|
ARCOS >
Registrant
Handbook
> Section 5
SECTION 5.0 - TRANSACTION
RECORD
5.1 TRANSACTION RECORD
Controlled substance transactions are reported using
the ARCOS transaction record.
This section discusses the transaction record
fields that apply to all manufacturers and distributors of reportable
controlled substances. Section 6 discusses transaction
record fields that apply only
to manufacturers.
The ARCOS transaction
record has two formats: one format for
reporting controlled substance transactions on magnetic tape, cartridge,
or diskette (automated reporting) and another, slightly different format,
for reporting transactions on DEA Form 333 (manual reporting). Exhibit
5.1: Transaction Record Formats, illustrates the record layouts for both
formats. The differences between the two formats are:
-
The automated reporting format contains an additional field, the document
Identifier field, which identifies the transaction as having
been generated by an automated system.
-
The transaction date field is a six-digit
field in the automated reporting format and a five-digit
field in the manual reporting format.
-
The quantity
field is an eight-position field in the automated
reporting format and a six-position field in the manual
reporting format
|
|
AUTOMATED |
MANUAL (DEA Form 333) |
|
FIELD NAME |
FIELD # |
FIELD LENGTH |
POSITION LOCATION |
FIELD # |
FIELD LENGTH |
COLUMN LOCATION |
|
REPORTING REGISTRANT
NUMBER
|
1 |
9 |
1-9 |
1 |
9 |
1-9 |
|
TRANSACTION CODE |
2 |
1 |
10 |
2 |
1 |
10 |
|
ACTION INDICATOR
(FORMERLY DELETE INDICATOR)
|
3 |
1 |
11 |
3 |
1 |
11 |
|
NATIONAL DRUG CODE (NDC NUMBER) |
4 |
11 |
12-22 |
4 |
11 |
12-22 |
|
QUANTITY |
5 |
8 |
23-30 |
5 |
6 |
23-28 |
|
UNIT |
6 |
1 |
31 |
6 |
1 |
29 |
|
ASSOCIATE REGISTRANT NUMBER |
7 |
9 |
32-40 |
7 |
9 |
30-38 |
|
DEA ORDER FORM NUMBER |
8 |
9 |
41-49 |
8 |
9 |
39-47 |
|
TRANSACTION DATE |
9 |
6 |
50-55 |
11-13 |
5 |
60-64 |
|
CORRECTION NUMBER (FORMERLY LOT NUMBER) |
10 |
8 |
56-63 |
9 |
8 |
48-55 |
|
STRENGTH |
11 |
4 |
64-67 |
10 |
4 |
56-59 |
|
TRANSACTION IDENTIFIER |
12 |
10 |
68-77 |
14 |
5 |
65-69 |
|
DOCUMENT IDENTIFIER |
13 |
3 |
78-80 |
NONE |
NONE |
NONE |
Exhibit 5.1: Transaction Record Formats
5.2 DEA FORM 333: DUPLICATING DATA
ARCOS uses the equal sign,
"=", to indicate that the data recorded in a transaction
record field on DEA Form 333 is to be duplicated in subsequent transaction
records. Under ARCOS the "=" is called the "duplicate
sign." The duplicate sign (=) may be used to avoid repeatedly writing
identical data within a field. The duplicate sign (=) must
be coded in the first position of each field being duplicated. Examples
applying to sections 5.2 and 5.2.1 illustrating the use of the duplicate
sign are found in:
-
Exhibit
5.2: Using the Duplicate Sign, Single Reporter and
-
Exhibit
5.3: Using the Duplicate Sign, Central Reporter
5.2.1 DEA Form 333: Duplicating
Transaction Date
The transaction date field is
composed of three segments: Fields 11, 12, and 13 (year, month, and day).
These three segments must be considered one, single field
for duplicating purposes. Therefore, the entire transaction date must
be the same when using the duplicate sign.

Exhibit 5.2: Using Duplicate Sign:
Single Reporter

Exhibit 5.3: Using the Duplicate
Sign, Central Reporter
5.3 REPORTING REGISTRANT NUMBER
5.3.1 Definition: Reporting Registrant
Number
The reporting registrant number
is the DEA registration number identifying the location where the
controlled substance activities being reported have occurred. This is a
9-character field.
