves as much as $100 million per year.
Reference:
E. Chason, et al, "Ion beams in silicon
processing and characterization," Journal
of Applied Physics, vol. 81, no. 10, pp.
6513-6561 (1997) [Report of BES study panel]
A. Agarwal, H.-J. Gossmann, D. J. Eaglesham, S. B.
Herner, A. T. Fiory, and T. E. Haynes,
"Boron-enhanced diffusion of boron from
ultra-low energy ion implantation," Applied
Physics Letters vol. 74, pp. 2435-2437 (1999).
J. M. Williams and R. A. Buchanan, "Ion
implantation of surgical Ti-6Al-4V alloy," Materials
Science and Engineering vol. 69, pp. 237-246
(1985).
|
|
 |
Preventing
Radioactive Contamination
|
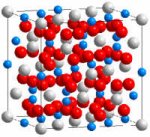 |
| Gadolinium
zirconate is a highly radiation-resistant
material that shows promise for use as a
durable storage material for immobilizing
plutonium and other actinides. The structure
of gadolinium zirconate (Gd2Zr2O7) above is
color coded: Blue=Gadolinium (Gd),
Gray=Zirconium (Zr), Red=Oxygen (O). |
Worldwide, nuclear
energy and weapons programs have created 1,350
metric tons of plutonium, an amount still growing by
70 metric tons annually. A major issue facing
society is how to dispose safely of plutonium, which
is radiotoxic and decays very slowly (it has a
half-life of 24,500 years). One strategy is to
immobilize it in chemically durable materials that
absorb harmful neutrons and resist radiation damage.
A 20-year collaboration between Rod Ewing at the
University of Michigan and Bill Weber of Pacific
Northwest National Laboratory has identified such
materials. Using simulation techniques, they
discovered that gadolinium zirconate materials
resist radiation damage for millennia. These
compounds absorb energy through the rearrangement of
atoms within the crystal structure without becoming
amorphous or structurally unstable—making them
superior to the titanate materials being considered
internationally for plutonium immobilization.
(Plutonium-bearing titanates would degrade much
faster.) The researchers also confirmed the mobility
of the disturbed atoms and the ease of incorporating
plutonium into the gadolinium-zirconate structure.
Scientific
Impact: These studies demonstrated a
systematic increase in radiation resistance as
zirconium is substituted for titanium in gadolinium
compounds. Discovery of these materials has
stimulated research elsewhere, including Los Alamos
National Laboratory, and led to identification of a
phase that seems to be the best candidate for
immobilizing plutonium.
Social
Impact: This material offers a promising
means of keeping future generations safe from the
dual threats of radioactive contamination caused by
plutonium decay, and the nuclear proliferation that
might result from further use of the plutonium in
weapons. Thus, this work may help resolve major
dilemmas of the nuclear age.
Reference:
S. X. Wang, B. D. Begg, L. M. Wang, R. C. Ewing, W.
J. Weber, and K. V. Govidan Kutty, "Radiation
Stability of Gadolinium Zirconate: A Waste Form for
Plutonium Disposition," J. Materials
Research 14 [12] (1999) 4470-4473.
W. J. Weber et al., "Radiation Effects in
Crystalline Ceramics for the Immobilization of
High-Level Nuclear Waste and Plutonium," J.
Materials Research, 13 [6] (1998) 1434-1484.
W. J. Weber and R. C. Ewing, "Plutonium
Immobilization and Radiation Effects," Science
289 (2000) 2051-2052.
B. D. Begg, N. J. Hess, D. E. McCready, S.
Thevuthasan, and W. J. Weber, "Heavy-Ion
Irradiation Effects in Gd2(ZrxTi1-x)2O7 Pyrochlores," J. Nuclear Materials 289
[1-2] (2001) 188-193.
|
|
 |
Explaining
and Applying Magnetism
|
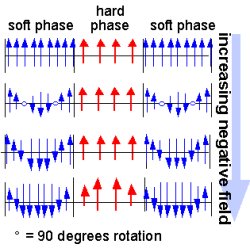 |
| Principle
of Operation: Hard phase stiffens response
of soft high-magnetization phase in magnetic
nanocomposite structures. |
Metallic magnetism is
a time-honored field of study that in recent years
has undergone a renaissance, thanks in part to
Office of Science support. Research at Argonne
National Laboratory on thin-film metallic
multilayers is widely recognized as providing the
basis for the discovery by others of giant
magnetoresistance (GMR), an effect used widely today
in recording heads in magnetic data storage devices.
Related research has resulted in new GMR materials
and structures, as well as contributions to the
development and understanding of colossal
magnetoresistance, a more powerful effect that may
be used in future recording devices. Research at
Brookhaven National Laboratory and Idaho National
Engineering and Environmental Laboratory on hard
magnets (permanently magnetized materials) explained
the link between microstructure and properties in
magnets made of rare earth materials; magnetic
properties were improved dramatically through the
design of microstructures on the nanoscale. Other
work focuses on understanding and exploiting
mixtures of hard and soft magnets with high magnetic
strength. (The magnetization in soft magnets can be
changed with applied magnetic fields.)
Scientific
Impact: These studies have advanced the
science of magnetic materials and paved the way for
manufacture of magnet structures with greater
mechanical strength and stability. Researchers
benefit from these materials through their use in
permanent magnet devices at Office of
Science-supported synchrotrons and most other light
sources around the world.
