Congressional Justification for FY 2012
National Eye Institute
Department of Health and Human Services
National Institutes of Health
Download the PDF* version of this document.
FY 2012 Budget:
- Organization Chart
- Appropriation Language
- Amounts Available for Obligation
- Budget Mechanism Table
- Major Changes in Budget Request
- Summary of Changes
- Budget Graphs
- Budget Authority by Activity
- Authorizing Legislation
- Appropriations History
- Justification of Budget Request
- Budget Authority by Object Class
- Salaries and Expenses
- Detail of Full-Time Equivalent Employment (FTE)
- Detail of Positions
- New Positions Requested
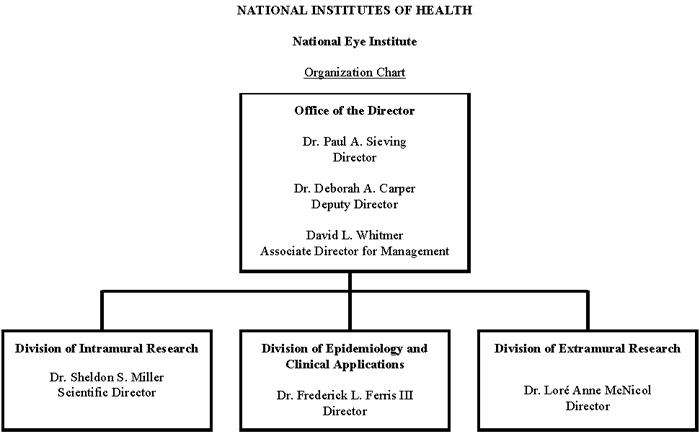
Organization Chart
Appropriation Language
For carrying out section 301 and title IV of the Public Health Services Act with respect to eye diseases and visual disorders $719,059,000.
NATIONAL INSTITUTES OF HEALTH
National Eye Institute
Amounts Available for Obligation 1
(Dollars in Thousands)
| Source of Funding | FY 2010 Actual | FY 2011 CR | FY 2012 PB |
|---|---|---|---|
| 1/ Excludes the following amounts for reimbursable activities carried out by this account: FY 2010- $16,007 FY 2011 - $17,060 FY 2012 - $17,060 Excludes $3,250 in FY 2010 for royalties. |
|||
| Appropriation | 707,036 | 707,036 | 719,059 |
| Type 1 Diabetes | 0 | 0 | 0 |
| Recession | 0 | 0 | 0 |
| Supplemental | 0 | 0 | 0 |
| Subtotal, adjusted appropriation | 707,036 | 707,036 | 719,059 |
| Real transfer under Director's one-percent transfer authority (GEI) | -1,130 | 0 | 0 |
| Real transfer under Secretary's one-percent transfer authority | -106 | 0 | Comparative transfer to NLM for NCBI and public Access | -288 | -601 | 0 | Comparative transfer under Director's one-percent transfer authority (GEI) | 1,130 | 0 | 0 |
| Subtotal, adjusted budget authority | 706,642 | 706,435 | 719,059 |
| Unobligated balance, start of year | 0 | 0 | 0 |
| Unobligated balance, end of year | 0 | 0 | 0 |
| Subtotal, adjusted budget authority | 706,642 | 706,435 | 719,059 |
| Unobligated balance lapsing | 0 | 0 | 0 |
| Total obligations | 706,642 | 706,435 | 719,059 |
NATIONAL INSTITUTES OF HEALTH
National Eye Institute
(Dollars in Thousands)
Budget Mechanism—Total 1
| MECHANISM | FY 2010 Actual | FY 2011 CR | FY 2012 PB | Change vs. FY2010 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number | Amount | Number | Amount | Number | Amount | Number | Amount | |||||
| 1. All items in italics are "non-adds": items in parenthesis are subtractions. | ||||||||||||
| Research Grants: | ||||||||||||
| Research Projects: | ||||||||||||
| Noncompeting | 854 | $335,229 | 814 | $317,520 | 765 | $308,620 | (89) | ($26,609) | ||||
| Administrative Supplements | 30 | 3,267 | 30 | 3,280 | 31 | 3,390 | 1 | 123 | ||||
| Competing: | ||||||||||||
| Renewal | 115 | 48,938 | 130 | 51,411 | 143 | 57,395 | 28 | 8,457 | ||||
| New | 133 | 47,470 | 150 | 59,458 | 166 | 66,224 | 33 | 18,754 | ||||
| Supplements | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Subtotal, Competing | 248 | $96,408 | 280 | $110,869 | 309 | $123,619 | 61 | $27,211 | ||||
| Subtotal, RPGs | 1,102 | $434,904 | 1,094 | $431,669 | 1,074 | $435,629 | (28) | $725 | ||||
| SBIR/STTR | 51 | $17,302 | 51 | $17,248 | 52 | $17,554 | 1 | $252 | ||||
| Research Project Grants | 1,153 | $452,206 | 1,145 | $448,917 | 1,126 | $453,183 | (27) | $977 | ||||
| Research Centers: | ||||||||||||
| Specialized/comprehensive | 40 | $27,604 | 40 | $27,604 | 40 | $27,880 | 0 | $276 | ||||
| Clinical research | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Biotechnology | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Comparative medicine | 0 | 150 | 0 | 150 | 0 | 150 | 0 | 0 | ||||
| Research Centers in Minority Institutions | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Research Centers | 40 | $27,754 | 40 | $27,754 | 40 | $28,030 | 0 | $276 | ||||
| Other Research: | ||||||||||||
| Research Careers | 64 | $12,696 | 64 | $13,053 | 65 | $13,184 | 1 | $387 | ||||
| Cancer Education | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Cooperative Clinical Research | 33 | 50,127 | 33 | 50,127 | 33 | 50,628 | 0 | 501 | ||||
| Biomedical Research Support | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Minority Biomedical Research Support | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Other | 18 | 12,059 | 18 | 12,059 | 18 | 12,180 | 0 | 121 | ||||
| Other Research | 115 | $74,983 | 115 | $75,239 | 116 | $75,992 | 1 | $1,009 | ||||
| Total Research Grants | 1,308 | $554,943 | 1,300 | $551,910 | 1,282 | $557,205 | (26) | $2,262 | ||||
| Research Training: | ||||||||||||
| Individual Awards | 67 FTTPs | $3,042 | 67 FTTPs | $3,103 | 67 FTTPs | $3,227 | 0 | $185 | ||||
