
News Releases
December 2020

Investigational COVID-19 Therapeutics to be Evaluated in Large Clinical Trials
December 17, 2020
Two randomized, controlled Phase 3 clinical trials have begun evaluating investigational monoclonal antibodies for their safety and efficacy in treating people hospitalized with COVID-19. The trials are part of the ACTIV-3 master protocol, which has an adaptive design allowing investigators to add new sub-studies of additional investigational agents. ACTIV-3 is sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health.

NIH Observational Study of Coronavirus Infection and Multisystem Inflammatory Syndrome in Children Begins
December 16, 2020
An observational study has launched to evaluate the short- and long-term health outcomes of SARS-CoV-2 infection in children, including multisystem inflammatory syndrome in children (MIS-C), and to characterize the immunologic pathways associated with different disease presentations and outcomes. SARS-CoV-2 is the virus that causes COVID-19. The study, called the Pediatric Research Immune Network on SARS-CoV-2 and MIS-C (PRISM), will enroll at least 250 children and young adults ages 20 years or younger from diverse racial and ethnic backgrounds at approximately 20 sites nationwide.

Baricitinib Plus Remdesivir Shows Promise for Treating COVID-19
December 11, 2020
The combination of baricitinib, an anti-inflammatory drug, and remdesivir, an antiviral, reduced time to recovery for people hospitalized with COVID-19, according to clinical trial results published in the New England Journal of Medicine. The study was supported by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health.
NIH Statement on World AIDS Day 2020
December 01, 2020
Today on World AIDS Day, we reflect both on the remarkable progress that has been made against HIV as well as the considerable challenges that remain. We now have highly effective HIV treatment and prevention methods, and work is underway to address the remaining challenges in delivering these tools to the people who need them most, as well as to develop new interventions. The National Institutes of Health continues to advance rigorous, innovative research to prevent new HIV transmissions and improve the health of people with HIV worldwide.
November 2020
NIH Announces Restructured HIV Clinical Trials Networks
November 30, 2020
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, today announced the clinical investigators and institutions that will lead four NIH HIV clinical trials networks over the next seven years to conduct the innovative, efficient clinical research needed to accelerate progress against the HIV pandemic. NIAID also awarded grants to 35 U.S. and international institutions selected as HIV clinical trials units (CTUs).
Experimental Vaccine for Deadly Tickborne Virus Effective in Cynomolgus Macaques
November 30, 2020
An experimental vaccine developed in Europe to prevent infection by Crimean-Congo hemorrhagic fever virus (CCHFV) has protected cynomolgus macaques in a new collaborative study from National Institutes of Health scientists. The animals received the DNA-based candidate vaccine through intramuscular injection immediately followed by electroporation—a process in development for human vaccines that helps cells absorb DNA. The study, published in Nature Microbiology, comes about three years after the same research group developed the macaque model for CCHFV.
Statement—Fourth Iteration of COVID-19 Treatment Trial Underway
November 25, 2020
The National Institute of Allergy and Infectious Diseases (NIAID), one of the National Institutes of Health, today announced that the fourth iteration of the Adaptive COVID-19 Treatment Trial (ACTT-4) has begun to enroll hospitalized adults with coronavirus disease 2019 (COVID-19) who require supplemental oxygen. The NIAID-sponsored trial will enroll up to 1,500 patients at approximately 100 sites in the United States and other countries.
Promising Interim Results from Clinical Trial of NIH-Moderna COVID-19 Vaccine
November 16, 2020
An independent data and safety monitoring board (DSMB) overseeing the Phase 3 trial of the investigational COVID-19 vaccine known as mRNA-1273 reviewed trial data and shared its interim analysis with the trial oversight group on Nov. 15, 2020. This interim review of the data suggests that the vaccine is safe and effective at preventing symptomatic COVID-19 in adults.

