TRND program supports preclinical development of therapeutic candidates intended to treat rare or neglected disorders, with the goal of enabling an Investigational New Drug (IND) application. Learn more.
-
Research
-
- Funding & Notices
- News & Media
- About Translation
-
- About NCATS
-
NCATS Programs & Initiatives
- 3-D Tissue Bioprinting Program
- Assay Development and Screening Technology (ADST)
- Biomedical Data Translator
- Bridging Interventional Development Gaps (BrIDGs)
- Chemistry Technology
- Discovering New Therapeutic Uses for Existing Molecules
- Genetic and Rare Diseases Information Center (GARD)
- Matrix Combination Screening
- Early Translation Branch (ETB)
- A Specialized Platform for Innovative Research Exploration (ASPIRE)
- About NCATS
- Home
- About NCATS
- NCATS Programs & Initiatives
- Therapeutics for Rare and Neglected Diseases (TRND)
Therapeutics for Rare and Neglected Diseases (TRND)
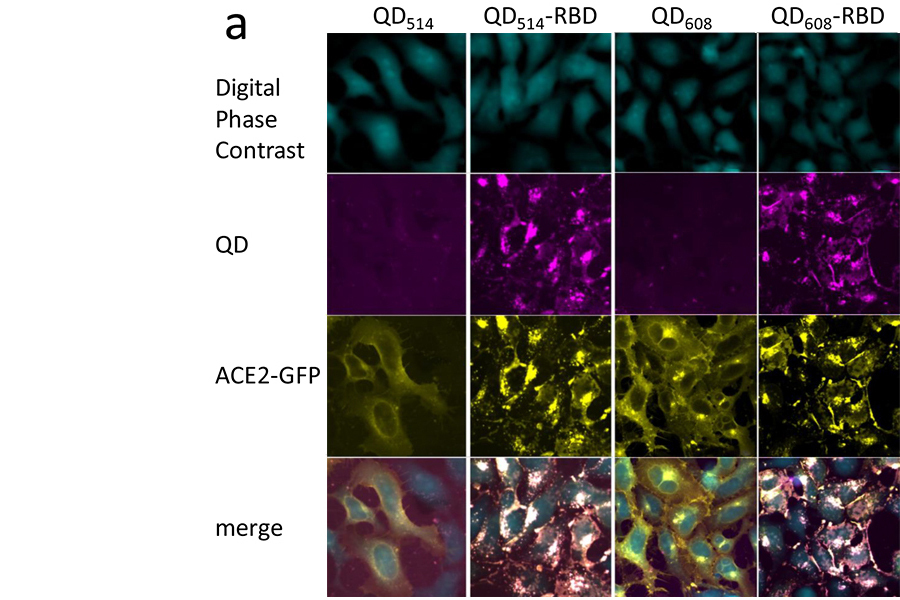
 Quantum Dot Probes Could Reveal New Ways to Address SARS-CoV-2
Quantum Dot Probes Could Reveal New Ways to Address SARS-CoV-2
Researchers from NCATS and the Naval Research Laboratory in Washington, D.C., developed a new tool that mimics how SARS-CoV-2 — the virus that causes COVID-19 — infects a cell, which could potentially speed the drug discovery process for COVID-19.

 Spreading a Message of Hope at Rare Disease Day at NIH
Spreading a Message of Hope at Rare Disease Day at NIH
The annual event — held this year on Feb. 28, 2020 — focused on forging connections in the rare diseases community, shortening the journey toward diagnosis and pursuing personalized medicine as a therapeutic approach for treating rare diseases.
 OpenData Portal: Sharing Data to Advance COVID-19 Research
OpenData Portal: Sharing Data to Advance COVID-19 Research
NCATS created a new resource for scientists, the OpenData Portal, to openly and quickly share COVID-19-related drug repurposing data and experiments for all approved drugs.
 About TRND
About TRNDTRND supports preclinical development from lead optimization through IND application.
 Scientific Capabilities
Scientific CapabilitiesTRND offers world-class in-house experts and connections to external resources.
 Work with TRND
Work with TRNDPartner with TRND to create and implement a therapeutic project plan for in-kind resources.
 TRND Projects
TRND ProjectsExplore active and completed therapeutic development projects supported by TRND, including projects with clinical activities.


TRND supports preclinical development from lead optimization through IND application.

TRND offers world-class in-house experts and connections to external resources.

Partner with TRND to create and implement a therapeutic project plan for in-kind resources.

Explore active and completed therapeutic development projects supported by TRND, including projects with clinical activities.
Work with Us
TRND stimulates therapeutic development research collaborations among NIH and academic scientists, nonprofit organizations, and pharmaceutical and biotechnology companies working on rare and neglected illnesses. Learn more and contact TRND.
Why TRND Matters
There are more than 6,500 identified rare and neglected diseases, yet only about 250 treatments are available for these conditions. One reason is that limited numbers of patients can make gathering information and designing drug studies difficult. As a result, scientists often know little about the symptoms and biology of these conditions. Another obstacle is that some private companies may find it difficult to justify the cost of developing drugs for such small rare disease markets. Even in the case of infectious diseases — such as malaria — that inflict health burdens on large numbers of people in the developing world, the private sector often neglects therapeutic development because of insufficient economic incentives. Learn more about TRND.
TRND Facts
Download the TRND fact sheet (PDF - 347KB).
NCATS Programs & Initiatives
- Funding & Notices
-