| Institutional Awards | 187 FTTPs | $7,368 | 187 FTTPs | $7,515 | 187 FTTPs | $7,816 | 0 | 448 | ||||
| Total Research Training | 254 FTTPs | $10,410 | 254 FTTPs | $10,618 | 254 FTTPs | $11,043 | 0 FTP | $633 | ||||
| Research & Development Contracts | 43 | $45,900 | 44 | $46,371 | 44 | $52,299 | 1 | $6,399 | ||||
| (SBIR/STTR) | 0 | $34 | 0 | $185 | 0 | $185 | 0 | $151 | ||||
| Intramural Research | 169 FTEs | $71,529 | 169 FTEs | $72,960 | 169 FTEs | $73,960 | 0 FTEs | $2,161 | ||||
| Research Management and Support | 81 | 23,860 | 82 | 24,576 | 82 | 24,822 | 1 | 962 | Construction | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | Building and Facilities | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total, NEI | 250 | $706,642 | 251 | $706,435 | 251 | $719,059 | 1 | $12,417 | ||||
Major Changes in the Fiscal Year 2012 Budget Request
Major changes by budget mechanism and/or budget activity detail are briefly described below. Note that there may be overlap between budget mechanism and activity detail and these highlights will not sum to the total change for the FY 2012 budget request for NEI, which is $12.417 million more than the FY 2010 level, for a total of $719.059 million.
Research Project Grants (+$0.977 million, total $453.183 million): NEI will support a total of 1,126 Research Project Grants (RPGs) in FY 2012. Noncompeting RPGs will decrease by 89 awards and decrease by $26.609 million. Competing RPGs will increase by 61 awards and increase by $27.211 million. The NIH budget policy for RPGs in FY 2012 includes 1% inflationary increases in noncompeting awards and in the average cost for competing RPGs.
Research Training (+$0.633 million, total $11.043 million): NIH will provide an increase of 4% for stipends levels under the Ruth L. Kirschstein National Research Service Award training program to continue efforts to attain the stipend levels recommended by the National Academy of Sciences. This will build on the 2% increase in stipend levels for FY 2011. Stipend levels were largely flat for several years, and the requested increase will help to sustain the development of a highly qualified biomedical research workforce.
Research and Development Contracts (+$6.399 million, total $52.299 million): Funds are included in R&D contracts to reflect NEI's share of NIH-wide funding required to support several trans-NIH initiatives, such as the Therapies for Rare and Neglected Diseases program, the Basic Behavioral and Social Sciences Opportunity Network (OppNet), and support for a new synchrotron at the Brookhaven National Laboratory. For example, each IC that will benefit from the new synchrotron will provide funding to total NIH's commitment to support this new technology--$10 million.
NATIONAL INSTITUTES OF HEALTH
National Eye Institute
Summary of Changes
(Dollars in Thousands)
| FY 2010 Actual | $706,642 |
|---|---|
| FY 2012 Estimate | 719,059 |
| Net change | $12,417 |
NATIONAL INSTITUTES OF HEALTH
National Eye Institute
Summary of Changes (continued)
| CHANGES | 2012 Current Estimate | Change from FY 2010 | ||
|---|---|---|---|---|
| FTEs | Budget Authority | FTEs | Budget Authority | |
| A. Built-in: | ||||
| 1. Intramural research: | ||||
| a. Annualization of January 2010 pay increase | $27,393 | $165 | ||
| b. January FY 2012 pay increase | 27,393 | 0 | ||
| c. One less day of pay (n/a for 2011) | 27,393 | (106) | ||
| d. Payment for centrally furnished services | 11,246 | 111 | ||
| e. Increased cost of laboratory supplies, materials, and other expenses | 35,051 | 344 | ||
| Subtotal | $514 | |||
| 2. Research management and support: | ||||
| a. Annualization of January 2010 pay increase | $11,346 | $68 | ||
| b. January FY 2012 pay increase | 11,346 | 0 | ||
| c. One less day of pay (n/a for 2011) | 11,346 | (44) | ||
| d. Payment for centrally furnished services | 4,165 | 41 | ||
| e. Increased cost of laboratory supplies, materials, and other expenses | 9,311 | 91 | ||
| Subtotal | $156 | |||
| Subtotal, Built-in | $670 | |||
NATIONAL INSTITUTES OF HEALTH
National Eye Institute
Summary of Changes (continued)
| CHANGES | 2012 Estimate | Change from FY 2010 | ||
|---|---|---|---|---|
| Number | Amount | Number | Amount | |
| B. Program: | ||||
| 1. Research Project Grants: | ||||
| a. Noncompeting | 765 | $312,010 | (89) | ($26,486) |
| b. Competing | 309 | 123,619 | 61 | 27,211 |
| c. SBIR/STTR | 52 | 17,554 | 1 | 252 |
| Total | 1,126 | $453,183 | (27) | $977 |
| 2. Research Centers | 40 | $28,030 | 0 | $276 | 3. Other Research | 116 | 75,992 | 1 | 1,009 |
| 4. Research Training | 254 | 11,043 | 0 | 633 |
| 5. Research and Development Contracts | 44 | 52,299 | 1 | 6,399 |
| Subtotal, Extramural | $620,547 | $9,294 | ||