Treatments for People with Early COVID-19 Infection is an Urgent Research Focus
November 11, 2020
COVID-19 treatments for people with early infection are needed urgently, according to a JAMA Viewpoint article by NIAID Director Anthony S. Fauci, M.D., and colleagues. Treating people early in the course of infection with SARS-CoV-2, the virus that causes COVID-19, would speed their recovery, reduce the likelihood that they develop severe outcomes and reduce demand on the healthcare system, they write.
Statement—NIH Study Finds Long-Acting Injectable Drug Prevents HIV Acquisition in Cisgender Women
November 09, 2020
A pre-exposure prophylaxis (PrEP) regimen containing an investigational long-acting form of the HIV drug cabotegravir injected once every eight weeks was safe and more effective than a daily oral PrEP regimen at preventing HIV acquisition among a group of cisgender women. The women, from southern and east Africa, are enrolled in a clinical trial sponsored by the National Institutes of Health.
October 2020
Statement—NIH-Sponsored ACTIV-3 Trial Closes LY-CoV555 Sub-Study
October 26, 2020
The ACTIV-3 clinical trial evaluating the investigational monoclonal antibody LY-CoV555 in hospitalized patients with COVID-19 will not enroll more participants into this sub-study following a recommendation from the independent Data and Safety Monitoring Board (DSMB). The trial is sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health.
Landmark TB Trial Identifies Shorter-Course Treatment Regimen
October 21, 2020
Results from an international, randomized, controlled clinical trial indicate that a four-month daily treatment regimen containing high-dose, or “optimized,” rifapentine with moxifloxacin is as safe and effective as the existing standard six-month daily regimen at curing drug-susceptible tuberculosis (TB) disease. This regimen is the first successful short-course treatment regimen for drug-susceptible TB disease in almost 40 years.

NIH Study Aims To Identify Promising COVID-19 Treatments for Larger Clinical Trials
October 13, 2020
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, today launched a study designed to determine whether certain approved therapies or investigational drugs in late-stage clinical development show promise against COVID-19 and merit advancement into larger clinical trials.

NIH Clinical Trial Testing Hyperimmune Intravenous Immunoglobulin Plus Remdesivir to Treat COVID-19 Begins
October 08, 2020
A clinical trial to test the safety, tolerability and efficacy of a combination treatment regimen for coronavirus disease 2019 (COVID-19) consisting of the antiviral remdesivir plus a highly concentrated solution of antibodies that neutralize SARS-CoV-2, the virus that causes COVID-19, has begun. The study is taking place in hospitalized adults with COVID-19 in the United States, Mexico and 16 other countries on five continents.
September 2020
BULLETIN—NIAID Stops Enrollment of Severely Ill COVID-19 Participants in Clinical Trial of Investigational Treatments
September 29, 2020
The Adaptive COVID-19 Treatment Trial 3 (ACTT-3), sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, will no longer enroll hospitalized participants with severe COVID-19 requiring high-flow oxygen, and will not begin to enroll patients requiring non-invasive or invasive mechanical ventilation.

Investigational COVID-19 Vaccine Well-Tolerated and Generates Immune Response in Older Adults
September 29, 2020
A Phase 1 trial of an investigational mRNA vaccine to prevent SARS-CoV-2 infection has shown that the vaccine is well-tolerated and generates a strong immune response in older adults. A report published today in the New England Journal of Medicine describes the findings from the study, which was supported by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health. SARS-CoV-2 is the virus that causes COVID-19 disease.

Scientists Discover Genetic and Immunologic Underpinnings of Some Cases of Severe COVID-19
September 24, 2020
New findings by scientists at the National Institutes of Health and their collaborators help explain why some people with COVID-19 develop severe disease. The findings also may provide the first molecular explanation for why more men than women die from COVID-19.

