Home
- Destinations
- Vaccinations
- News & Announcements
- Travel Notices
- Diseases
- Yellow Book
- Find a Clinic
- Specific Groups & Settings
- Seasonal Flu & Travel
- Earthquake, Tsunami, and Radiation Release in Japan: Travel Information
- Traveling with Children
- Special Needs
- Disaster Relief
- Avian Flu & Travel
- Air & Cruise Ship Travel
- Air Travel Information For Travelers
- Cruise Ship Information for Travelers
- For Industry: Air
- CDC Measles Guidance for Commercial Aircraft Operators
- Interim Guidance for Protecting Travelers on Commercial Aircraft Serving Haiti During the Cholera Outbreak
- Reporting Onboard Deaths or Illnesses to CDC
- Onboard Death and Illness Response Tool for Cabin Crew
- Onboard Death and Illness Reporting Tool for Pilots
- CDC Guidance for Commercial Aircraft Operators: Seasonal Influenza
- Infection Control Guidelines for Cabin Crew Members on Commercial Aircraft
- For Industry: Cruise Ships
- Stay Healthy & Safe
- Illness & Injury Abroad
- Resources & Training
- Travel Podcasts
- RSS Feeds
Related Links
Chapter 3Infectious Diseases Related To Travel
Hepatitis A
Umid M. Sharapov
INFECTIOUS AGENT
Hepatitis A virus (HAV) is an RNA virus classified as a picornavirus.
MODE OF TRANSMISSION
Transmission can occur through direct person-to-person contact; through exposure to contaminated water, ice, or shellfish harvested from sewage-contaminated water; or from fruits, vegetables, or other foods that are eaten uncooked and that were contaminated during harvesting or subsequent handling. HAV is shed in the feces of infected people. The virus reaches peak levels 1–2 weeks before onset of symptoms and diminishes rapidly after liver dysfunction or symptoms appear, which is concurrent with the appearance of circulating antibodies to HAV. Infants and children, however, may shed virus for up to 6 months after infection.
EPIDEMIOLOGY
Worldwide, geographic areas can be characterized by high, intermediate, low, or very low levels of endemicity (Map 3-03). The map indicates the seroprevalence of antibody to HAV (total anti-HAV) as measured in selected cross-sectional studies among each country’s residents. Levels of endemicity are related to hygienic and sanitary conditions in the area. The estimates show that all high-income regions have very low HAV endemicity levels, all low-income regions have high endemicity levels, and most middle-income regions have a mix of intermediate and low endemicity levels.
HAV infection is common throughout the developing world, where infections most frequently are acquired during early childhood and usually are asymptomatic or mild, resulting in a high proportion of adults in the population that are immune to HAV and where epidemics of hepatitis A are uncommon. In developed countries, HAV infection is less common, but communitywide outbreaks may occur.
Hepatitis A is one of the most common vaccine-preventable infections acquired during travel. In 2007 in the United States, among cases for which information regarding exposures was collected, the most frequently identified risk factor for hepatitis A was international travel (reported by 18% of patients). As in previous years, most travel-related cases (85%) were associated with travel to Mexico, Central America, or South America. As HAV transmission in the United States has decreased, cases among unimmunized travelers to countries in which hepatitis is endemic have accounted for an increased proportion of all cases. The risk of HAV infection for US residents traveling abroad varies with living conditions, length of stay, and the incidence of HAV infection in the area visited. Risk is highest for those who live in or visit rural areas, trek in backcountry areas, or frequently eat or drink in settings of poor sanitation. However, cases of travel-related hepatitis A can also occur in travelers to developing countries with “standard” tourist itineraries, accommodations, and eating behaviors.
Map 3-03. Estimated prevalence of hepatitis A virus1,2
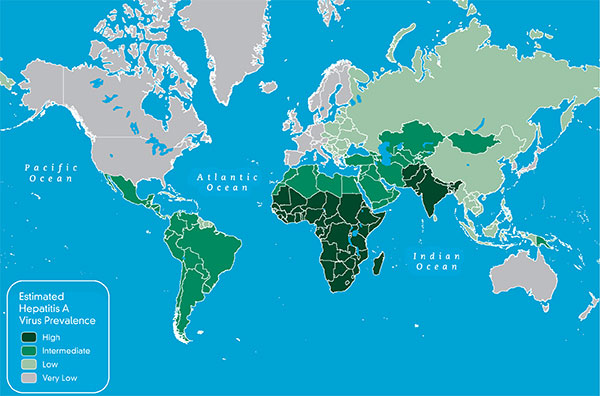
View Larger Map PDF Version (printable)
1Jacobsen KH, Wiersma ST. Hepatitis A virus seroprevalence by age and world region, 1990 and 2005. Vaccine. 2010 Sep 24;28(41):6653–7. Data used with permission from Elsevier.