| 6. Intramural Research | 169 FTEs | $73,690 | 0 FTEs | 1,647 |
| 7. Research Management and Support | 82 FTEs | 24,822 | 1 FTEs | 806 |
| 8. Construction | FTEs | 0 | FTEs | 0 |
| 9. Buildings and Facilities | FTEs | 0 | FTEs | 0 |
| Subtotal, program | 251 | $719,059 | 1 | $11,747 |
| Total changes | $719,059 | $12,417 | ||
Budget Graphs
History of Budget Authority and FTEs:
Full-Time Employees by Fiscal Year
| Fiscal Year | FTEs |
|---|---|
| 2008 | 230 |
| 2008 | 241 |
| 2010 | 250 |
| 2011 | 251 |
| 2012 | 251 |
Funding Levels by Fiscal Year
| Fiscal Year | Dollars in Millions |
|---|---|
| 2008 | 670.7 |
| 2009 | 688.3 |
| 2010 | 706.6 |
| 2011 | 706.4 |
| 2012 | 719.1 |
Distribution by Mechanism:
FY 2012 Budget Mechanism
| Mechanism | Percent of Budget |
|---|---|
| Research Project Grants | 63 |
| RM&S | 3 |
| Other Research | 11 |
| R&D Contracts | 7 |
| Intramural Research | 10 |
| Research Training | 2 |
| Research Centers | 4 |
Change by Selected Mechanism:
FY 2012 Estimate
Percent Change from FY 2010 Mechanism
| Mechanism | Percent Change |
|---|---|
| Research Project Grants | 0.2 |
| Research Centers | 1.0 |
| Other Research | 1.3 |
| Research Training | 6.1 |
| R&D Contracts | 13.9 |
| Intramural Research | 3.0 |
| Res. Mgmt. & Support | 4.0 |
NATIONAL INSTITUTES OF HEALTH
National Eye Institute
Budget Authority by Activity
(Dollars in Thousands)
| FY 2010 Actual | FY 2011 CR | FY 2012 PB | Change vs. FY 2010 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FTEs | Amount | FTEs | Amount | FTEs | Amount | FTEs | Amount | |||||||
| 1. Includes FTEs which are reimbursed from the NIH Common Fund for Medical Research. 2. Includes Real Transfers and Comparable Adjustments as detailed in the "Amounts Available for Obligation" table. | ||||||||||||||
| Extramural Research | ||||||||||||||
| Detail: | ||||||||||||||
| Retinal Disease Research | $272,317 | $271,268 | $276,458 | $4,141 | ||||||||||
| Corneal Diseases, Cataract, and Glaucoma Research | 174,370 | 173,698 | 177,020 | 2,650 | ||||||||||
| Sensorimotor Disorders and Rehabilitation Research | 164,566 | 163,933 | 167,069 | 2,503 | ||||||||||
| Subtotal, Extramural | $611,253 | $608,899 | $620,547 | $9,294 | ||||||||||
| Intramural Research | 169 | $71,529 | 169 | $72,960 | 169 | $73,690 | 0 | $2,161 | ||||||
| Research Management & Support | 81 | $23,860 | 82 | $24,576 | 82 | $24,822 | 1 | $962 | ||||||
| TOTAL | 250 | $706,642 | 251 | $706,435 | 251 | $719,059 | 1 | $12,417 | ||||||
NATIONAL INSTITUTES OF HEALTH
National Eye Institute
Authorizing Legislation
| PHS Act/Other Citation | U.S. Code Citation | 2011 Amount Authorized | FY 2010 Actual | 2012 Amount Authorized | FY 2012 PB | |
|---|---|---|---|---|---|---|
| Research and Investigation | Section 301 | 42§241 | Indefinite | Indefinite | ||
| National Eye Institute | Section 401(a) | 42§281 | Indefinite | Indefinite | ||
| Total, Budget Authority | 706,642,000 | 719,059,000 |
Appropriations History
| Fiscal Year |
Budget Estimate to Congress |
House Allowance |
Senate Allowance |
Appropriation |
|---|---|---|---|---|
| 1/ Reflects enacted supplementals, rescissions, and reappropriations. | ||||
| 2/ Excludes funds for HIV/AIDS research activities consolidated in the NIH Office of AIDS Research. | ||||
| 2003 | $625,666,000 | $625,666,000 | $637,290,000 | 581,366,000 |
| Rescission | ($4,142,000) | |||
| 2004 | $652,738,000 | $648,299,000 | $657,199,000 | $657,199,000 |
| Rescission | ($4,147,000) | |||
| 2005 | $671,578,000 | $671,578,000 | $680,300,000 | $674,578,000 |
| Rescission | ($5,508,000) | |||
| 2006 | $673,491,000 | $673,491,000 | $693,559,000 | $673,491,000 |
| Rescission | ($6,735,000) | |||
| 2007 | $661,358,000 | $661,358,000 | $666,898,000 | $667,166,000 |
| Rescission | $0 | |||
| 2008 | $667,820,000 | $677,039,000 | $681,962,000 | $678,978,000 |
| Rescission | ($11,862,000) | |||
| Supplemental | $3,548,000 | |||
| 2009 | $667,764,000 | $690,721,000 | $687,346,000 | $688,276,000 |
| Rescission | $0 | |||
| 2010 | $695,789,000 | $713,072,000 | $700,158,000 | $707,036,000 |
| Rescission | $0 | |||
| 2011 | $724,360,000 | $723,220,000 | ||
| Rescission | ||||
| 2012 | $719,059,000 | |||
Justification of Budget Request
National Eye Institute
Authorizing Legislation: Section 301 and title IV of the Public Health Service Act, as amended. Budget Authority (BA):
| FY 2010 Actual | FY 2011 Continuing Resolution | FY 2012 Budget Request | Fy 2012 +/- FY 2010 | |
|---|---|---|---|---|
| BA | $706,642,000 | $706,435,000 | $719,059,000 | $12,417,000 |
| FTE | 250 | 251 | 251 | 1 |
Program funds are allocated as follows: Competitive Grants/Cooperative Agreements; Contracts; Direct Federal/Intramural and Other.