5.3.2 Specifications: Reporting
Registrant Number
| a. Field Number: |
1 |
| b. Field Name: |
reporting registrant number |
| c. Field Length: |
9 Characters |
| d. Positions/Columns: |
1-9 |
| e. Type: |
Alphanumeric |
| f. Special Rules: |
|
 1.
Mandatory Entry 1.
Mandatory Entry |
 2.
Use Capital Letters for Alphabetic Data 2.
Use Capital Letters for Alphabetic Data |
 3.
Position/Column 1 is always 'P' or ‘R’ 3.
Position/Column 1 is always 'P' or ‘R’ |
5.3.3 Discussion: Reporting Registrant
Number
A central reporter uses its own
registrant reporting number when reporting for itself and the
subsidiaries’s registrant reporting number when reporting for a
subsidiary.
5.4 TRANSACTION CODE
5.4.1 Definition: Transaction Code
The transaction code is a
single-character field which identifies each specific ARCOS-reportable
activity.
5.4.2 Specifications: Transaction
Code
| a. Field Number: |
2 |
| b. Field Name: |
transaction code |
| c. Field Length: |
1 Character |
| d. Position/Column: |
10 |
| e. Type: |
Alphanumeric |
| f. Special Rules: |
|
 1.
Only Specific Codes Permitted 1.
Only Specific Codes Permitted |
 2.
Mandatory Entry 2.
Mandatory Entry |
 3.
Use Capital Letters for Alphabetic Data 3.
Use Capital Letters for Alphabetic Data |
5.4.2 Specifications: Transaction Code
| a. Field Number: |
2 |
| b. Field Name: |
transaction code |
| c. Field Length: |
1 Character |
| d. Position/Column: |
10 |
| e. Type: |
Alphanumeric |
| f. Special Rules: |
|
 1.
Only Specific Codes Permitted 1.
Only Specific Codes Permitted |
 2.
Mandatory Entry 2.
Mandatory Entry |
 3.
Use Capital Letters for Alphabetic Data 3.
Use Capital Letters for Alphabetic Data |
5.4.3 Discussion: Transaction Code
Manufacturers and distributors are
responsible for reporting all ARCOS activities and inventories. The transaction
code field identifies the reporting registrant’s controlled substance
acquisitions, dispositions, inventories, and miscellaneous transactions. The
transaction codes listed below have been grouped into four categories for
ease in selecting the correct code under which to report a specific business
or manufacturing activity. These four categories are: inventory,
acquisition, disposition, and miscellaneous. They are discussed in detail in
Section 5.5 through 5.7. See Appendix 1, ARCOS
Transaction Matrix: Automated Reports or Appendix
2, ARCOS Transaction Matrix: Manual Reports, to determine which fields
must be completed for a specific transaction code.
Inventory Transaction Codes
transaction code 1:
Schedule Change Inventory
transaction code 3: Year-End Inventory
transaction code 4: Year-End In-Process
Inventory (manufacturers only)
transaction code 5: Special Inventory
transaction code 8: No Year-End Inventory
Acquisition Transaction Codes (Increases to Inventory)
transaction code P:
Purchase or Receipt
transaction code R: Return
transaction code V: Unsolicited Return
transaction code G: Government Supplied
transaction code W: Recovered Waste
(manufacturers only)
transaction code M: Manufactured
(manufacturers only)
transaction code L: Reversing (manufacturers
only)
transaction code J: Return of Sample to
Inventory (manufacturers only)
Disposition Transaction Codes (Decreases
to Inventory)
transaction code S:
Sale, Disposition, or Transfer
transaction code Y: Destroyed
transaction code T: Theft
transaction code Z: Receipt by Government
(seizures, samples, etc.)
transaction code N: Nonrecoverable Waste
(manufacturers only)
transaction code U: Used in Production
(manufacturers only)
transaction code Q: Sampling (manufacturers
only)
transaction code K: Used in Preparations
(manufacturers only)
Miscellaneous Transaction Codes
transaction code 7:
No ARCOS Activity for the Current Reporting Period
transaction code F: Reorder DEA Form 333
transaction code X:
Lost-in-Transit
5.5 INVENTORY TRANSACTION CODES
Transaction code
1, Schedule Change Inventory; transaction code 3, Year-End Physical
Inventory; transaction code 4, In-Process Inventory; and transaction
code 5, Special Inventory are used when reporting inventory data to DEA
(ARCOS).