Fourth Large-Scale COVID-19 Vaccine Trial Begins in the United States
September 23, 2020
A fourth Phase 3 clinical trial evaluating an investigational vaccine for coronavirus disease 2019 (COVID-19) has begun enrolling adult volunteers. The trial is designed to evaluate if the investigational Janssen COVID-19 vaccine (JNJ-78436725) can prevent symptomatic COVID-19 after a single dose regimen. Up to 60,000 volunteers will be enrolled in the trial at up to nearly 215 clinical research sites in the United States and internationally.

Probiotic Skin Therapy Improves Eczema in Children, NIH Study Suggests
September 09, 2020
An experimental treatment for eczema that aims to modify the skin microbiome safely reduced disease severity and increased quality of life for children as young as 3 years of age, a National Institutes of Health study has found. These improvements persisted for up to eight months after treatment stopped, researchers report Sept. 9 in Science Translational Medicine.
August 2020
Phase 3 Clinical Testing in the U.S. of AstraZeneca COVID-19 Vaccine Candidate Begins
August 31, 2020
A multi-site, Phase 3 clinical trial evaluating an investigational COVID-19 vaccine known as AZD1222 has begun. The trial will enroll approximately 30,000 adult volunteers at 80 sites in the United States to evaluate if the candidate vaccine can prevent symptomatic coronavirus disease 2019 (COVID-19). The United Kingdom-based global biopharmaceutical company AstraZeneca is leading the trial as regulatory sponsor.

NIAID Establishes Centers for Research in Emerging Infectious Diseases
August 27, 2020
The National Institute of Allergy and Infectious Diseases (NIAID), one of the National Institutes of Health, today announced that it has awarded 11 grants with a total first-year value of approximately $17 million to establish the Centers for Research in Emerging Infectious Diseases (CREID). The global network will involve multidisciplinary investigations into how and where viruses and other pathogens emerge from wildlife and spillover to cause disease in people. NIAID intends to provide approximately $82 million over 5 years to support the network.
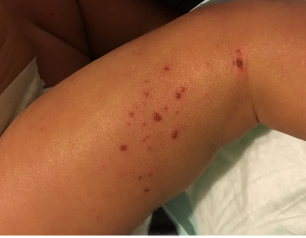
NIH-Supported Scientists Demonstrate How Genetic Variations Cause Eczema
August 14, 2020
New research supported by the National Institutes of Health delineates how two relatively common variations in a gene called KIF3A are responsible for an impaired skin barrier that allows increased water loss from the skin, promoting the development of atopic dermatitis, commonly known as eczema. This finding could lead to genetic tests that empower parents and physicians to take steps to potentially protect vulnerable infants from developing atopic dermatitis and additional allergic diseases.

Clinical Trials of Monoclonal Antibodies to Prevent COVID-19 Now Enrolling
August 10, 2020
Two Phase 3, randomized, placebo-controlled, double-blind clinical trials testing whether experimental monoclonal antibodies (mAbs) can prevent infection by SARS-CoV-2 coronavirus are now enrolling healthy adults at clinical trial sites in the United States.
NIH Clinical Trial Testing Remdesivir Plus Interferon Beta-1a for COVID-19 Treatment Begins
August 05, 2020
A randomized, controlled clinical trial evaluating the safety and efficacy of a treatment regimen consisting of the antiviral remdesivir plus the immunomodulator interferon beta-1a in patients with coronavirus disease 2019 (COVID-19) has begun. The study, called the Adaptive COVID-19 Treatment Trial 3 (ACTT 3), is anticipated to enroll more than 1,000 hospitalized adults with COVID-19 at as many as 100 sites in the United States and abroad.

NIH-Moderna Investigational COVID-19 Vaccine Shows Promise in Mouse Studies
August 05, 2020
The investigational vaccine known as mRNA-1273 protected mice from infection with SARS-CoV-2, the virus that causes COVID-19, according to research published today in Nature. Scientists at the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, and the biotechnology company Moderna, based in Cambridge, Massachusetts, along with collaborators from the University of North Carolina at Chapel Hill, Vanderbilt University Medical Center in Nashville, and the University of Texas at Austin conducted the preclinical research.

