Weekly U.S. Influenza Surveillance Report

2016-2017 Influenza Season Week 42 ending October 22, 2016
All data are preliminary and may change as more reports are received.
Synopsis:
During week 42 (October 16-22, 2016), influenza activity was low in the United States.
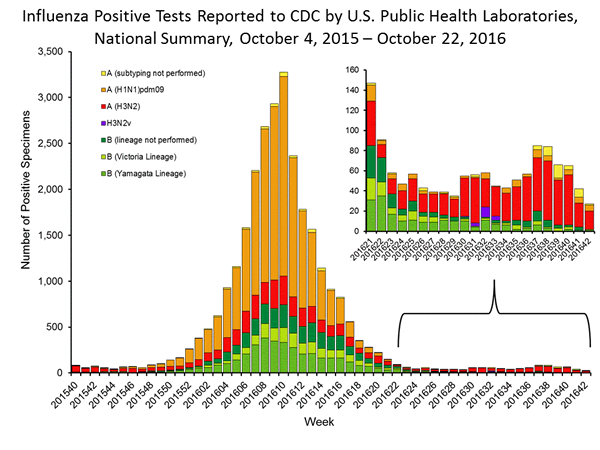
- Viral Surveillance: The most frequently identified influenza virus type reported by public health laboratories during week 42 was influenza A. The percentage of respiratory specimens testing positive for influenza in clinical laboratories was low.
- Pneumonia and Influenza Mortality: The proportion of deaths attributed to pneumonia and influenza (P&I) was below the system-specific epidemic threshold in the National Center for Health Statistics (NCHS) Mortality Surveillance System.
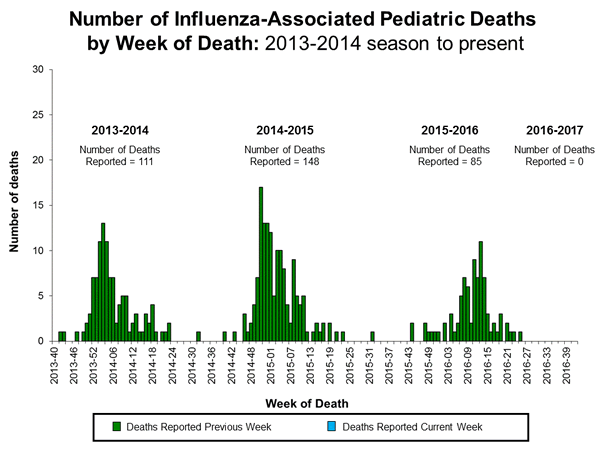
- Influenza-associated Pediatric Deaths: No influenza-associated pediatric deaths were reported.
- Outpatient Illness Surveillance: The proportion of outpatient visits for influenza-like illness (ILI) was 1.3%, which is below the national baseline of 2.2%. All 10 regions reported ILI below region-specific baseline levels. New York City, Puerto Rico, and all 50 states experienced minimal ILI activity and the District of Columbia had insufficient data.
- Geographic Spread of Influenza: The geographic spread of influenza in Guam was reported as widespread; Puerto Rico reported regional activity; one state reported local activity; the District of Columbia, the U.S. Virgin Islands and 41 states reported sporadic activity; and eight states reported no activity.