5.5.1 Code 1: Schedule Change Inventory
Transaction code 1,
Schedule Change Inventory, is used when reporting bulk or dosage form
products that become reportable controlled substances due to a controlled
substance schedule change. Specific instructions will be provided by DEA
(ARCOS) when a controlled substance schedule change affecting ARCOS
reporting occurs.
Example:
Prior to March 21, 1991, glutethimide was
a Schedule III controlled substance which was only reportable by ARCOS
registrants actually manufacturing the drug. On March 21, 1991, glutethimide
became a Schedule II controlled substance. At this point, glutethimide
became reportable by all ARCOS registrants. All ARCOS registrants not
previously reporting this controlled substance were required to report a
Schedule Change Inventory Transaction (transaction code 1), if
glutethimide was in their inventory on March 21, 1991.
Acquisition After Report
ARCOS registrants acquiring new stock
after a Schedule Change Inventory report has been submitted, report this new
stock as a "purchase" (transaction code P) or as another
type of acquisition transaction when reporting that period’s activities.
Out-of-Stock or Do Not Carry
ARCOS registrants that carry a
newly-reportable controlled substance, but are out-of-stock or
ARCOS registrants that do not carry a newly-reportable controlled substance must
not report a Schedule Change Inventory. If an ARCOS registrant
acquires the newly-reportable controlled substance at a later date, this
acquisition would be reported as a "purchase" (transaction code
P) or as another type of acquisition transaction when reporting that
period’s activities.
Exhibit 5.4: Schedule Change Reporting
Requirements, summarizes reporting requirements when a controlled substance
schedule change makes a formerly non-reportable controlled substance
reportable.
|
Inventory Status |
ARCOS Transaction Code |
Submission Required |
|
Out-of-stock |
None |
Schedule Change Inventory Not
Required |
|
In-stock |
transaction code 1 |
Schedule Change Inventory
Required
|
|
Received After Schedule Change Date |
transaction code P or
Other Applicable Acquisition Code |
Schedule Change Inventory Not
Required
|
Exhibit 5.4: Schedule Change Reporting Requirements
5.5.2 Code 3: Year-End Physical Inventory
All ARCOS registrants are required to
take a physical, year-end inventory (YEI). This inventory must
reflect the actual amount of each reportable
controlled substance on the registrant’s premises as of the close of
business on December 31st of each year. This is the only
valid date which is accepted for transaction code 3. If the
year-end inventory report is not dated December 31, the transaction
record is rejected. If year-end inventory transactions are
included in the December or fourth quarter report, the reporting registrant must
write "YEI INCLUDED" in the "Notes"
block of the bar code label. This indicates that the December or fourth
quarter report also contains the year-end inventory report and will prevent
the registrant from being placed in a delinquent reporting status.
5.5.3 Code 4: Year-end In-process
Inventory
On December 31st of each year, a
manufacturer may have some quantities of controlled substances that are
still in the manufacturing production chain (i.e., not in a bulk or finished
dosage form). The amount of controlled substance at this stage of the
production chain is termed an "In-process Inventory" quantity.
Year-end In-process Inventory is reported using transaction code 4.
The Year-end In-process Inventory is to be taken at the close of business on
December 31st of the reporting year. This is the only date
which will be accepted for transaction code 4. See
Section 6, Manufacturing Activities, for examples of In-process
Inventory reporting.
5.5.4 Code 5: Special Inventory
A Special Inventory is any inventory
other than a Year-end, Year-end In-process, or Schedule Change Inventory
being taken at the direction of DEA. Report a Special
Inventory using transaction code 5.