Social
Impact: Magnetic materials are used in many
industrial and consumer devices such as motors,
generators, and computers. Improvements in magnet
properties and processing characteristics will
enhance energy efficiency; for example, the use of
rare earth magnets in more efficient electric motors
could save the nation several billion dollars
annually.
Reference:
L. H. Lewis, A. R. Moodenbaugh, D. O. Welch and V.
Panchanathan, "Stress, Strain and Technical
Magnetic Properties in "Exchange-Spring"
Nd2Fe14B + a-Fe Nanocomposite Magnets", J.
Phys. D.: Appl. Phys. 34 (2001) 744-751.
D. J. Branagan, M. J. Kramer, Yali Tang, R. W.
McCallum, D. C. Crew and L. H. Lewis,
"Engineering Magnetic Nanocomposite
Microstructures", J. Materials Science,
35(14): 3459-3466, July 2000.
E. E. Fullerton, C. H. Sowers, J. E. Pearson, X. Z.
Wu, D. Lederman, and S. D. Bader, "A General
Approach to the Epitaxial Growth of
Rare-Earth-Transition-Metal Films," Appl.
Phys. Lett. 69, 2438 (1996).
E. E. Fullerton, M. J. Conover, J. E. Mattson, C. H.
Sowers, and S. D. Bader, "Oscillatory
interlayer coupling and giant magnetoresistance in
epitaxial Fe/Cr(211) and (100) superlattices", Phys.
Rev. B 48, 15755 (1993).
|
|
 |
Modeling
Metals
|
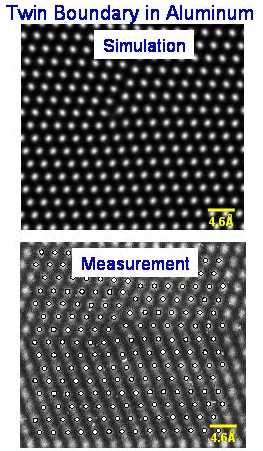 |
|
The Embedded Atom Method (EAM) can reliably simulate not only atomic structure of perfect crystals, but also defects and grain boundaries. An example is shown
above where the high resolution TEM measurement of an Aluminum grain boundary is directly compared to the EAM calculation. |
For many years,
scientists did not fully understand how and why
metals and alloys sometimes cracked. The mystery was
finally solved in the early 1980s, when Murray Daw
and Mike Baskes at Sandia National Laboratories
developed a way to accurately describe the
embrittlement of steel by hydrogen or other
corrosive impurities, a problem of interest in
defense applications. Using concepts from density
functional theory, they constructed the embedded
atom method (EAM) from a "first
principles" quantum mechanical framework for
describing metal bonding. The EAM simplified the
quantum behavior so that high-speed calculations
could easily be made on very large systems
containing hundreds of thousands of atoms. Whereas
most fracture studies previously had been conducted
on scales of inches or even feet, the EAM revealed
much finer detail by focusing instead on
atomic-scale processes, such as slight movements of
electrons that weaken metal bonds. The EAM
accurately describes important quantities such as
cohesion and deformation of metals, making possible
computer simulations that are useful in designing
and predicting the behavior of complex materials and
engineering components.
Scientific
Impact: The EAM revolutionized
computational materials science by enabling
large-scale simulations of the atomic structure and
evolution of metals; the method successfully
simulates complex processes such as metal
deformation, embrittlement, and fracture. The method
is currently used by more than 100 groups worldwide
and has resulted in more than 1,000 published works
with more than 2,700 citations to the original
paper.
Social
Impact: The EAM is used in industry to
design alloys for use in metallic parts and
products.
Reference:
"Parallel Molecular Dynamics With the Embedded
Atom Method", S. J. Plimpton and B. A.
Hendrickson, in Materials Theory and Modelling,
edited by J. Broughton, P. Bristowe, and J. Newsam, MRS
Proceedings 291, Pittsburgh, PA, 1993, p 37.
|
|
 |
Organic-Based
Magnets: A New Frontier
|
 |
| Photograph
of a coating of the V[TCNE]x magnet on a
Teflon tape being attracted to a Co5Sm
magnet at room temperature in the air. |
Once considered to be
a scientific impossibility, organic magnets
(containing little or no metallic material) were
discovered by chemist Joel Miller then at Du Pont,
and physicist Arthur Epstein of The Ohio State
University and both supported by the Office of
Science. In 1986, they discovered the first organic
material to become magnetically ordered (at very low
temperature, -268 degrees C / -441 degrees F),
demonstrating for the first time that a magnet could
be made using organic chemistry and without the
usual high temperature, energy intensive processing.
(Magnetic ordering refers to the orientation of each
atoms' electron spins, which behave like tiny
magnets; when many adjacent electron spins align in
the same direction, the material can be a strong
magnet.) Miller and Epstein's compound is composed
of molecular units. These are the first soluble as
well as nonmetallurgically prepared magnets, and are
more magnetic then iron metal. (Due to the high
density of iron, organic magnets can never be as
magnetic as iron on a volume basis.) Their research
led to the 1991 discovery of the first
organic/polymeric material to exhibit magnetism
above room temperature, opening the door to many
potential applications. More recently, the two
researchers made thin magnetic films using a unique
low-temperature process as well as another material
that becomes magnetically ordered far above room
temperature (~100 degrees C, or 212 degrees F).