National and Regional Summary of Select Surveillance Components
| HHS Surveillance Regions* | Data for current week | Data cumulative since October 2, 2016 (week 40) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Out-patient ILI† | Number of jurisdictions reporting regional or widespread activity§ | % respiratory specimens positive for flu in clinical laboratories‡ | A(H1N1)pdm09 | A (H3) | A (Subtyping not Performed) |
B Victoria lineage | B Yamagata lineage | B lineage not performed | Pediatric Deaths | |
| Influenza test results from public health laboratories only | ||||||||||
| Nation | Normal | 2 of 54 | 1.6% | 17 | 92 | 13 | 3 | 4 | 5 | 0 |
| Region 1 | Normal | 0 of 6 | 0.6% | 0 | 10 | 0 | 0 | 0 | 0 | 0 |
| Region 2 | Normal | 1 of 4 | 0.9% | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| Region 3 | Normal | 0 of 6 | 0.6% | 2 | 1 | 1 | 0 | 0 | 0 | 0 |
| Region 4 | Normal | 0 of 8 | 3.8% | 0 | 10 | 5 | 1 | 2 | 2 | 0 |
| Region 5 | Normal | 0 of 6 | 0.7% | 0 | 18 | 1 | 0 | 1 | 0 | 0 |
| Region 6 | Normal | 0 of 5 | 1.6% | 2 | 0 | 0 | 1 | 0 | 0 | 0 |
| Region 7 | Normal | 0 of 4 | <0.1% | 0 | 4 | 0 | 1 | 0 | 0 | 0 |
| Region 8 | Normal | 0 of 6 | 0.9% | 11 | 5 | 0 | 0 | 0 | 0 | 0 |
| Region 9 | Normal | 1 of 4 | 1.0% | 2 | 22 | 0 | 0 | 1 | 1 | 0 |
| Region 10 | Normal | 0 of 4 | 2.2% | 0 | 22 | 6 | 0 | 0 | 1 | 0 |
*HHS regions (Region 1 CT, ME, MA, NH, RI, VT; Region 2: NJ, NY, Puerto Rico, US Virgin Islands; Region 3: DE, DC, MD, PA, VA, WV; Region 4: AL, FL, GA, KY, MS, NC, SC, TN; Region 5: IL, IN, MI, MN, OH, WI; Region 6: AR, LA, NM, OK, TX; Region 7: IA, KS, MO, NE; Region 8: CO, MT, ND, SD, UT, WY; Region 9: AZ, CA, Guam, HI, NV; and Region 10: AK, ID, OR, WA).
† Elevated means the % of visits for ILI is at or above the national or region-specific baseline
§ Includes all 50 states, the District of Columbia, Guam, Puerto Rico, and U.S. Virgin Islands
‡ National data are for current week; regional data are for the most recent three weeks
U.S. Virologic Surveillance:
WHO and NREVSS collaborating laboratories, which include both public health and clinical laboratories located in all 50 states, Puerto Rico, and the District of Columbia, report to CDC the total number of respiratory specimens tested for influenza and the number positive for influenza by virus type. In addition, public health laboratories also report the influenza A subtype (H1 or H3) and influenza B lineage information for the viruses they test and the age or age group of the persons from whom the specimens were collected.
Additional data are available at http://gis.cdc.gov/grasp/fluview/fluportaldashboard.html and http://gis.cdc.gov/grasp/fluview/flu_by_age_virus.html.
The results of tests performed by clinical laboratories during the current week are summarized below.
| Week 42 | Data Cumulative since October 2, 2016 (Week 40) |
|
|---|---|---|
| No. of specimens tested | 10,837 | 35,844 |
| No. of positive specimens (%) | 176 (1.6%) | 555 (1.6%) |
| Positive specimens by type | ||
| Influenza A | 94 (53.4%) | 311 (56.0%) |
| Influenza B | 82 (46.6%) | 244 (44.0%) |

View National and Regional Level Graphs and Data | View Chart Data | View Full Screen | View PowerPoint Presentation
The results of tests performed by public health laboratories, as well as the age group distribution of influenza positive tests, during the current week are summarized below.
| Week 42 | Data Cumulative since October 2, 2016 (Week 40) |
|
|---|---|---|
| No. of specimens tested | 400 | 1,691 |
| No. of positive specimens | 27 | 134 |
| Positive specimens by type/subtype | ||
| Influenza A | 25 (92.6%) | 122 (91.0%) |
| A(H1N1)pmd09 | 6 (24.0%) | 17 (13.9%) |
| H3 | 18 (72.0%) | 92 (75.4%) |
| Subtyping not performed | 1 (4.0%) | 13 (10.7%) |
| Influenza B | 2 (7.4%) | 12 (9.0%) |
| Yamagata lineage | 1 (50.0%) | 4 (33.3%) |
| Victoria lineage | 1 (50.0%) | 3 (25.0%) |
| Lineage not performed | 0 (0%) | 5 (41.7%) |
View National and Regional Level Graphs and Data | View Chart Data | View Full Screen | View PowerPoint Presentation

View Interactive Application | View Full Screen
Influenza Virus Characterization:
CDC characterizes influenza viruses through one or more tests including genomic sequencing, hemagglutination inhibition (HI) and/or neutralization assays. These data are used to compare how similar currently circulating influenza viruses are to the reference viruses used for developing influenza vaccines, and to monitor for changes in circulating influenza viruses. Historically, HI data have been used most commonly to assess the similarity between reference viruses and circulating viruses to suggest how well the vaccine may work until such a time as vaccine effectiveness estimates are available.