5.5.5 Code 8: No Year-End Inventory
Transaction code 8
is used to report zero (0) year-end inventory. If, at the close of business
on December 31st, an ARCOS registrant has no (i.e., zero) physical inventory
for all the reportable controlled substances that it handles,
the firm must report this lack of physical
inventory to DEA (ARCOS). It is not necessary to report the
lack of inventory for each reportable controlled substance that the firm has
handled through the year. A single transaction code 8 is sufficient. The
"no-year-end inventory" report will prevent DEA
(ARCOS) from placing the registrant in a delinquent reporting status. transaction
code 8 requires the following fields:
(December 31st of each year)
transaction Identifier
5.6 ACQUISITION TRANSACTION CODES
An ARCOS registrant can acquire
controlled substances by buying them, by having previously sold controlled
substances returned, or by receiving them from the government. If the ARCOS
registrant is a manufacturer, the registrant can acquire controlled
substances by four additional means: (1) manufacturing them, (2) recovering
them from manufacturing waste, (3) having samples of controlled substances
returned, or (4) by decomposing a substance into its component substances;
one of which is a controlled substance. Acquisitions of controlled
substances, irrespective of the manner in which they are acquired, increase
the ARCOS registrant’s inventory.
5.6.1 Code P: Purchase or Other Receipt
Transaction code P,
purchase or other receipt, is used to report controlled substance
acquisitions under three different scenarios:
-
The acquisition of a controlled substance by one ARCOS
registrant from another.
-
The transfer of a controlled substance from one physical
location to another.
-
Establishing the initial stock on hand of a controlled
substance for a new ARCOS registrant
Controlled substances that are shipped directly
from a supplier to an ARCOS registrant’s customer (third party shipments)
and are never physically on the registrant’s premises,
(i.e., a drop shipment) are reported to DEA (ARCOS) by the supplier and the
recipient. The ARCOS registrant must not report these
transactions.
5.6.2 Code R: Return
Transaction code R
is used to report the receipt of returned controlled substances when the
manufacturer or distributor requests their return. Returns
reported under transaction code R do not necessarily
involve a monetary transaction. Reportable controlled substances received by
a supplier may include substances being returned for credit, salvage,
re-work, or non-GMP (good manufacturing process) quality, as well as
outdated or unused controlled substances.
5.6.3 Code V: Unsolicited Return
Transaction code V
is used to report the receipt of an unsolicited return of a reportable
controlled substance. An "unsolicited return" is a return that has
not been requested by the manufacturer or
distributor. The situations needing transaction code V are described
below and summarized in Exhibit 5.5: Using Transaction Code V.
|
UNSOLICITED RETURN |
|
|
SHIPPER’S IDENTITY
|
SCHEDULE
|
ORDER FORM # REQUIRED?
|
TRANSACTION CODE
|
ASSOCIATE REGISTRANT NUMBER ENTRY
|
|
1 |
Unknown |
III narcotic |
NO |
V |
"UNKNOWN" |
|
2 |
Unknown |
I, II |
YES |
V |
"UNKNOWN" |
|
3 |
Known |
III narcotic |
NO |
V |
USE SHIPPER’S DEA REGISTRATION
NUMBER |
|
4 |
Known |
I, II |
YES |
V |
USE SHIPPER’S DEA REGISTRATION
NUMBER |
Exhibit 5.5: Using Transaction Code V
Unknown Shipper
-
Use transaction code V when a
Schedule III narcotic shipment is received without any
identifying markings indicating the name of the firm that shipped the
drugs. In the associate registrant number field (Field 7), enter
"UNKNOWN" in all capital letters and left
justified.
-
Use transaction code V when a
Schedule I or II product is received without the prior
issuance of an order form (U.S. Official Order Forms - Schedules I &
II) DEA Form 222, and without any
identifying markings indicating the name of the firm that shipped the
product. In the associate registrant number field (Field 7),
enter "UNKNOWN" in all capital letters
and left justified. Additionally, a DEA Order Form Number must
be acquired to cover the shipment. An "after-the-fact" DEA
Order Form Number must be issued by the receiving firm
using its own blank Order Form, after
obtaining approval from the local DEA field office.
Known Shipper
-
Use transaction code V when a
Schedule III narcotic shipment is received without prior
notification and the shipper is known. If the registrant decides
to keep the shipment, the registrant must obtain the approval of the
local DEA field office. The shipper’s DEA Registration
Number is entered into the associate registrant number field
(Field 7).