Director's Overview
The National Eye Institute (NEI) was established by Congress in 1968 to protect and prolong the vision of the American people. NEI research leads to sight-saving treatments, reduces visual impairment and blindness, and improves the quality of life for people of all ages. NEI supports vision research through approximately 1,600 research grants and training awards made to scientists at more than 250 medical centers, hospitals, and universities across the country and around the world. NEI also conducts laboratory and patient-oriented research at its own facilities located on the NIH campus in Bethesda, Maryland.
Accelerating Discovery with New Technologies
The causes of common diseases are complex in that there are potentially many different environmental factors and genetic variants that can contribute to disease. New technologies such as genome-wide association studies (GWAS) allow investigators to scan the genomes of patients to identify genetic risk variants for common diseases. Individually, each of these variants may only contribute to a small percentage of cases, so GWAS often require many subjects to identify low frequency risk variants. In the largest GWAS study in vision research to date, NEI investigators recently sequenced DNA from over 18,000 patients and control subjects and identified three new genes associated with age-related macular degeneration (AMD), the most common cause of vision loss in older Americans. Two of these genes are involved with high-density lipoprotein cholesterol metabolism, implicating a new biochemical pathway involved in the pathogenesis of AMD. These findings will allow researchers to better understand the disease mechanisms underlying AMD and develop therapies that address the root cause of vision loss.
In FY 2010, NEI established the NEI Glaucoma Human Genetics Collaboration (NEIGHBOR), a consortium of clinicians and geneticists at 12 institutions throughout the U.S. dedicated to identifying the genetics of glaucoma. NEIGHBOR has collected and is now sequencing 6000 DNA samples for large-scale GWAS. The data from these DNA samples are expected to be released to the vision research community in 2011.
Refractive errors (nearsightedness and farsightedness) are the leading cause of vision impairment in the world, affecting an estimated two billion people and growing. Severe nearsightedness increases the risk of other sight-threatening conditions such as retinal detachment, macular degeneration, cataract, and glaucoma. Although refractive error is heritable, no genes have been identified yet. Two recent GWAS studies involving over 15,000 subjects identified a susceptibility locus for refractive errors on chromosome 15 that may harbor regulatory elements for two genes expressed in the retina. The next step is to study the role of these genes in refractive error with the ultimate goals of understanding the causes of refractive error and developing preventative treatments.
Therapeutic Development Pipeline
Positive results of ongoing clinical trials of gene therapy for Leber congenital amaurosis, a severe, early onset retinal disease, have encouraged applications of this approach to many other eye diseases. In the past year, NEI investigators demonstrated proof-of-concept of gene therapy in animal models of AMD, achromatopsia, Leber's hereditary optic neuropathy, retinitis pigmentosa, and red-green color blindness. Previous work has established the utility of gene therapy in juvenile retinoschisis, optic neuritis, and Stargardt disease. These studies now allow investigators to conduct the pre-clinical work necessary to pursue regulatory approval for clinical trials. In addition, novel gene delivery systems, such as the use of nanoparticles, have shown promise in animal models. Such vectors will be helpful in expanding the reach of gene therapy to target a variety of ocular tissues such as ganglion and photoreceptor cell layers.
Clinical Trials of Comparative Effectiveness
Diabetic macular edema (DME) is a common sight-threatening complication of diabetes in which fluid from leaky blood vessels causes the retina to swell. For the past 25 years, DME has been treated with a laser to destroy abnormal blood vessels. Although laser therapy slows disease progression, the effects are temporary, and repeated treatments can damage healthy retinal tissue and impair vision. In recent years, ophthalmologists have been supplementing laser treatment with ocular injections of either ranibizumab, a drug that prevents blood vessel growth, or triamcinolone, a corticosteroid to reduce inflammatory complications. An ongoing clinical trial comparing the safety and efficacy of these two drugs is being conducted by the NEI-supported Diabetic Retinopathy Clinical Research Network (DRCR.net). After one year, ranibizumab + laser treatment was superior in both safety and efficacy to triamcinolone + laser or to laser alone. The triamcinolone treatment group also experienced a far greater incidence of cataract and glaucoma. This landmark clinical trial identified the first new safe and effective treatment regimen for DME in more than two decades. In addition, the study demonstrated that intravitreal triamcinolone, which had been used in 60 percent of patients with DME, had significant side effects and was not better than laser alone. These results are already being used by community ophthalmologists to greatly improve the quality of life for people living with diabetes.
The treatment of cataracts in infants is challenging for pediatric ophthalmologists and parents. Removing and substituting for the opaque lens is critical to preventing permanent loss of vision in the eye. Contact lenses have been the preferred method to overcome the loss of the naturallens. However, it is difficult and stressful for parents to insert a contact lens into an infant's eye. Surgical implantation of intraocular lenses (IOLs) in adults is common but had not been fully characterized in infants. An NEI-supported clinical trial found no difference in visual acuity with contact lenses compared to IOLs at one year after cataract removal. However, IOLs caused significant numbers of surgical complications. Based on these results, the use of contact lenses has been determined to be the safest effective treatment for infants with cataract.
New Ideas and New Investigators in Vision Research
In 2011, NEI will renew its national plan to identify current needs, gaps, and opportunities in research to improve eye health. NEI is reinvigorating its strategic planning process which consists of three new components: 1) assembling expert panels in all six major program areas to develop a national plan; 2) developing an implementation guide to describe NEI operations, policies, and priorities; and 3) periodically convening workshops that address emerging needs and opportunities in vision research. A recent multidisciplinary NEI workshop on Ocular Pain and Sensitivity was the first in this series. Experts in ocular surface disease met with experts in pain and sensitivity in more thoroughly studied non-ocular tissues (e.g., itch in skin; headache pain; burning mouth) in order to gain knowledge about the neural mechanisms of pain. A workshop report has been posted to the NEI Web site. These workshops will draw attention to emerging scientific and clinical needs, identify emerging biomedical research areas, and attract new investigators into vision research.
