NCATS develops novel approaches to improve the process of joining basic science discoveries with initial testing of therapies in humans.
-
Research
-
- Funding & Notices
- News & Media
- About Translation
-
- About NCATS
-
NCATS Programs & Initiatives
- 3-D Tissue Bioprinting Program
- Assay Development and Screening Technology (ADST)
- Biomedical Data Translator
- Bridging Interventional Development Gaps (BrIDGs)
- Chemistry Technology
- Discovering New Therapeutic Uses for Existing Molecules
- Genetic and Rare Diseases Information Center (GARD)
- Matrix Combination Screening
- Early Translation Branch (ETB)
- A Specialized Platform for Innovative Research Exploration (ASPIRE)
- About NCATS
Preclinical Innovation

 NCATS Patent Book Showcases Innovation, Collaboration and Opportunities for New Partnerships
NCATS Patent Book Showcases Innovation, Collaboration and Opportunities for New Partnerships
A new digital book showcases 22 patents issued to NCATS and its collaborators that can help speed the process of translating discoveries into medical interventions. NCATS seeks additional commercial partners to license and use the patents.

 Commonly Used Antibiotic Shows Promise for Combating Zika Infections
Commonly Used Antibiotic Shows Promise for Combating Zika Infections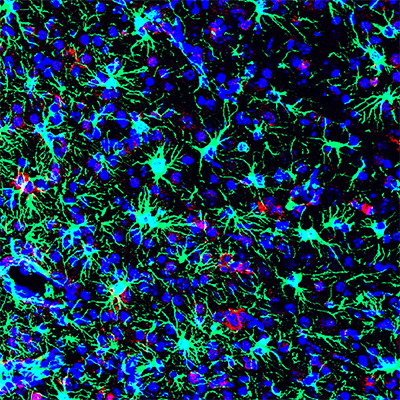
NIH-supported preclinical research suggests that the widely used antibiotic methacycline was effective at preventing brain infections and reducing neurological problems associated with the Zika virus.
 Apply Now for the New NCATS/FDA Translational Science Interagency Fellowship
Apply Now for the New NCATS/FDA Translational Science Interagency Fellowship
NCATS and the U.S. Food and Drug Administration are jointly sponsoring the new, three-year Translational Science Interagency Fellowship to provide fellows with training in both translational science and regulatory science.
 Improving the Drug Development Process
Improving the Drug Development ProcessCreating and testing innovative methods to improve and accelerate the drug development process and find new treatments for patients faster.
 Repurposing Drugs
Repurposing DrugsDiscovering new uses for approved drugs to provide the quickest possible transition from bench to bedside.
 Testing & Predictive Models
Testing & Predictive ModelsDeveloping better model systems for drug and toxicity testing.
 Core Technologies
Core TechnologiesUsing state-of-the-art resources to enable the ongoing operation of all NCATS translational research activities.


Improving the Drug Development Process
Creating and testing innovative methods to improve and accelerate the drug development process and find new treatments for patients faster.

Discovering new uses for approved drugs to provide the quickest possible transition from bench to bedside.

Developing better model systems for drug and toxicity testing.

Using state-of-the-art resources to enable the ongoing operation of all NCATS translational research activities.
Work with Us
NCATS preclinical programs and resources focus on key obstacles and inefficiencies in the translational process, overcoming bottlenecks that slow the development of new treatments for patients.
Learn more about how to access NCATS programs and expertise.
Inside the NCATS Laboratories
Take a video tour of our labs and see firsthand how our researchers work to get more treatments to more patients more quickly.
Learn more about NCATS’ Achievements in Advancing Translational Sciences.
NCATS Programs & Resources Supporting Preclinical Innovation
- 3-D Tissue Bioprinting Program
- Biomedical Data Translator
- Bridging Interventional Development Gaps (BrIDGs)
- Chemistry Technology
- Discovering New Therapeutic Uses for Existing Molecules (New Therapeutic Uses)
- Extracellular RNA Communication
- Matrix Combination Screening
- NCATS Chemical Genomics Center (NCGC)
- NCATS Pharmaceutical Collection
- Pfizer's Centers for Therapeutic Innovation (CTI) for NIH Researchers
- RNA Interference (RNAi)
- Therapeutics for Rare and Neglected Diseases (TRND)
- Tissue Chip for Drug Screening
- Toxicology in the 21st Century (Tox21)
- Funding & Notices
-