2Estimates of prevalence of antibody to hepatitis A virus (anti-HAV IgG), a marker of previous HAV infection, are based on systematic literature review conducted for the period of 1990–2005. In addition, anti-HAV prevalence might vary within countries by subpopulation and locality. As used on this map, the terms “high,” “medium,” “low” and “very low” endemicity reflect available evidence of how widespread HAV infection is within each country, rather than precise quantitative assessments.
CLINICAL PRESENTATION
The incubation period for hepatitis A averages 28 days (range, 15–50 days). HAV infection may be asymptomatic, or its clinical manifestations may range in severity from a mild illness lasting 1–2 weeks to a severely disabling disease lasting several months. Clinical manifestations of hepatitis A often include the abrupt onset of fever, malaise, anorexia, nausea, and abdominal discomfort, followed within a few days by jaundice. The likelihood of having symptoms with HAV infection is related to the age of the infected person. In children aged <6 years, most (70%) infections are asymptomatic; if illness does occur, its duration is usually <2 months. Ten percent of infected people have prolonged or relapsing symptoms over a 6- to 9-month period. The overall case-fatality ratio is 0.3%; however, the ratio is 1.8% among adults aged >50 years.
DIAGNOSIS
Demonstration of IgM anti-HAV in the serum of acutely or recently ill patients establishes the diagnosis. IgM anti-HAV becomes detectable 5–10 days after exposure. A 4-fold or larger rise in specific antibodies in paired sera, detected by commercially available EIA, also establishes the diagnosis. If laboratory tests are not available, epidemiologic evidence may provide support for the diagnosis in a clinically compatible case. HAV RNA can be detected in blood and stools of most people during the acute phase of infection through nucleic acid amplification methods, but these generally are not used for diagnostic purposes.
TREATMENT
No specific treatment is available for people with hepatitis A; therefore, treatment is supportive.
PREVENTIVE MEASURES FOR TRAVELERS
Vaccine and Immune Globulin
Monovalent Vaccines
Two monovalent hepatitis A vaccines are licensed in the United States for people aged ≥12 months (Tables 3-02 and 3-03). Both vaccines are made of inactivated HAV adsorbed to aluminum hydroxide as an adjuvant. A full vaccination series includes 2 doses, giving the second dose 6–12 months (Havrix) or 6–18 months (Vaqta) after the first. All hepatitis A vaccines should be administered intramuscularly in the deltoid muscle.
Combination Vaccine
A combined hepatitis A and hepatitis B vaccine is licensed for people aged ≥18 years (Table 3-04). Primary immunization consists of 3 doses, given on a 0-, 1-, and 6-month schedule, the same schedule as commonly used for monovalent hepatitis B vaccine. An accelerated schedule (doses at days 0, 7, and 21–30) for travelers has been approved by the Food and Drug Administration; however, a booster dose should be given at 12 months to promote long-term immunity. The combination vaccine contains aluminum phosphate and aluminum hydroxide as adjuvants. The immunogenicity of the combination vaccine is equivalent to that of the monovalent hepatitis vaccines when tested after completion of the licensed schedule.
Indications for Use
All susceptible people traveling for any purpose, frequency, or duration to countries with high or intermediate hepatitis A endemicity should be vaccinated or receive immunoglobulin (IG) before departure. Providers also may consider its administration to travelers to any destination. The first dose of hepatitis A vaccine should be administered as soon as travel to countries with high or intermediate endemicity is considered.
People who are traveling for international adoption should be advised that hepatitis A vaccination is recommended for all previously unvaccinated household members and other people who anticipate close personal contact (such as regular babysitters) with an international adoptee from a country of high or intermediate endemicity during the first 60 days after arrival of the adoptee in the United States. The first dose of hepatitis A vaccine should be administered as soon as adoption is planned, ideally ≥2 weeks before the arrival of the adoptee (see Chapter 7, International Adoption).
Vaccine Administration
One dose of monovalent hepatitis A vaccine administered at any time before departure can provide adequate protection for most healthy people aged ≤40 years. Hepatitis A vaccine at the age-appropriate dose is preferred to IG. The vaccine series should be completed according to the licensed schedule for long-term protection. Many people will have detectable anti-HAV in response to the monovalent vaccine by 2 weeks after the first vaccine dose. The proportion of people who develop a detectable antibody response at 2 weeks may be lower when smaller vaccine doses are used, such as with the combination vaccine.