Overall Budget Policy: The FY 2012 request for NEI is $719.059 million, an increase of $12.417 million or 1.8% over the FY 2010 Actual level. NEI will continue to support new investigators and to maintain an adequate number of competing RPGs. NEI will provide a 1% inflationary increase for non-competing and competing grants, and continue funding the highest quality investigator-initiated research as evaluated through the peer review system.
Program Descriptions and Accomplishments
Retinal Diseases Research: The light-sensitive retina is susceptible to many sight-threatening conditions, including age-related macular degeneration, diabetic retinopathy, retinopathy of prematurity, retinitis pigmentosa, Usher's syndrome, ocular albinism, retinal detachment, uveitis (inflammation), and eye cancer. The goals of this program are to increase the understanding of disease mechanisms that cause vision loss and to develop improved methods of prevention, diagnosis, and treatment. To meet these goals, NEI supports research on the cell biology, physiology, and immunology of the retina and on the role of gene expression, gene regulation, and the environment in retinal health and disease. NEI investigators have identified gene variants for many of these diseases and have made significant progress in discovering the underlying biological mechanisms of vision loss. With this knowledge, efforts are now increasingly focused on translational research to advance novel gene-based therapies to clinical trials.
Budget Policy: The FY 2012 budget estimate for these activities is $276.458 million, an increase of $4.141 million or 1.5% over the FY 2010 Actual. FY 2012 program plans will focus on an acceleration of research on the genetic and environmental basis for AMD, including the role of possible immunological factors. This effort will include meta-analysis of genome wide association studies and related efforts in bioinformatics. NEI will support projects that address the possible restoration of vision in retinal degenerative diseases by building on recent advances in gene transfer, stem cell biology, photosensitive replacement molecules, and visual prostheses. Research will continue in efforts to control abnormal blood vessel growth (angiogenesis) in several eye diseases. Program plans also include the continuation of the Age-Related Eye Disease Study 2 (AREDS2), a multi-center study to evaluate the use of additional oral supplements for the treatment of AMD and cataract. NEI will also support an ocular follow-up study of patients in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) Trial to gain important information on the long-term progression of diabetic retinopathy.
Program Portrait: Translational Research Expansion
FY 2010 Level: $5.4 million
FY 2012 Level: $6.3 million
Change: $0.9 million
NEI investigators have made significant progress in understanding underlying disease mechanisms that result in vision loss and have established proof-of-concept in animal models for several novel therapies. NEI is enhancing its translational research program by increasing support of a grant mechanism, the R24, specifically designed to support collaborative, multidisciplinary research programs focused on new therapeutic approaches such as gene therapy, pharmacological approaches, and development of appropriate delivery systems.
New blood vessel growth, or neovascularization, is an important developmental process, but abnormal neovascularization is a common cause of vision loss in diabetic retinopathy and age-related macular degeneration. Inhibition of neovascularization in these diseases prevents catastrophic vision loss by preventing complications such as ocular hemorrhage and edema. NEI investigators have discovered a naturally occurring enzyme with potent anti- neovascular properties. NEI awarded an R24 grant to further characterize and optimize the enzyme's inhibition of neovascularization as well as to develop a drug delivery system for use in clinical trials. Work supported by two other R24 awards will be evaluating small molecules to treat Stargardt disease, an early onset form of macular degeneration. Stargardt disease results from a defect in the visual cycle, the set of chemical interactions that convert light into an electrical signal that is then processed by the brain. This defect leads to accumulation of harmful vitamin A by-products in the retina. One grant is evaluating a compound that slows the visual cycle to limit the accumulation of vitamin A metabolites. It is thought that this approach could result in long-term preservation of vision. A second grant proposes to screen small molecules for their therapeutic potential and to develop a gene transfer technique for treatment of Stargardt disease.
Corneal Diseases, Cataract, and Glaucoma Research: Corneal diseases, cataract, and glaucoma cause more visits to ophthalmologists than any other vision disorders. Corneal injuries, infections, and diseases can be extremely painful and require immediate medical attention. NEI grantees are exploring how infectious, inflammatory, and immunological processes affect the cornea, and how the cornea heals after a wound. Important proteins that promote and deter wound healing have been identified, providing an opportunity to develop therapies that prevent or ameliorate corneal disease. Worldwide, cataracts are the leading cause of blindness. NEI cataract research seeks to understand the physiological basis of lens transparency at the cellular and molecular levels and investigates strategies to prevent cataract formation and progression. Glaucoma is a blinding disease that most often results from increased intraocular pressure. NEI investigators aim to understand the complex genetic and biological factors that cause the disease and to develop treatments that protect optic nerves from the damage that leads to vision loss.
Budget Policy: The FY 2012 budget estimate for these activities is $177.020 million, an increase of $2.650 million or 1.5% over the FY 2010 Actual. FY 2012 program plans include following up on a recent finding that certain receptors that bind to vascular endothelial growth factor may play an important role in maintaining the normal transparency of the cornea. NEI will continue to fund projects that study the cellular, biochemical, biophysical and molecular bases of lens transparency and its relation to cataractogenesis. Furthermore, proteins highly enriched in the ocular lens (such as crystallins, connexins, and aquaporins) will continue to be investigated with a particular focus on their role in cataract development. NEI expects to fund new projects to identify therapeutic approaches to limit and/or reduce corneal pain. Projects will be funded to examine the possible contribution of defects in gap junctions in the development of cataracts. Genome wide association studies and related bioinformatics efforts will continue to explore the role of genetics and the environment on the development of glaucoma and to understand better the differential response of individuals to glaucoma medications. Other projects will relate to optic nerve regeneration and functional re-connection of the injured optic nerve to the brain.