-
Use transaction code V when a
Schedule I or II shipment is received without the prior
issuance of an order form, DEA Form 222, and the shipper is known.
When such a shipment is received, the recipient firm must:
(1) obtain the approval of the local DEA field office to issue an
"after-the-fact" DEA order form number and (2) issue the DEA
order form number using its own blank order form. The shipper’s
DEA registration number is entered into the associate registrant
number field (Field 7) and the order form number is entered in the DEA
order form number field (Field 8).
5.6.4 Code G: Government Supplied
Transaction code G
is used when the government supplies controlled substances or returns seized
materials or samples to the reporting registrant. The registration number of
the DEA or FDA divisional office is required in the associate registrant
number field (Field 7). Contact your local DEA office to obtain the
appropriate associate registrant number, if it is unknown.
5.6.5 Manufacturing Acquisition Codes
The following acquisition transaction
codes are discussed in Section 6, Manufacturing Activities:
transaction code W Recovered
Waste (Manufacturers Only)
transaction code M Manufactured (Manufacturers
Only)
transaction code L Reversing (Manufacturers
Only)
transaction code J Return of Sample to
Inventory (Manufacturers Only)
5.7 DISPOSITION TRANSACTION CODES
Controlled substances can be sold,
destroyed, taken by the government, or stolen (theft). Under the
manufacturing process there are a number of additional ways to dispose of
controlled substances: (1) non-recoverable waste, (2) used in manufacturing,
(3) distribution of samples, and (4) used in preparations. Dispositions of
controlled substances, irrespective of the manner in which they are
disposed, decrease the ARCOS registrant’s inventory.
5.7.1 Code S: Sale, Disposition, or
Transfer
Transaction code S is
used when a controlled substance is physically transferred to another DEA
registrant. This is not necessarily a monetary transaction.
Samples to customers are included in this category. However, a sample that
does not leave the ARCOS registrant’s premises is reported
as a transaction code Q. See Section 6, Manufacturing Codes. The sale
of reportable controlled substances reduces the manufacturer’s or
distributor’s inventory.
In cases where a controlled substance is
shipped directly from a supplier to an ARCOS registrant’s customer and is never
physically on the registrant’s premises (e.g., the registrant only
does the billing), the registrant must not
report the transaction. The supplier and the recipient do the reporting.
5.7.2 Code Y: Destroyed
Transaction code Y is
used for reporting authorized destructions of controlled substances. Enter
the registration number of the local DEA area office in the associate
registrant number field (Field 7). If
necessary, contact the DEA area office or the Data Systems Unit (ARCOS) to
obtain the DEA registration number. For all destructions,
DEA Form 41, Registrants Inventory of Drugs
Surrendered, must be filed with the local DEA area office.
Exhibit 5.6: DEA Form 41, contains a sample form.
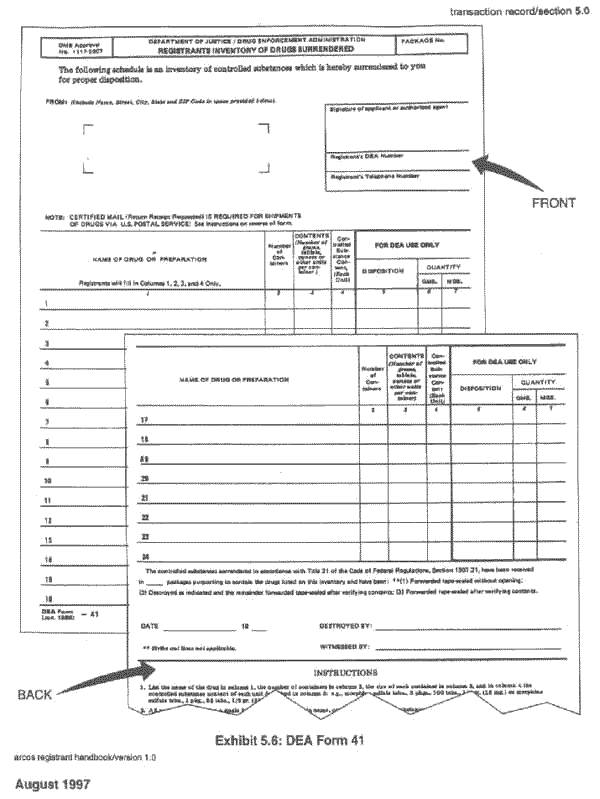
Exhibit 5.6:
DEA Form 41
5.7.3 Code T: Theft
Transaction code T is
used to report controlled substances stolen from your premises.