For optimal protection, adults aged >40 years, immunocompromised people, and people with chronic liver disease or other chronic medical conditions planning to depart to an area in <2 weeks should receive the initial dose of vaccine along with IG (0.02 mL/kg) at a separate anatomic injection site. Travelers who receive hepatitis A vaccine <2 weeks before traveling to an endemic area and who do not receive IG (either by choice or because of lack of availability) will be at lower risk for infection than those who do not receive hepatitis A vaccine or IG.
Although vaccination of an immune traveler is not contraindicated and does not increase the risk for adverse effects, screening for total anti-HAV before travel can be useful in some circumstances to determine susceptibility and eliminate unnecessary vaccination or IG prophylaxis. Such serologic screening for susceptibility might be indicated for adult travelers who are aged >40 years and those born in areas of the world with intermediate or high endemicity who are likely to have had prior HAV infection, if the cost of screening (laboratory and office visit) is less than the cost of vaccination or IG, and if testing will not delay vaccination and interfere with timely receipt of vaccine or IG before travel. Postvaccination testing for serologic response is not indicated.
Travelers who are aged <12 months, are allergic to a vaccine component, or who otherwise elect not to receive vaccine should receive a single dose of IG (0.02 mL/kg), which provides effective protection against HAV infection for up to 3 months (Table 3-05). Those who do not receive vaccination and plan to travel for >3 months should receive an IG dose of 0.06 mL/kg, which must be repeated if the duration of travel is >5 months. In addition, clinicians should be alert to opportunities to vaccinate all travelers whose plans might include future travel to an area of high or intermediate endemicity, including those whose current medical evaluation is for travel to an area where hepatitis A vaccination is not currently recommended.
Other Vaccine Considerations
Using the vaccines according to the licensed schedules is preferable. However, an interrupted series does not need to be restarted. Given their similar immunogenicity, a series that has been started with one brand of monovalent vaccine may be completed with the other brand. In adults and children who have completed the vaccine series, anti-HAV persists for ≥5–12 years after vaccination. Results of mathematical models indicate that, after completion of the vaccination series, anti-HAV will likely persist for ≥20 years. For children and adults who complete the primary series, booster doses of vaccine are not recommended.
Vaccine Safety and Adverse Reactions
Among adults, the most frequently reported side effects occurring 3–5 days after a vaccine dose are tenderness or pain at the injection site (53%–56%) or headache (14%–16%). Among children, the most common side effects reported are pain or tenderness at the injection site (15%–19%), feeding problems (8% in one study), or headache (4% in one study). No serious adverse events in children or adults have been found that could be definitively attributed to the vaccine, nor have increases in serious adverse events among vaccinated people compared with baseline rates been identified.
IG for intramuscular administration prepared in the United States has few side effects (primarily soreness at the injection site) and has never been shown to transmit infectious agents (hepatitis B virus, hepatitis C virus, or HIV). Since December 1994, all IG products commercially available in the United States have had to undergo a viral inactivation procedure or test negative for hepatitis C virus RNA before release.
Precautions and Contraindications
These vaccines should not be administered to travelers with a history of hypersensitivity to any vaccine component. Twinrix should not be administered to people with a history of hypersensitivity to yeast. Because hepatitis A vaccine consists of inactivated virus, and hepatitis B vaccine consists of a recombinant protein, no special precautions are needed for vaccination of immunocompromised travelers.
Pregnancy
The safety of hepatitis A vaccine for pregnant women has not been determined. However, because hepatitis A vaccine is produced from inactivated HAV, the theoretical risk to either the pregnant woman or the developing fetus is thought to be low. The risk of vaccination should be weighed against the risk of hepatitis A in female travelers who might be at high risk for exposure to HAV. Pregnancy is not a contraindication for using IG.
Personal Protection Measures
Boiling or cooking food and beverage items for ≥1 minute to 185°F (85°C) inactivates HAV. Foods and beverages heated to this temperature and for this length of time cannot serve as vehicles for HAV infection, unless they become contaminated after heating. Adequate chlorination of water, as recommended in the United States, will inactivate HAV. Travelers should follow these recommendations, as well as those described in Chapter 2, Food and Water Precautions.