These accomplishments have been profiled on the
covers of 15 journals and recognized by the American
Chemical Society's 2000 National Award for Chemistry
of Materials.
Scientific
Impact: This work created a new class of
materials and a thriving field of research that
could lead to many new technologies. Since the
original discovery, several new classes of polymeric
organic magnets have been identified, and research
consortiums have formed in both Europe and Japan.
Social
Impact: Organic magnets are lighter, more
flexible, and less energy intensive to make than
conventional metal and ceramic magnets. Applications
now being pursued include magnetic shielding,
magneto-optical switching, and "smart"
materials. The magnetic properties of these
materials change when exposed to light, making them
candidates for high-density optical data storage
systems.
Reference:
Ferromagnetic Properties of One-Dimensional
Decamethylferrocenium Tetracyanoethylenide (1:1):
[Fe(C5Me5)2] + [TCNE]. J.S. Miller, J.C. Calabrese,
A J. Epstein, R.W. Bigelow, J. H. Zhang, W.M. Reiff,
J. Chem. Soc. Chem. Commun. 1026-1028
(1986).
Organic and Organometallic Magnetic Materials -
Designer Magnets, J. S. Miller, A. J. Epstein, Angew.
Chem. internat. edit. 33, 385-415
(1994).
Designer Magnets, J.S. Miller, A. J. Epstein, Chem.
Eng. News, 73(#40), 30-41 (1995).
A Room Temperature Molecular/Organic-Based Magnet,
J.M. Manriquez, G.T. Yee, R.S. McLean, A. J.
Epstein, J. S. Miller, Science, 252,
1415-1417 (1991).
Tetracyanoethylene-based Organic Magnets, J.S.
Miller, A.J. Epstein, J. Chem. Soc., Chem.
Commun. 1319-1325 (1998).
Thin Film V[TCNE]x Magnets, K. I. Pokhodnya, A. J.
Epstein, J. S. Miller. Adv. Mater. 12,
410-413 (2000).
Enhancement of the Magnetic Ordering Temperature
(and Air Stability) of a Mixed Valent Vanadium
Hexacyanochromate(III) Magnet to 99 C (372 K), O.
Hatlevik, W. E. Buschmann, J. Zhang, J. L. Manson,
J. S. Miller, Adv. Mater. 11, 914-918
(1999).
Organometallic- and Organic-based Magnets: New
Chemistry and New Materials for the New Millennium,
J. S. Miller, Inorg. Chem. 39, 4392-4408
(2000).
|
|
 |
Manipulating
Light in Photonic Crystals
|
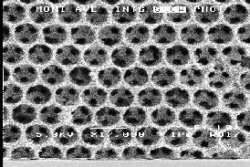 |
| A
photonic band gap crystal produced by a
colloidal crystallization technique using
sized, template spheres. |
Not long ago, theory
and experiments failed to agree on the question of
how light propagates in crystals. But in 1990,
researchers at Ames Laboratory proved the theorists
correct by demonstrating the existence of structures
with a "photonic bandgap" (PBG), a range
of frequencies within which a specific wavelength of
light is blocked. Scientists then knew they could
custom-design crystals to trap and manipulate light,
sending it down assigned routes and even around
loops and bends. Among their novel optical
properties, PBG crystals can manipulate light
without absorption; the energy not emitted in one
frequency region is redirected into other
frequencies, a useful feature in energy-efficient
devices. Early photonic crystals had a bandgap in
the microwave region of the electromagnetic
spectrum. Using a layered lattice design and
microfabrication capabilities at Sandia National
Laboratories, scientists moved the bandgap to
shorter wavelengths, in the infrared, for
applications such as optical communications. Ames
also produced computer programs that allow for the
rapid design, analysis, and optimization of PBG
structures.
Scientific
Impact: The Ames' work spawned a growing
global research community and knowledge base
focusing on PBG crystals and related atomic
properties and behavior. The high accuracy of Ames?
theoretical calculations assists in the
interpretation and design of PBG experiments and
devices, and the layered lattice approach has been
used to make the smallest PBG crystal ever
fabricated.
Social
Impact: PBG crystals could revolutionize
the control of light propagation, emission, and
absorption in optical devices; thus, they have many
potential uses in compact and efficient sensors,
antennas, lasers, electronics, lighting, solar
cells, and telecommunications equipment (e.g.,
optical switches, waveguides). The microfabrication
method developed at Sandia is economical and lends
itself to mass production.
Reference:
"Photonic Crystals," M.M. Sigalas, R.
Biswas, G. Tuttle, C.M. Soukoulis, and K.M. Ho, Wiley
Encyclopedia of Electrical and Electronic
Engineering, Volume 16, 345 (John Wiley, 1999).