For nearly all virus positive surveillance samples received at CDC, next-generation sequencing is performed to ascertain genomic data of circulating influenza viruses. Viruses can be classified into genetic groups/clades based on analysis of their HA gene segments using phylogenetics and key amino acid changes (Klimov Vaccine 2012).
Genetic Characterization
During May 22 – October 22, 2016, 1,297 influenza positive specimens were collected and reported by public health laboratories in the United States (figure, left). CDC genetically characterized 250 influenza viruses [21 influenza A (H1N1)pdm09, 152 influenza A (H3N2), and 77 influenza B viruses] collected by U.S. laboratories. The HA gene segment of all influenza A (H1N1)pdm09 viruses analyzed belonged to genetic group 6B.1. Influenza A (H3N2) virus HA gene segments analyzed belonged to genetic groups 3C.2a or 3C.3a. Genetic group 3C.2a includes a newly emerging subgroup known as 3C.2a1. The HA of influenza B/Victoria-lineage viruses all belonged to genetic group V1A. The HA of influenza B/Yamagata-lineage viruses analyzed all belonged to genetic group Y3.
The majority of U.S. viruses submitted for characterization come from state and local public health laboratories. Specimen submission guidance issued to the laboratories request that, if available, 2 influenza A (H1N1), 2 A influenza (H3N2), and 2 influenza B viruses be submitted every other week. Because of this, the number of each virus type/subtype characterized should be approximately equal. In the figure below, the results of tests performed by public health labs are presented on the left and sequence results by genetic group of specimens submitted to CDC are presented on the right.

View Chart Data | View Full Screen | View PowerPoint Presentation
Antigenic Characterization
During May 22 – October 22, 2016, CDC has antigenically characterized 150 influenza viruses [10 influenza A (H1N1)pdm09, 77 influenza A (H3N2), and 63 influenza B viruses] collected by U.S. laboratories.
Influenza A Virus [87]
- A (H1N1)pdm09 [10]: All 10 (100%) influenza A (H1N1)pdm09 viruses were antigenically characterized using ferret post-infection antisera as A/California/7/2009-like, the influenza A (H1N1) component of the 2016-2017 Northern Hemisphere vaccine.
- A (H3N2) [77]: 65 (84.4%) of 77 influenza A (H3N2) viruses were antigenically characterized as A/Hong Kong/4801/2014-like, a virus that belongs in genetic group 3C.2a and is the influenza A (H3N2) component of the 2016-2017 Northern Hemisphere vaccine, by HI testing or neutralization testing. Among the viruses which reacted poorly with ferret antisera raised against A/Hong Kong/4801/2014-like viruses, 11 of 12 (91.7%) are more closely related to A/Switzerland/9715293/2013), a virus belonging to genetic group 3C.3a.
Influenza B Virus [63]
- Victoria Lineage [30]: All 30 (100%) B/Victoria-lineage viruses were antigenically characterized using ferret post-infection antisera as B/Brisbane/60/2008-like, which is included as an influenza B component of the 2016-2017 Northern Hemisphere trivalent and quadrivalent influenza vaccines.
- Yamagata Lineage [33]: All 33 (100%) B/Yamagata-lineage viruses were antigenically characterized using ferret post-infection antisera as B/Phuket/3073/2013-like, which is included as an influenza B component of the 2016-2017 Northern Hemisphere quadrivalent influenza vaccines.