This does not eliminate the requirement to prepare an official theft
report, U.S. Department of Justice, Drug Enforcement Administration,
Report of Theft or Loss of Controlled Substances, DEA Form 106. Contact
your local DEA office for further details. See Section 5.8.3 Code
X, for in-transit theft or loss. Exhibit 5.7: DEA Form
106, contains a
sample theft report form.

Exhibit 5.7: DEA Form 106
5.7.4 Code Z: Receipt by Government or
Seizures
Transaction code Z
is used to report the transfer (e.g., samples or seizure) of reportable
controlled substances from a manufacturer or distributor to a government
official. When a controlled substance is received by an agent of DEA or FDA,
the DEA registration number of their Area Office must be entered in the associate
registrant number field (Field 7). "OFFICER"
is the entry in Field 7 for any other non-registered government official such
as a customs officer; an agent of the Bureau of Alcohol, Tobacco, and
Firearms; or state and local police officers. This entry must be
in all capital letters and
left justified within Field 7.
5.7.5 Manufacturing Disposition Codes
The following disposition transaction codes
are discussed in Section 6, Manufacturing Activities:
transaction code N Nonrecoverable
Waste (Manufacturers Only)
transaction code U Used in Production (Manufacturers Only)
transaction code Q Sampling (Manufacturers Only)
transaction code K Used In Preparations (Manufacturers Only)
5.8 MISCELLANEOUS TRANSACTION CODES
5.8.1 Code 7: No ARCOS Activity for the
Current Reporting Period
Transaction code 7
is used to report the lack of controlled substance activity for the current
reporting period. This lack of activity must be reported
to DEA (ARCOS). transaction code 7 is used when there has been no
business activity for any and all controlled substances during the
reporting period. It is not necessary to report no (i.e., zero) activity for
each NDC product; a single transaction code 7 will suffice. Submitting
a transaction code 7 will prevent DEA from placing the ARCOS Registrant
in a delinquent reporting status. Transaction code 7 requires the
following fields:
5.8.2 Code F: Reorder DEA Form 333
Transaction code F
is used to re-order a supply of DEA Form 333. The quantity requested cannot
exceed 500 sheets for any single re-order. When the quantity field
(Field 5) is left blank 100 forms will be sent. Complete only the following
fields:
| Field 1: |
reporting
registrant number |
| Field 2: |
transaction
code F |
| Field 5: |
quantity |
| Field 14: |
transaction
identifier |
5.8.3 Code X: Lost-in-Transit
Transaction code X
is used by the seller to report the loss or theft of an in-transit shipment of
a reportable controlled substance. It is reported in addition to
the normal sales transaction (transaction code S). Transaction code
X is an explanatory transaction code which does not
affect an ARCOS registrant’s inventory. Enter the DEA registration number of
the intended purchaser in the associate registrant number field (Field
7). If the product lost in transit was a Schedule I or II controlled substance
the selling ARCOS registrant must file an official theft report,
U.S. Department of Justice, Drug Enforcement Administration, Report of Theft
or Loss of Controlled Substances, DEA Form 106, with the local DEA office.
Exhibit 5.7: DEA Form 106, contains a sample theft report form. If the ARCOS
registrant purchasing the product still wants it, this registrant must
supply a new order form with a new DEA order form
number to replace the original one.
5.9 ACTION INDICATOR (Formerly DELETE
INDICATOR)
5.9.1 Definition: Action Indicator
The action indicator was formerly
called the delete indicator. The name has been changed to reflect the
fact that the function of this field has been expanded.