"Existence
of a photonic gap in periodic dielectric structures,"
K. M. Ho, C. T. Chan, and C. M. Soukoulis, Phys.
Rev. Lett. 65, 3152 (1990).
|
|
 |
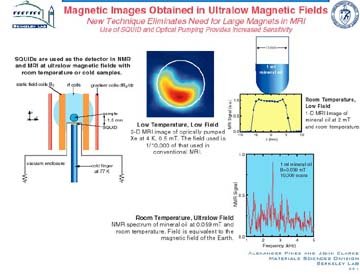
Extending
the Power of Nuclear Magnetic Resonance Techniques
|
 |
| A
larger .pdf version of this figure is
available at the URL listed below. |
Scientists at
Lawrence Berkeley National Laboratory, led by Alex
Pines, are world leaders in magnetic identification
and imaging concepts and techniques used worldwide
in science and industry. Both nuclear magnetic
resonance (NMR) spectroscopy and magnetic resonance
imaging (MRI) are based on the tiny magnetic moments
produced by the "spin" of atomic nuclei;
NMR provides spectra for use in identifying
molecules, whereas MRI produces recognizable images.
The Pines group's research has extended the
applicability of NMR to a wide range of problems and
materials, including biological systems. They also
applied their new methods to study novel materials
such as nanocrystals, liquid crystals, and zeolites.
Recently, they helped develop a technique in which
hyperpolarized gas molecules transfer added momentum
to other atomic nuclei, increasing the sensitivity
of NMR of molecules in solution and MRIs of
materials and organisms, a major step toward
extending the power of these techniques in chemistry
and biology. The group also developed methods that
allow for the use of very low magnetic fields in MRI
and may eliminate the need for large, costly magnets
in these instruments.
Scientific
Impact: These novel concepts and techniques
have revolutionized the study of structure,
dynamics, and function in solid materials and other
systems that previously were inaccessible to NMR
investigations. Low-field MRI is ideal for studying
highly porous, magnetic materials and fossils and
rocks.
Social
Impact: These techniques and instruments
have been licensed and incorporated into commercial
NMR technology used worldwide. A company has
licensed the low-field MRI technology to develop
medical applications. In addition, Pines has trained
about 200 scientists, of whom many now hold leading
positions in academia and industry.
Reference:
Y.Q. Song, B.M. Goodson, and A. Pines, "NMR and
MRI Using Laser-Polarized Xenon," Spectroscopy,
(14) 26-33 (July 1999).
|
|
 |
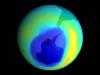
Saving
the Earth's Ozone Layer
|
 |
|
The stratospheric
layer of ozone 15 to 50 kilometers above the Earth
absorbs ultraviolet radiation, preventing it from
reaching the planet's surface. For many years,
scientists assumed that this protective ozone would
not be affected by release into the atmosphere of
chlorofluorocarbons (CFCs), chemically inert and
nontoxic gases once common in aerosol sprays and
refrigerants. But in fact, CFCs do threaten the
ozone layer, as explained in 1974 by F. Sherwood
Rowland of the University of California, Irvine, and
Mario Molina of the Massachusetts Institute of
Technology. Rowland was supported by predecessors to
the Office of Science for his research in hot-atom
chemistry. Initially interested in species formed as
a result of nuclear reactions, he extended his work
to study the photochemical formation of chlorine
atoms. Roland and Molina theorized that CFC
molecules could be split apart by solar radiation to
produce chlorine atoms, which could catalyze the
destruction of ozone. They were right, as underlined
later by discovery of the "ozone hole"
over the Antarctic. Rowland and Molina, together
with Paul Crutzen of the Max-Planck-Institute for
Chemistry in Germany, shared the 1995 Nobel Prize in
Chemistry for their work on the formation and
decomposition of ozone.
Scientific
Impact: Discovery of the effect of CFCs on
the ozone layer was a seminal contribution to
atmospheric chemistry.
Social
Impact: Rowland and Molina's work initially
led to restrictions on CFC releases; after discovery
of the ozone hole, an international agreement was
signed to limit the manufacture and use of these
compounds. Thus, this research has helped mitigate a
global environmental problem with potentially
catastrophic consequences. It will take at least 100
years for the ozone layer to recover fully.
Reference:
Molina, M. J., and F. S. Rowland, Stratospheric Sink
for Chlorofluoromethanes: Chlorine Catalyzed
Destruction of Ozone, Nature, 249, 810-814 (1974)
|
|
 |
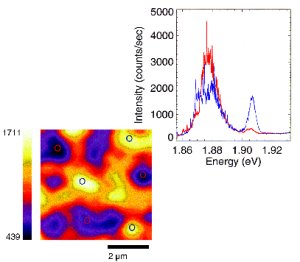
Making
Solar Energy More Affordable
|
 |
| Solar
Panels |
 |
|
TEM
Structure Analysis
|
The cost of solar
electricity has been reduced 100-fold over the past
two decades, but further reductions are needed
before solar power is widely used. Scientists at the
National Renewable Energy Laboratory took major
steps toward this goal by designing photovoltaic
cells (which convert sunlight to electricity) with
30 percent efficiency, much higher than the 10-20
percent levels achieved previously. The new cells
consist of thin layers of semiconductors applied to
a low-cost backing, such as glass or plastic. The
researchers received Office of Science support to
develop a basic understanding of the opto-electronic
properties of various semiconductors. Calculations
of electronic structure provided the knowledge
needed to precisely engineer layered semiconductors.
Then, a tandem device was designed with two solar
cells made of materials that respond to different
parts of the solar spectrum; the top cell (made of
gallium indium phosphide) absorbs the high-energy
component of sunlight and passes the rest to the
bottom cell (made of gallium arsenide) for
absorption. Researchers are working on the addition
of a third cell to push efficiency to more than 40
percent, to open up new opportunities for
terrestrial and space applications.