Table 3-02. Licensed doses and schedules for Havrix1
| AGE GROUP (y) | DOSE (ELU)2 | VOLUME | NUMBER OF DOSES | SCHEDULE (MONTHS) |
|---|---|---|---|---|
| 1–18 | 720 | 0.5 mL | 2 | 0, 6–12 |
| ≥19 | 1,440 | 1.0 mL | 2 | 0, 6–12 |
1Hepatitis A vaccine, inactivated, GlaxoSmithKline.
2ELU, ELISA units of inactivated hepatitis A virus.
Table 3-03. Licensed doses and schedules for Vaqta1
| AGE GROUP (y) | DOSE (U)2 |
VOLUME | NUMBER OF DOSES | SCHEDULE (MONTHS) |
|---|---|---|---|---|
| 1–18 | 25 | 0.5 mL | 2 | 0, 6–18 |
| ≥19 | 50 | 1.0 mL | 2 | 0, 6–18 |
1Hepatitis A vaccine, inactivated, Merck & Co., Inc.
2U, units of hepatitis A virus antigen.
Table 3-04. Licensed doses and schedules for Twinrix1
| AGE GROUP (y) | VOLUME2 | NUMBER OF DOSES | SCHEDULE |
|---|---|---|---|
| ≥18 (primary immunization schedule) | 1.0 mL | 3 | 0, 1, 6 months |
| ≥18 (accelerated schedule) | 1.0 mL | 4 |
0, 7, 21–30 days + 12 months |
1Combined hepatitis A and hepatitis B vaccine, GlaxoSmithKline.
2Each 1.0mL dose contains 720 ELISA units of inactivated hepatitis A virus and 20 micrograms of hepatitis B surface antigen
Table 3-05. Recommended doses of immune globulin (IG) to protect against hepatitis A
| SETTING | DURATION OF COVERAGE | DOSE (mL/kg)1 |
|---|---|---|
| Preexposure | Short-term (1–2 months) Long-term (3–5 months) |
0.02 0.062 |
| Postexposure | Not applicable | 0.02 |
1IG should be administered by intramuscular injection into either the deltoid or gluteal muscle. For children <12 months of age, IG can be administered in the anterolateral thigh muscle.
2Repeat every 5 months if continued exposure to hepatitis A virus occurs.
BIBLIOGRAPHY
- Bacaner N, Stauffer B, Boulware DR, Walker PF, Keystone JS. Travel medicine considerations for North American immigrants visiting friends and relatives. JAMA. 2004 Jun 16;291(23):2856–64.
- Bell BP, Feinstone SM. Hepatitis A vaccine. In: Plotkin SA, Orenstein WA, editors. Vaccines. 4th ed. Philadelphia: WB Saunders; 2004. p. 269–97.
- CDC. Update: Prevention of hepatitis A after exposure to hepatitis A virus and in international travelers. Updated recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep. 2007 Oct 19;56(41):1080–4.
- CDC. Updated recommendations from the Advisory Committee on Immunization Practices (ACIP) for use of hepatitis A vaccine in close contacts of newly arriving international adoptees. MMWR Morb Mortal Wkly Rep. 2009 Sep 18;58(36):1006–7.
- Daniels D, Grytdal S, Wasley A. Surveillance for acute viral hepatitis—United States, 2007. MMWR Surveill Summ. 2009 May 22;58(3):1–27.
- Fiore AE. Hepatitis A transmitted by food. Clin Infect Dis. 2004 Mar 1;38(5):705–15.
- Fiore AE, Wasley A, Bell BP. Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2006 May 19;55(RR-7):1–23.
- Jacobsen KH, Wiersma ST. Hepatitis A virus seroprevalence by age and world region, 1990 and 2005. Vaccine. 2010 Sep 24;28(41):6653–7.
- Mutsch M, Spicher VM, Gut C, Steffen R. Hepatitis A virus infections in travelers, 1988–2004. Clin Infect Dis. 2006 Feb 15;42(4):490–7.
- Van Damme P, Banatvala J, Fay O, Iwarson S, McMahon B, Van Herck K, et al. Hepatitis A booster vaccination: is there a need? Lancet. 2003 Sep 27;362(9389):1065–71.
- Winokur PL, Stapleton JT. Immunoglobulin prophylaxis for hepatitis A. Clin Infect Dis. 1992 Feb;14(2):580–6.
Contact Us:
- Centers for Disease Control and Prevention
1600 Clifton Rd
Atlanta, GA 30333 - 800-CDC-INFO
(800-232-4636)
TTY: (888) 232-6348 - New Hours of Operation
8am-8pm ET/Monday-Friday
Closed Holidays - Contact CDC-INFO