Antiviral Resistance:
No antiviral resistance data is available for specimens collected after October 1, 2016.
During May 22-Septemer 30, 2016, 228 specimens (26 influenza A (H1N1)pdm09, 128 influenza A (H3N2), and 74 influenza B viruses) collected in the United States were tested for susceptibility to the neuraminidase inhibitors (oseltamivir, zanamivir, and peramivir). Among the 26 influenza A (H1N1)pdm09 viruses tested for oseltamivir and peramivir, one (3.8%) was resistant to both drugs. A total of 22 influenza A (H1N1)pdm09 viruses were tested for zanamivir susceptibility and all were susceptible. None of the 128 influenza A (H3) and 74 influenza B viruses were found to be resistant to either oseltamivir, zanamivir, or peramivir.
The majority of recently circulating influenza viruses are susceptible to the neuraminidase inhibitor antiviral medications, oseltamivir, zanamivir, and peramivir; however, rare sporadic instances of oseltamivir-resistant and peramivir-resistant influenza A (H1N1)pdm09 viruses and oseltamivir-resistant influenza A (H3N2) viruses have been detected worldwide. Antiviral treatment as early as possible is recommended for patients with confirmed or suspected influenza who have severe, complicated, or progressive illness; who require hospitalization; or who are at high risk for serious influenza-related complications. Additional information on recommendations for treatment and chemoprophylaxis of influenza virus infection with antiviral agents is available at http://www.cdc.gov/flu/antivirals/index.htm.
Pneumonia and Influenza (P&I) Mortality Surveillance:
Based on National Center for Health Statistics (NCHS) mortality surveillance data available on October 26, 2016, 5.3% of the deaths occurring during the week ending October 8, 2016 (week 40) were due to P&I. This percentage is below the epidemic threshold of 6.5% for week 40.
P&I percentages for recent weeks may be artificially low due to a backlog of records requiring manual processing. Percentages will likely increase to levels more similar to the baseline as more data becomes available.
Region and state-specific data are available at http://gis.cdc.gov/grasp/fluview/mortality.html.

View Regional and State Level Data | View Chart Data | View Full Screen | View PowerPoint Presentation
Influenza-Associated Pediatric Mortality:
No influenza-associated pediatric deaths were reported to CDC during week 42.
Additional data can be found at: http://gis.cdc.gov/GRASP/Fluview/PedFluDeath.html.Influenza-Associated Hospitalizations:
The Influenza Hospitalization Surveillance Network (FluSurv-NET) conducts population-based surveillance for laboratory-confirmed influenza-related hospitalizations in select counties in the Emerging Infections Program (EIP) states and Influenza Hospitalization Surveillance Project (IHSP) states. FluSurv-NET estimated hospitalization rates will be updated weekly starting later this season. Additional FluSurv-NET data can be found at: http://gis.cdc.gov/GRASP/Fluview/FluHospRates.html and http://gis.cdc.gov/grasp/fluview/FluHospChars.html.
Outpatient Illness Surveillance:
Nationwide during week 42, 1.3% of patient visits reported through the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) were due to influenza-like illness (ILI). This percentage is below the national baseline of 2.2%.
(ILI is defined as fever (temperature of 100°F [37.8°C] or greater) and cough and/or sore throat.)
Additional data are available at http://gis.cdc.gov/grasp/fluview/fluportaldashboard.html.

View National and Regional Level Graphs and Data | View Chart Data | View Full Screen | View PowerPoint Presentation
On a regional level, the percentage of outpatient visits for ILI ranged from 0.5% to 1.8% during week 42. All 10 regions reported a proportion of outpatient visits for ILI below their region-specific baseline levels.
ILINet State Activity Indicator Map:
Data collected in ILINet are used to produce a measure of ILI activity* by state. Activity levels are based on the percent of outpatient visits in a state due to ILI and are compared to the average percent of ILI visits that occur during weeks with little or no influenza virus circulation. Activity levels range from minimal, which would correspond to ILI activity from outpatient clinics being below, or only slightly above, the average, to high, which would correspond to ILI activity from outpatient clinics being much higher than average.