Scientific
Impact: These advances have added to the
scientific and engineering knowledge base needed to
make solar power more practical and useful. For
instance, the material used in the top layer of the
new device is much more resistant to radiation
damage than conventional silicon and thus will have
a longer useful life.
Social
Impact: The technology was transferred to a
major supplier of photovoltaic cells for space
power, and four satellites using it are in orbit,
flashing back telephone and television signals. The
new solar cells provide as much as 50 percent more
power than previous cells, so the satellites can
carry more communications links, experiments, or
other projects and operate more economically.
Reference:
Cotal, H. L.; Lillington, D. R.; Ermer, J. H.; King,
R. R.; Karam, N. H.; Kurtz, S. R.; Friedman, D. J.;
Olson, J. M.; Ward, S.; Duda, A.; Emery, K. A.;
Moriarty, T. (2000). "Highly Efficient 32.3%
Monolithic GaInP/GaAs/Ge Triple Junction
Concentrator Solar Cells." Program and
Proceedings: NCPV Program Review Meeting 2000,
16-19 April 2000, Denver Colorado. BK-520-28064.
Golden, CO: National Renewable Energy Laboratory;
pp. 111-112; NICH Report No. CP-520-29664.
|
|
 |
Enhancing
Separations and Analysis
|
 |
|
For many decades, the
Office of Science and predecessor agencies have
supported studies of the scientific principles
underlying chemical separations and analysis. The
most notable achievement was the development of the
host-guest complexation concept by Donald J. Cram of
the University of California, Los Angeles. This
concept, which explains how molecules recognize and
react with each other, changed how scientists think
about separation and sequestration of elements. Cram
also synthesized organic molecules that imitated the
primary functions of enzymes, helping to fulfill a
long-standing dream of chemists. For his pioneering
work, Cram shared the 1987 Nobel Prize in Chemistry
with two other scientists. Other researchers with
Office of Science support developed inductively
coupled plasma to produce sample materials for
chemical analysis; wrote a powerful software
program, SIMION, to help design particle beams and
traps for fundamental studies; performed important
basic research on supercritical fluids (liquefied
gases used in solvent extraction); and developed
laser-based detection schemes that enhanced the
sensitivity of important analytical methods.
Scientific
Impact: Cram helped to lay the foundation
for one of the most active fields of chemical
research, known as host-guest or supramolecular
chemistry, which is directly applicable to
separations. The work on inductively coupled plasma
ushered in the era of ultra-trace multi-element
analysis, enabling the rapid and accurate
determination of up to 70 elements in metals,
alloys, and organic compounds (such as oil, serum,
blood, and soils). SIMION is used at every national
laboratory.
Social
Impact: Inductively coupled plasma is used
in environmental testing and the production of
ultrapure materials for the semiconductor and
nuclear industries. The research on supercritical
fluids contributed to the recent introduction of
"green chemistry" for commercial dry
cleaning and polymer manufacturing. SIMION is used
by instrument suppliers that design mass
spectrometers.
Reference:
DOE-BES Chemical Sciences, Highlights of
Progress in Separations Sciences, 1980-1999,
Edited by Charles H. Byers, IsoPro International
Inc., 2140 Santa Cruz Ave, #C304, Menlo Park, CA
94025
|
|
 |
Sequencing the First Plant Genome
|
 |
|
Arabidopsis
thaliana
|
Arabidopsis
thaliana, a small plant in the mustard family,
has become the model for molecular genetic research
on plants because of its small size, rapid growth
cycle (6 weeks), large production of seed, and small
genome (the smallest known of any flowering plant).
In 1990, when Arabidopsis was being tested
as a laboratory model for plant genetics, the DOE
Plant Research Laboratory at Michigan State
University initiated a project to analyze expressed
sequence tags (ESTs), mirror images of fragments of
genes and the proteins they make. Because they can
be used to scan for and tag active genes, ESTs
rapidly became important tools for identifying and
isolating plant genes. Subsequently, other federal
agencies provided support, resulting in a
multinational computer database linked to U.S. and
European stock centers, which distribute seeds and
DNA of Arabidopsis to researchers
worldwide. The research that grew out of this work
laid the groundwork for the Arabidopsis
Genome Initiative, which began 1996 as a
multinational effort to sequence this plant's
genome. The entire sequence (130 million pairs of
chemical units) was officially completed recently
and is largely available on the World Wide Web. As a
direct model for 250,000 closely related species, Arabidopsis
will help scientists understand the molecular basis
of plant growth and development and address
fundamental questions in plant physiology,
biochemistry, cell biology, and pathology.
Scientific
Impact: The MSU effort was instrumental in
establishing Arabidopsis as a model
organism for identifying and studying plant genes at
the molecular level. Since the early sequencing of
ESTs, studies using Arabidopsis have
yielded many significant advances, including the
discovery of plant hormone and signal receptor
action and components of disease resistance.
Social
Impact: Concentrated research on this
single plant will provide detailed information that
can be applied to a wide range of plant attributes
relevant to energy, manufacturing, the environment,
agriculture, and even human health. One result has
been crops that are more resistant to the cold;
further insights will help scientists make other
plants easier to grow under adverse conditions,
healthier to eat, and more disease resistant.