Program Portrait: Pluripotent Stem Cells in Ocular Regenerative Medicine
FY 2010 Level: $0.0 million
FY 2012 Level: $1.0 million
Change: $1.0 million
Regenerative medicine is an emerging field that holds promise to restore lost vision due to injury or disease. Many of the leading causes of visual impairment and blindness are due in part to the loss of photoreceptor cells in the retina or either a loss of cells or opacities in the normally transparent cornea. While there is some evidence that embryonic retinal cells can differentiate into photoreceptors, thus far they do not integrate well with the host retina and are not yet suitable for therapeutic use. Likewise, although stem cells derived from the outer ring of the corneal epithelium have had limited success in replacing damaged corneal tissue, more research is needed to expand the utility of stem cells in generating replacements for the corneal epithelium and other layers of the cornea.
NEI plans to augment its existing regenerative medicine portfolio. Applications will be solicited to 1) encourage research to better understand the factors that promote differentiation of pluripotent stem cells into retinal and corneal cells; 2) promote methods to create in vitro cell cultures that facilitate self-renewal and differentiation of these stem cells as possible sources for transplantation; and 3) foster the discovery of factors that direct stem cells to the target tissue and promote their integration in order to repair/treat the retina or the cornea.
Sensorimotor Disorders, Visual Processing, and Rehabilitation Research: Strabismus (misalignment of the eyes) and amblyopia (known as "lazy eye") are common disorders that develop during childhood. Program goals center on gaining a better understanding of the neuromuscular control of gaze and the development of the visual system in children at high risk for these disorders. Refractive errors, such as nearsightedness, farsightedness, and astigmatism, are commonly correctable with eye glasses or contact lenses in the United States, but remain a tremendous economic and personal burden globally. Major goals of this program are to discover the biochemical pathways that govern eye growth and to uncover the risk factors associated with refractive errors. Much of the cerebral cortex is devoted to processing the visual information that floods our eyes. Vision scientists seek to understand how the brain processes visual information, how neural activity is related to visual perception, and how the visual system interacts with cognitive and motor systems. Low vision is the term used to describe chronic visual conditions that are not correctable by eye glasses or contact lenses. NEI supports rehabilitation research on improving the quality of life of persons with visual impairments by helping them maximize theuse of remaining vision and by devising improved aids and strategies to assist those without useful vision.
Budget Policy: The FY 2012 budget estimate for these activities is $167.069 million, an increase of $2.503 million or 1.5% over the FY 2010 Actual. FY 2012 program plans include pursuing research to identify genetic risk factors for strabismus, myopia, and other ocular diseases. Gene therapy trials are underway for treating Leber's Hereditary Optic Neuropathy, an inherited disease that frequently results in a substantial loss of central vision. The Neuro-Ophthalmology Research Disease Investigator consortium will expand to include many new sites in order to facilitate patient recruitment and the conduct of clinical trials. Investigators will also pursue new findings about how the activity of certain brain cells allows us to perceive faces and objects and allow us to provide a stable view of our surroundings despite constant head and eye movements. A better understanding of the rules of visual system development will lead to new strategies for improving binocular vision in amblyopia and for stimulating the recovery of function in other ocular diseases. Further advances are expected in the development of retinal implants and other neural interfaces to provide artificial stimulation of visual pathways and the use of brain electrical activity to control computer displays and robotic arms.
Intramural Research: NEI conducts world-class research programs that explore clinical and translational studies concerned with the cause, prevention, and treatment of major eye diseases and vision disorders; cellular and molecular mechanisms of eye development, including the expression and function of genes within the eye; immunology and infectious diseases of the eye; the mechanisms of visual perception by the brain; and developing a better understanding of our ability to guide movements under sensory control. The National Ophthalmic Disease Genotyping Network (eyeGENE), an NEI intramural collaboration with patients, clinicians, and investigators throughout the United States, has expanded its operations by collecting more than 2200 patient DNA samples. eyeGENE enables patients to receive a genetic diagnosis for many rare eye diseases in exchange for donating DNA samples for research and participating in a clinical trial registry Through this unique collaboration, eyeGENE is enhancing patient care, education and research.
Budget Policy: The FY 2012 budget estimate for these activities is $73.690 million, an increase of $2.161 million or 3% over the FY 2010 Actual. In FY 2012, NEI will continue recruiting investigators for the Laboratory of Neurobiology, Neurodegeneration and Repair to integrate basic, pre-clinical, and translation research in developing and testing therapeutic interventions in neurodegenerative eye diseases. NEI is building its capacity in pre-clinical animal model evaluation and development, expanding its Genetic Engineering Facility in support of a new initiative to develop human embryonic stem cells for therapeutic use in retinal degenerative and other ocular diseases, and developing a new animal Visual Function Facility with expertise in electroretinography and optical coherence tomography. NEI is also recruiting for a new laboratory in computational medicine and plans to enhance electroretinography and optical coherence tomography to assess patients in the Ophthalmic Genetics and Visual Function Branch. NEI continues to expand the eyeGENE network to facilitate research on the genetic causes of ocular diseases.
Program Portrait: Bioinformatics Support and Resource Center for Vision Research
FY 2010 Level: $0.0 million
FY 2012 Level: $3.0 million
Change: $3.0 million
New medical research technologies are evolving rapidly, leading to an exponential increase in the volume of data generated from genomics, proteomics, and metabolomics studies. These data need to be matched to genetic information (genotype) and disease characteristics from patients (phenotype) with eye diseases. The immense quantity of information will require novel and sophisticated mathematical and statistical methods to help uncover the nature of these complex interactions in health and disease.