During week 42, the following ILI activity levels were experienced:
- New York City, Puerto Rico, and all 50 states experienced minimal ILI activity.
- Data were insufficient to calculate an ILI activity level from the District of Columbia.
*This map uses the proportion of outpatient visits to health care providers for ILI to measure the ILI activity level within a state. It does not, however, measure the extent of geographic spread of flu within a state. Therefore, outbreaks occurring in a single city could cause the state to display high activity levels.
Data collected in ILINet may disproportionally represent certain populations within a state, and therefore, may not accurately depict the full picture of influenza activity for the whole state.
Data displayed in this map are based on data collected in ILINet, whereas the State and Territorial flu activity map is based on reports from state and territorial epidemiologists. The data presented in this map is preliminary and may change as more data are received.
Differences in the data presented here by CDC and independently by some state health departments likely represent differing levels of data completeness with data presented by the state likely being the more complete.
Geographic Spread of Influenza as Assessed by State and Territorial Epidemiologists
The influenza activity reported by state and territorial epidemiologists indicates geographic spread of influenza viruses, but does not measure the severity of influenza activity.
During week 42, the following influenza activity was reported:
- Widespread influenza activity was reported by Guam.
- Regional influenza activity was reported by Puerto Rico.
- Local influenza activity was reported by one state (Alabama).
- Sporadic influenza activity was reported by the District of Columbia, the U.S. Virgin Islands and 41 states (Alaska, Arizona, Arkansas, California, Colorado, Connecticut, Delaware, Florida, Georgia, Hawaii, Indiana, Iowa, Kansas, Kentucky, Louisiana, Maine, Maryland, Massachusetts, Michigan, Minnesota, Missouri, Montana, Nevada, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, South Carolina, South Dakota, Tennessee, Texas, Utah, Virginia, Washington, Wisconsin, and Wyoming).
- No activity was reported by eight states (Idaho, Illinois, Mississippi, Nebraska, New Hampshire, Rhode Island, Vermont, and West Virginia).
Flu Activity data in XML Format | View Full Screen
Additional National and International Influenza Surveillance Information
FluView Interactive: FluView includes enhanced web-based interactive applications that can provide dynamic visuals of the influenza data collected and analyzed by CDC. These FluView Interactive applications allow people to create customized, visual interpretations of influenza data, as well as make comparisons across flu seasons, regions, age groups and a variety of other demographics. To access these tools, visit http://www.cdc.gov/flu/weekly/fluviewinteractive.htm.
U.S. State and local influenza surveillance: Click on a jurisdiction below to access the latest local influenza information.
World Health Organization: Additional influenza surveillance information from participating WHO member nations is available through FluNet and the Global Epidemiology Reports.
WHO Collaborating Centers for Influenza located in Australia, China, Japan, the United Kingdom, and the United States (CDC in Atlanta, Georgia).
Europe: For the most recent influenza surveillance information from Europe, please see WHO/Europe and the European Centre for Disease Prevention and Control at http://www.flunewseurope.org/.
Public Health Agency of Canada: The most up-to-date influenza information from Canada is available at http://www.phac-aspc.gc.ca/fluwatch/
Public Health England: The most up-to-date influenza information from the United Kingdom is available at https://www.gov.uk/government/statistics/weekly-national-flu-reports
Any links provided to non-Federal organizations are provided solely as a service to our users. These links do not constitute an endorsement of these organizations or their programs by CDC or the Federal Government, and none should be inferred. CDC is not responsible for the content of the individual organization web pages found at these links.
An overview of the CDC influenza surveillance system, including methodology and detailed descriptions of each data component, is available at: http://www.cdc.gov/flu/weekly/overview.htm.
--------------------------------------------------------------------------------- Page last reviewed: October 28, 2016
- Page last updated: October 28, 2016
- Content source:
- Centers for Disease Control and Prevention, National Center for Immunization and Respiratory Diseases (NCIRD)
- Page maintained by: Office of the Associate Director for Communication, Digital Media Branch, Division of Public Affairs


 ShareCompartir
ShareCompartir