Reference:
Arabidopsis thaliana Genome Sequencing
Completed," Nature, December 14, 2000.
|
|
 |
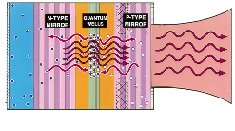
New Opto-electronic Materials and Devices
|
 |
| Vertical
Cavity Surface-Emitting Laser (VCSEL)
(sideview). It is a sandwich of high tech
materials that traps and uses electric
current to generate a laser beam. |
Stacks of ultrathin
layers—each less than one-thousandth the thickness
of a human hair—are the secret to a class of
artificially grown materials that have enabled
numerous advances in technology over the past
generation. In 1981, scientists at Sandia National
Laboratories were the first to predict the unique
electronic and optical properties of strained-layer
semiconductor (SLS) superlattices, and, a few years
later, the first to make devices from them. These
crystalline materials got their name because the
spacing between the atoms in different layers is
mismatched initially, but the thinness of the layers
allows alignment by elastic strain without causing
dislocations or other defects. Because the number,
composition, and thickness of the layers can be
varied over wide limits, scientists can tailor the
electrical and optical properties to design
materials and devices with targeted properties. This
work has won a number of awards, including the
American Physical Society's International Prize for
New Materials in 1993.
Scientific
Impact: This work established new areas of
materials science and electronics as well as new
research technologies; for instance, SLS materials
are used to make transistors for high-frequency,
low-noise electronic amplifiers, such as those found
in radiotelescopes. These materials made it possible
for scientists to tailor the wavelength (or color)
of light-emitting devices (such as light-emitting
diodes) and increase the speed of electrons in
transistors.
Social
Impact: The SLS technology revolutionized
the multibillion-dollar field of opto-electronics
and is a key to wireless communications. These
materials enhance the performance and efficiency of
semiconductor lasers and make possible new types of
lasers with applications in optical communications,
supermarket scanners, remote sensing, and medical
diagnostics.
Reference:
"Laser Gain and Threshold Properties in
Compressive-Strained and Lattice-Matched
GaInNAs/GaAs Quantum Wells", W. W. Chow, E. D.
Jones, Appl. Phys. Lett 75, pp. 2891-93
(1999).
"Pressure Dependence of the Bandgap Energy and
the Conduction-Band Mass for an N-Type InGaAs/GaAs
Single-Strained Quantum Well", E. D. Jones, S.
W. Tozer, and T. Schmiedel, Physica E 2,
pp.146-150 (1998).
"Study of Cyclotron Resonance and
Magneto-Photoluminescence of N-Type Modulation-Doped
In GaAs Quantum Well Layers and Their
Characterizations", N. Kotera, E. D. Jones, K.
Tanaka, H. Arimoto, M. Ohno, N. Miura, T. Mishima,
edited by S. C. Shen, D. Y. Tang, G. Z. Zheng, and
G. Bauer (World Scientific, Singapore, 199)
pp. 591-598.
"Room-Temperature Continuous Wave InGaAsN
Quantum Well Vertical-Cavity Lasers Emitting at 1.3
Microns", K. D. Choquette, J. F. Klem, A. J.
Fischer, O. Blum, A. A. Allerman, I. J. Fritz, S. R.
Kurtz, W.G. Breiland, R. Sieg, K. M. Electronics
Letters Vol. 36, 1388 (2000).
"GaAsSb/InGaAs Type-II Quantum Wells for
Long-Wavelength Lasers on GaAs Substrates", J.
F. Klem, O. Blum, S. R. Kurtz, I. J. Fritz, and K.
D. Choquette, J. Vac. Sci. Technol. B, Vol.
18, 1605 (2000). "Strained-layer semconductor
superlattices from lattice mismatched
materials." Osbourn, J.C. J. Applied
Physics (53) p1586 (1982).
"InGaAs strained-layer semiconductor
superlattices: A proposal for useful new electronic
materials." Osbourn, J.C. Phy Rev. B.
(27) p5126 (1983).
|
|
 |
Unraveling the Mystery of High-Temperature
Superconductivity
|
 |
| Stripe
Formation. Picture of the spin and charge
densities in the copper-oxygen planes where
the conducting electrons are located. Lower
density antiferromagnetic regions are seen
to alternate with higher charge density
regions. Such an effect is far more probable
in two dimensional, i.e., planar,
geometrical arrangements. Courtesy of
researchers at Brookhaven National
Laboratory. |
Since the discovery
in the 1980s of high-temperature superconductors,
the Office of Science has supported research
designed to explain and improve the physical
behavior of these materials and develop methods of
making wires and other objects from them. These
materials conduct electricity with virtually no
resistance at temperatures high enough to be cooled
by liquid nitrogen (-196 degrees C, or -321 degrees
F) instead of more costly helium. Studies at various
national laboratories have led to discoveries
concerning, for example, the relationships between
magnetic behavior and superconductivity, and between
material layering and current-carrying capability.
Argonne National Laboratory clarified the nature of
several different phases of vortex matter (compounds
often break down at the vortex, where the molecules
of different materials meet), leading to new
configurations that improve conductivity. Argonne
also built the first superconducting motor and
developed a process for welding lengths of wire in a
way that maintains superconductivity. Other
investigators have observed "charge
stripes" in materials exhibiting colossal
magnetoresistance, an unusual and powerful effect
that may be exploited in future magnetic recording
devices. Years of research at Oak Ridge National
Laboratory led to the development of processes that
may enable the manufacture of long lengths of
superconducting wires and tape.