To meet these challenges, NEI initiated a three-pronged approach this past year with a new institutional training grant that partners vision researchers with statistical and mathematical scientists to train the next generation of vision researchers in computational methods. NEI is also soliciting applications from investigators to develop novel computational and bioinformatics approaches and tools to analyze large data sets of genomic and phenotypic data. To increase availability and accessibility of these data sets, in the coming year NEI plans to establish a bioinformatics resource center for sharing diverse sets of biological data and research tools for vision research. The infrastructure developed under this program will support the various needs and activities of both intramural and extramural vision research scientists and will facilitate computational biology/bioinformatics efforts.
Research Management and Support: Research Management and Support (RMS) sustains, guides, and monitors the extramural and intramural research programs. Included in these funds are the support necessary for personnel to carry out leadership and management functions, human resource support, training, travel, purchasing, facilities, budget, planning, information technology, and extramural grant award and management. NEI currently oversees more than 1,600 grants and contracts, including research project grants, core center grants, research career development awards, cooperative clinical research agreements, and research and development contracts.
Budget Policy: The FY 2012 budget estimate for these activities is $24.822 million, an increase of $0.962 million or 4% over the FY 2010 Actual to continue to enhance the activities described above.
NATIONAL INSTITUTES OF HEALTH
National Eye Institute
Budget Authority by Object
(Dollars in Thousands)
| Total compensable workyears | FY 2010 Enacted | FY 2012 PB | Increase or Decrease | Percent Change |
|---|---|---|---|---|
| Includes FTEs which are reimbursed from the NIH Common Fund for Medical Research | ||||
| Full-time employment | 250 | 251 | 1 | 0.4% |
| Full-time equivalent of overtime and holiday hours | 0 | 0 | 0 | 0% |
| Average ES salary | $167 | $167 | $0 | 0% |
| Average GM/GS grade | 12.4 | 12.4 | 0.0 | 0% |
| Average GM/GS salary | $102 | $102 | $0 | 0% |
| Average salary, grade established by act of July 1, 1944 (42 U.S.C. 207) | $90 | $90 | $0 | 0% |
| Average salary of ungraded positions | 123 | 123 | 0 | 0% |
Budget Authority by Object
(continued)
| OBJECT CLASSES | FY 2010 Estimate | FY 2012 Estimate | Increase or Decrease | Percent Change |
|---|---|---|---|---|
| Includes FTEs which are reimbursed from the NIH Roadmap for Medical Research | ||||
| Personnel Compensation: | ||||
| 11.1 Full-time permanent | $15,038 | $15,164 | $126 | 0.8% |
| 11.3 Other than full-time permanent | 11,111 | 11,149 | 38 | 0.3% |
| 11.5 Other personnel compensation | 978 | 983 | 5 | 0.5% |
| 11.7 Military personnel | 253 | 262 | 9 | 3.6% |
| 11.8 Special personnel services payments | 3,251 | 3,258 | 7 | 0.2% |
| Total, Personnel Compensation | $30,631 | $30,816 | $185 | 0.6% |
| 12.0 Personnel benefits | $7,691 | $7,731 | $40 | 0.5% |
| 12.2 Military personnel benefits | 192 | 192 | 0 | 0% |
| 13.0 Benefits for former personnel | 0 | 0 | 0 | 0% |
| Subtotal, Pay Costs | $38,514 | $38,739 | $225 | 0.6% |
| 21.0 Travel and transportation of persons | $857 | $935 | $78 | 9.1% |
| 22.0 Transportation of things | 77 | 83 | 6 | 7.8% |
| 23.1 Rental payments to GSA | 0 | 0 | 0 | 0% |
| 23.2 Rental payments to others | 9 | 9 | 0 | 0% |
| 23.3 Communications, utilities and miscellaneous charges | 525 | 583 | 58 | 11.0% |
| 24.0 Printing and reproduction | 70 | 77 | 7 | 10% |
| 25.1 Consulting services | 46 | 52 | 6 | 13% |
| 25.2 Other services | 7,030 | 7,641 | 611 | 8.7% |
| 25.3 Purchase of goods and services from government accounts | 57,008 | 64,368 | 7,360 | 12.9% |
| 25.4 Operation and maintenance of facilities | 1,085 | 1,189 | 104 | 9.6% |
| 25.5 Research and development contracts | 24,595 | 24,687 | 92 | 0.4% |
| 25.6 Medical care | 138 | 149 | 11 | 8% |
| 25.7 Operation and maintenance of equipment | 4,434 | 4,828 | 394 | 8.9% |
| 25.8 Subsistence and support of persons | 0 | 0 | 0 | 0% |
| 25.0 Subtotal, Other Contractual Services | $94,336 | $102,914 | $8,578 | 9.1% |
| 26.0 Supplies and materials | $4,383 | $4,738 | $355 | 8.1% |
| 31.0 Equipment | 2,516 | 2,731 | 215 | 8.5% |
| 32.0 Land and structures | 0 | 0 | 0 | 0% |
| 33.0 Investments and loans | 0 | 0 | 0 | 0% |
| 41.0 Grants, subsidies and contributions | 565,353 | 568,248 | 2,895 | 0.5% |