The action indicator is a
single-character field which initiates three different ARCOS data base
operations: (1) the deletion of a transaction record, (2) the revision
(adjustment) of data in a transaction record, and (3) the
insertion of a late transaction record. These three data base
operations are components of ARCOS error processing. Section
7.5, Correcting Transaction Records, contains a full discussion of the
error processing.
5.9.2 Specifications: Action Indicator
| a. Field Number: |
3 |
| b. Field Name: |
action indicator (Formerly delete
indicator) |
| c. Field Length: |
1 Character |
| d. Position/Column: |
11 |
| e. Type: |
Alphabetic |
| f. Special Rules: |
|
 1.
Code "D" to delete a transaction
record 1.
Code "D" to delete a transaction
record |
 2.
Code "A" to adjust (revise) data in a transaction
record 2.
Code "A" to adjust (revise) data in a transaction
record |
 3.
Code "I" to insert (add) a late transaction
record 3.
Code "I" to insert (add) a late transaction
record |
 4.
Use only capital letters 4.
Use only capital letters |
 5.
Leave blank when unused 5.
Leave blank when unused |
5.9.3 Discussion: Action Indicator
See Section
7.5, Correcting Transaction Records for additional action indicator
instructions.
5.10 NATIONAL DRUG CODE (NDC)
5.10.1 Definition: NDC
The National Drug Code (NDC) used by ARCOS
is an 11-character code that identifies controlled substance products. This
code is divided into three segments: the labeler code, the product code, and
the package size code. General specifications are presented in Section 5.10.2,
Specifications: NDC, followed by detailed specifications for each segment.
5.10.2 Specifications: NDC
| a. Field Number: |
4 |
| b. Field Name: |
NDC Number |
| c. Field Length: |
11 Characters |
| d. Position/Column: |
12-22 |
| e. Type: |
Alphanumeric |
| f. Special Rules: |
|
 1.
Always coded except for transaction codes 7, 8, and F. 1.
Always coded except for transaction codes 7, 8, and F. |
 2.
Entries must be made in each segment (Labeler Code, Product Code, 2.
Entries must be made in each segment (Labeler Code, Product Code,  Package Size Code).
Package Size Code). |
 3.
Do not use hyphens (-), slashes (/), or blanks. 3.
Do not use hyphens (-), slashes (/), or blanks. |
 4.
Do not enter product’s name in lieu of the NDC. 4.
Do not enter product’s name in lieu of the NDC. |
 5.
Use capital letters for alphabetic data 5.
Use capital letters for alphabetic data |
 6.
See specific segment for additional special rules. 6.
See specific segment for additional special rules. |
5.10.2.1 NDC Segment Specifications
Labeler Code
The National Drug Code Directory1
defines a labeler as "...any firm that manufactures or distributes a drug
product." The labeler code is assigned by the FDA.
| a. Segment Name: |
Labeler Code |
| b. Field Length: |
5 Characters |
| c. Positions/Columns: |
12-16 |
| d.
Type: |
Alphanumeric |
| e.
Special Rules: |
|
 1.
Right justified. 1.
Right justified. |
 2.
Leading zeros must be entered to fill blank segment
positions or 2.
Leading zeros must be entered to fill blank segment
positions or
 columns. columns. |
| |
|
|
|
|
|
Product Code
The National Drug Code Directory defines
the product code as the segment that "identifies a specific strength,
dosage form, and formulation for a particular labeler." The product code
is assigned by the labeler.
| a. Segment Name: |
Product Code |
| b. Field Length: |
4 Characters |
| c. Positions/Columns: |
17-20 |
| d.
Type: |
Alphanumeric |
| e.
Special Rules: |
|
 1.
Right justified. 1.
Right justified. |
 2.
Leading zeros must be entered to fill blank segment positions
or 2.
Leading zeros must be entered to fill blank segment positions
or
 columns. columns. |
Package Size Code
The National Drug Code Directory defines
package size code as the segment that "identifies trade package
sizes." The package size code is assigned by the labeler.
| a. Segment
Name: |
Package
Size Code |
| b. Field
Length: |
2 Characters |
| c.
Positions/Columns: |
21-22 |
| d.