Scientific
Impact: This research has greatly increased
scientific understanding of high-temperature
superconductors. As yet, there is no comprehensive
theory that explains all of the experimental
phenomena; this remains a key question in condensed
matter physics.
Social
Impact: Superconducting wires and tape can
carry 100 to 200 times more electric current than
conventional wires. These innovations could enable
the widespread commercialization of more efficient
types of power generation, transmission, and
electrical equipment and devices, offering
tremendous energy savings and emissions reductions.
Reference:
S.L. Bud'ko, G. Lapertot, C. Petrovic, C.E.
Cunningham, N. anderson, and P.C. Canfield.
"Boron Isotope Effect in Superconducting
MgB2," Physical Review Letters,
February 26, 2001.
|
|
 |
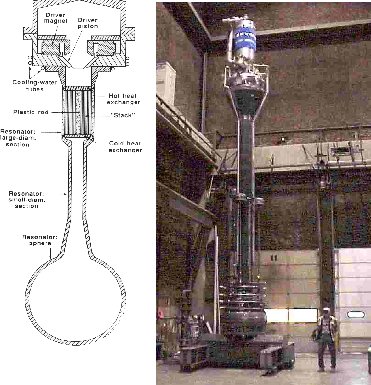
Harnessing the "Thermoacoustic"
Effect
|
 |
|
Thermoacoustic
Refrigeration
|
A sound wave consists
of oscillations in pressure, temperature, and
displacement. Although the temperature oscillations
are small, research during the past two decades has
shown that this "thermoacoustic" effect
can be harnessed to produce powerful, reasonably
efficient heat engines, including heat pumps, and
refrigerators. Thermoacoustic engines typically have
no moving parts; at most, there is a single
oscillating part (such as a loudspeaker) with no
sliding seals. Thus, these engines have the
potential to be both simple and reliable. Research
by Greg Swift at Los Alamos National Laboratory on
the thermodynamics of the thermoacoustic process has
led to the development of prototype refrigerators
with cooling powers up to tens of watts, and
prototype engines with efficiencies approaching
those of conventional engines. The research has
spawned collaborative efforts that have resulted in
advances in the theory, design, and construction of
thermoacoustic devices.
Scientific
Impact: Los Alamos' leadership in both the
scientific and technological aspects of
thermoacoustics since the mid-1980s has generated a
sizeable academic research community around the
world. The first international workshop on
thermoacoustics will be held in 2001.
Social
Impact: Thermoacoustic energy conversion
(including conversion of heat to acoustic power,
acoustic power to refrigeration, and acoustic power
to mixture separation) is reasonably efficient and
should be inexpensive and reliable in mass
production. Efforts are under way to develop a
natural-gas liquefier for use in remote locations, a
residential co-generation system to produce both
electricity and gas heat, an electric generator for
deep-space probes, and a water chiller for use on
submarines.
Reference:
S. Backhaus and G.W. Swift. "A
thermoacoustic-Stirling heat engine." Nature,
399:335-338, 1999.
G. W. Swift. "Thermoacoustic engines and
refrigerators." Physics Today, pages
22-28, July 1995.
G. W. Swift. "Thermoacoustic engines." J.
Acoust. Soc. Am., 84:1145-1180, 1988.
|
|
 |
Metallic Glasses with Extraordinary
Properties
|
 |
|
Bulk
Metallic Glasses
|
|
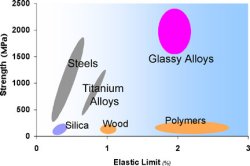 |
| Typical
strengths and elastic limits for various
materials. Metallic glasses are unique. |
Metallic glasses
with extraordinary magnetic properties, and
practical methods for processing these
materials, have been developed over the past
four decades with support from the Office of
Science and predecessor agencies. Pol Duwez at
the California Institute of Technology produced
the first ribbons of metallic glasses, which had
unusual mechanical strength, magnetic behavior,
and resistance to wear and corrosion that set
them apart from conventional crystalline
materials. The processing method involved
chilling molten metal at rates in excess of
1,000,000 degrees C per second. Duwez and his
student William L. Johnson also discovered other
alloys that could be made into metallic glasses
for high-efficiency magnets, but expensive
processing was required to fabricate forms
useful for motors and transformers. During the
1980s, Johnson developed new compositions that
could be processed without rapid cooling in bulk
or three-dimensional form (bulk forms are more
than 20 times thicker than the roughly
40-micrometer ribbons), suitable for casting or
possibly molding into complex shapes for
precision parts, without the costs or wastes
associated with machining. Recently, scientists
at Los Alamos National Laboratory produced a
bulk ferromagnetic glass with a record-low
magnetic energy loss that does not require
expensive processing, a form appropriate for
energy conversion devices.
Scientific
Impact: This research opened up a new
area of materials science and technology—for
which DOE was the sole U.S. supporter until
recently—that offers opportunities for
increasing the efficiency of magnets, motors,
and transformers. Los Alamos is a world leader
in research on bulk ferromagnetic glasses, the
only form appropriate for motors and
transformers.
Social
Impact: These materials are used in
products ranging from motor components to golf
clubs and also have great potential for military
applications. The use of bulk ferromagnetic
glasses in energy-conversion devices would
reduce costly losses from power-distribution
systems and corrosion damage, and the consequent
reduced use of energy from fossil sources would
reduce the rate of release of carbon dioxide
into the atmosphere.