| 42.0 Insurance claims and indemnities | 0 | 0 | 0 | 0% |
| 43.0 Interest and dividends | 2 | 2 | 0 | 0% |
| 44.0 Refunds | 0 | 0 | 0 | 0% |
| Subtotal, Non-Pay Costs | $668,128 | $680,320 | $12,192 | 1.8% |
| Total Budget Authority by Object | $706,642 | $719,059 | $12,417 | 1.8% |
NATIONAL INSTITUTES OF HEALTH
National Eye Institute
Salaries and Expenses
(Dollars in Thousands)
| OBJECT CLASSES | FY 2010 Actual | FY 2012 PB | Increase or Decrease | Percent Change |
|---|---|---|---|---|
| Personnel Compensation: | ||||
| Full-time permanent (11.1) | $15,038 | $15,164 | $126 | 0.8% |
| Other than full-time permanent (11.3) | 11,111 | 11,149 | 38 | 0.3% |
| Other personnel compensation (11.5) | 978 | 983 | 5 | 0.5% |
| Military personnel (11.7) | 253 | 262 | 9 | 3.6% |
| Special personnel services payments (11.8) | 3,251 | 3,258 | 7 | 0.2% |
| Total Personnel Compensation (11.9) | $30,631 | $30,816 | $185 | 0.6% |
| Civilian personnel benefits (12.1) | $7,691 | $7,731 | $40 | 0.5% |
| Military personnel benefits (12.2) | 192 | 192 | 0 | 0% |
| Benefits to former personnel (13.0) | 0 | 0 | 0 | 0% |
| Subtotal, Pay Costs | $38,514 | $38,739 | $225 | 0.6% |
| Travel (21.0) | $857 | $935 | $78 | 9.1% |
| Transportation of things (22.0) | 77 | 83 | 6 | 7.8% |
| Rental payments to others (23.2) | 9 | 9 | 0 | 0% |
| Communications, utilities and miscellaneous charges (23.3) | 525 | 583 | 58 | 11% |
| Printing and reproduction (24.0) | 70 | 77 | 7 | 10% |
| Other Contractual Services: | ||||
| Advisory and assistance services (25.1) | 46 | 52 | 6 | 13% |
| Other services (25.2) | 7,030 | 7,641 | 611 | 8.7% |
| Purchases from government accounts (25.3) | 37,773 | 39,114 | 1,341 | 3.6% |
| Operation and maintenance of facilities (25.4) | 1,085 | 1,189 | 104 | 9.6% |
| Operation and maintenance of equipment (25.7) | 4,434 | 4,828 | 394 | 8.9% |
| Subsistence and support of persons (25.8) | 0 | 0 | 0 | 0% |
| Subtotal Other Contractual Services | $50,368 | $52,824 | $2,456 | 4.9% |
| Supplies and materials (26.0) | $4,330 | $4,681 | $351 | 8.1% |
| Subtotal, Non-Pay Costs | $56,236 | $59,192 | $2,956 | 5.3% |
| Total, Administrative Costs | $94,750 | $97,931 | $3,181 | 3.4% |
Detail of Full-Time Equivalent employment (FTE)
| Office/Division | FY 2010 Actual | FY 2011 CR | FY 2012 PB | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Civilian | Military | Total | Civilian | Military | Total | Civilian | Military | Total | |
|
Includes FTEs which are reimbursed from the NIH Common Fund for Medical Research FTEs supported by funds from Cooperative Research and Development Agreements, 0 |
|||||||||
| Office of the Director | 79 | 3 | 82 | 80 | 3 | 83 | 80 | 3 | 83 |
| Division of Intramural Research Programs | 125 | 0 | 125 | 125 | 0 | 125 | 125 | 0 | 125 |
| Division of Epidemiology and Clinical Applications | 12 | 0 | 12 | 12 | 0 | 12 | 12 | 0 | 12 |
| Division of Extramural Research | 31 | 0 | 31 | 31 | 0 | 31 | 31 | 0 | 31 |
| Total | 247 | 3 | 250 | 248 | 3 | 251 | 248 | 3 | 251 |
Details of Full-Time Equivalent Employment (FTEs)
(continued)
| FISCAL YEAR | Average GM/GS Grade |
|---|---|
| 2008 | 12.0 |
| 2009 | 12.1 |
| 2010 | 12.4 |
| 2011 | 12.4 |
| 2011 | 12.4 |
Detail of Positions
| Grade | FY 2010 Actual | FY 2011 CR | FY 2012 PB |
|---|---|---|---|
| Total, ES Positions | 1 | 1 | 1 |
| Total, ES Salary | 167 | 167 | 167 |
| GM/GS-15 | 35 | 36 | 36 |
| GM/GS-14 | 18 | 18 | 18 |
| GM/GS-13 | 29 | 29 | 29 |
| GS-12 | 26 | 26 | 26 |
| GS-11 | 35 | 35 | 35 |
| GS-10 | 0 | 0 | 0 |
| GS-9 | 10 | 10 | 10 |
| GS-8 | 5 | 5 | 5 |
| GS-7 | 4 | 4 | 4 |
| GS-6 | 1 | 1 | 1 |
| GS-5 | 0 | 0 | 0 |
| GS-4 | 0 | 0 | 0 |
| GS-3 | 0 | 0 | 0 |
| GS-2 | 0 | 0 | 0 |
| GS-1 | 0 | 0 | 0 |
| Subtotal | 163 | 164 | 164 |
| Grades established by Act of July 1, 1944 (42 U.S.C. 207): | |||
| Assistant Surgeon General | 0 | 0 | 0 |
| Director Grade | 0 | 0 | 0 |
| Senior Grade | 2 | 2 | 2 |
| Full Grade | 1 | 1 | 1 |
| Senior Assistant Grade | 0 | 0 | 0 |
| Assistant Grade | 0 | 0 | 0 |
| Subtotal | 3 | 3 | 3 |
| Ungraded | 87 | 87 | 87 |
| Total permanent positions | 165 | 165 | 166 |
| Total positions, end of year | 254 | 255 | 255 |
| Total full-time equivalent (FTE) employment, end of year | 250 | 251 | 251 |
| Average ES salary | 167 | 167 | 167 |
| Average GM/GS grade | 12.4 | 12.4 | 12.4 |
| Average GM/GS salary | 102 | 102 | 102 |
New Positions Requested
| FY 2012 | |||
|---|---|---|---|
| Grade | Number | Annual Salary | |
| Health Science Administrator, Translational Research | GS-15 | 1 | $150,000 |
| Total Requested | 1 | $150,000 | |