Reference:
Masuhr A, Busch R, Johnson WL. "Rheometry
and Crystallization of Bulk Metallic Glass
Forming Alloys at High Temperatures." ISMANAM
1997 - Materials Science Forum. Barcelona,
Spain. Switzerland: Trans Tech Publications,
1998: 779-84.
|
|
 |
Extending the Science of Transition Metal
Nitrides
|
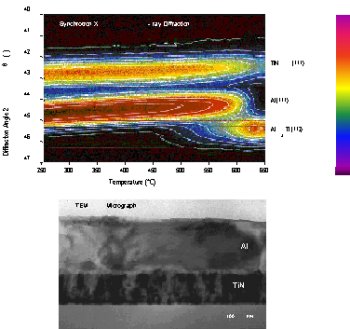 |
| Advanced
Nitrides by Design. Synchrotron XRD and TEM
microanalyses were used to determine
interfacial reaction paths and mechanisms.
The results allowed functional design of
dramatically improved diffusion barriers. |
Novel semiconductors,
superconductors, and corrosion-resistant materials
have been developed recently through nanoscale
research on transition metal nitrides. J. E. Greene,
I. Petrov, and colleagues at the University of
Illinois Seitz Materials Research Laboratory, with
Office of Science support, combined theoretical
modeling with fundamental growth and
characterization experiments to improve the basic
mechanical and electrical properties of nitrides.
They developed new processes for depositing these
materials with control of atomic-scale reaction and
diffusion, thereby designing whole families of
alloys with unique properties that are impossible to
achieve under equilibrium conditions. To achieve
these properties, it was necessary to control grain
size and texture on a scale on the order of 10
nanometers (nm), and to achieve interfacial widths
of 0.1 nm to 1.0 nm. This work has many applications
and has been recognized by many awards, including
the 1999 David Turnbull Lectureship of the Materials
Research Society, the 1998 David Adler Prize from
the American Physical Society, and the Tage Erlanger
Prize in Physics (the second-ranking Swedish prize
in science after the Nobel Prize).
Scientific
Impact: This work extended the science of
transition metal nitrides, making possible the
design of entirely new materials. These achievements
also demonstrate the value of research on the
nanoscale, an emerging field of great importance.
Social
Impact: Transition metal nitrides already
have practical uses; titanium aluminum nitride, for
example, has become ubiquitous in wear-, corrosion-,
and diffusion-resistant coatings for products such
as cutting tools. The new alloys have enabled the
use of copper interconnects in integrated circuits
through the creation of improved diffusion barriers,
thus paving the way for a new generation of faster
computer chips.
Reference:
J. S. Chun, I. Petrov, and J.E. Greene, "Dense
fully 111-textured TiN diffusion barriers: Enhanced
lifetime through microstructure control during layer
growth" J. Appl. Phys., 86 3633
(1999).
D. Gall, I. Petrov, P. Desjardins, and J.E. Greene,
"Microstructural evolution and Poisson ratio of
epitaxial ScN grown on TiN(001)/MgO(001) by
ultrahigh vacuum reactive magnetron sputter
deposition" J. Appl. Phys., 86 5524
(1999).
|
|
 |
A New Type of Microscopy
|
 |
| Image
of a strontium titanate grain boundary taken
with Z-contrast TEM. |
A microscope invented
with Office of Science support is the first
technique to produce a direct image of a complex
atomic structure while also identifying the atoms
involved. Steve Pennycook of Oak Ridge National
Laboratory combined elements of three existing
electron microscopes to make the Z-contrast
microscope, which uses electrons bounced off
(scattered from) a sample to form an image of the
atoms. Because the scattered intensity depends on
the atomic number (Z) of the chemical element being
probed, the image intensity provides a means of
identifying the atoms. The method improves on
scanning electron microscopes (which produce clear
images but cannot penetrate materials), transmission
electron microscopes (which produces images that
cannot be interpreted directly as atomic structure),
and the hybrid scanning transmission electron
microscope (which produces outstanding microanalysis
but poor-quality images). Z-contrast microscopy is
particularly suited to the viewing of interfaces,
grain boundaries, and defects in materials-features
that cannot be analyzed well using indirect means.
The Z-contrast microscope won an R&D 100 award
from R&D Magazine as a significant new
technology. Pennycook also received the Materials
Research Society Medal and the Kurt J. Heinrich
Award of the Microbeam Analysis Society.
Scientific
Impact: Z-contrast microscopy has had major
impact on the study of materials structure. It has
achieved the highest resolution of a crystal
structure ever recorded in a microscope and provided
new information on the atomic-scale structure and
chemistry of a variety of materials—correcting
previously published quasicrystal structures, for
example.
Social
Impact: Z-contrast microscopes are
commercially manufactured. This tool is likely to
lead to dramatic advances in structural materials,
superconductors, and semiconductors, especially in
the smoothness of interfaces where different
materials join, and thereby pave the way for
improved computers, fiber-optic communications,
medical imaging, and laser-disc players.
Reference:
M. F. Chisholm and S. J. Pennycook, "Z-Contrast
Imaging of Grain-Boundary Core Structures in
Semiconductors," MRS Bulletin 22, 53
(1997).
|
|
 |
|
|
|