Type: |
Alphanumeric
or "**" for bulk finished (e.g., unpackaged bulk dosage
units) or bulk raw material (e.g., powder or liquid) |
| e.
Special Rules: |
|
 1.
Right justified. 1.
Right justified. |
 2.
Leading zeros must be entered to fill blank segment positions
or 2.
Leading zeros must be entered to fill blank segment positions
or
 columns. columns. |
-
U.S Department of
Health and Human Services, Food and Drug Administration, National Drug
Code Directory, Vol I, June 1995.
5.10.2.2 Formatting Summary
Exhibit 5.8: NDC
Formatting Summary, briefly describes the formatting specifications for
the NDC. These specifications apply to controlled substance transactions
reported on automated media as well as DEA Form 333 for the following
products:
-
Bulk Raw Powder
-
Bulk Dosage Formulations
-
Bulk Solutions
-
Trade Packages
| TYPE OF MATERIAL |
Bulk raw powder; bulk dosage forms;
bulk solutions |
| LABELER CODE POSITIONS or COLUMNS:
12-16 |
alphanumeric, right justified, fill
blanks with leading zeros |
| PRODUCT CODE POSITIONS or COLUMNS:
17-20 |
alphanumeric, right justified, fill
blanks with leading zeros |
| PACKAGE CODE POSITIONS or COLUMNS:
21-22 |
** |
| |
| TYPE OF MATERIAL |
Trade Packages |
| LABELER CODE POSITIONS or COLUMNS:
12-16 |
alphanumeric, right justified, fill
blanks with leading zeros |
| PRODUCT CODE POSITIONS or COLUMNS:
17-20 |
alphanumeric, right justified, fill
blanks with leading zeros |
| PACKAGE CODE POSITIONS or COLUMNS:
21-22 |
alphanumeric, right justified, fill
blanks with leading zero |
Exhibit 5.8: NDC Formatting Summary
5.10.3 NDC Coding Examples
The NDC configuration for ARCOS reporting
is composed of a 5-character Labeler Code, a 4-character Product Code, and a
2-character Package Code. When the NDC for a product does not
conform to the configuration required under ARCOS, the following changes must
be made in the configuration:
-
A leading zero must be
added to the Labeler Code when this segment contains 4-characters.
-
A leading zero must be
added to the Product Code when this segment contains 3-characters.
-
A leading zero must be
added to the Package Size Code when this segment contains
1-character.
-
"**" must be
placed in the Package Size Code segment when the product does not
have a Package Size Code. The "**" indicates the product is in
bulk form.
Transactions with incorrectly formatted NDC
fields are rejected as erroneous and must be corrected and resubmitted. The
exhibits in this section illustrate correct coding for the NDC field. Exhibit
5.9: Converting the NDC for a Non-bulk Product, illustrates how to convert an
NDC for a non-bulk product to the format required by ARCOS. Exhibit 5.10:
Converting the NDC for a Bulk Product, illustrates how to convert an NDC for a
bulk product to the ARCOS format.
Fictitious Non-bulk Product (NDC Directory Configurations):
(a) 1234 - 1234 - 12 (b) 12345 - 123 - 34 (c)
12345 - 1234 - 7
Converting To ARCOS NDC Configurations
a. 01234 - 1234 - 12
 ^_________________________leading
zero added ^_________________________leading
zero added
b. 12345 - 0123 - 34
 ^________________leading
zero added ^________________leading
zero added
c. 12345 - 1234 - 07
 ^_______________________leading
zero added ^_______________________leading
zero added
|
|
Exhibit 5.9: Converting the NDC for a
Non-bulk Product
Fictitious Bulk Product (NDC Directory
Configurations):
(a) 1234 - 1234 (b) 12345 - 123 (c) 12345 - 1234
Converting To ARCOS NDC Configurations
|
Exhibit 5.10: Converting the NDC for a
Bulk Product
5.10.4 NDC Assignment
Identification of reportable controlled
substances is always based on the Food and Drug Administration’s National
Drug Code (NDC). All transaction records, except
for transaction codes 7, 8, and F must contain an NDC. Any transaction
record with missing or invalid NDC will be rejected and must be corrected
before being resubmitted. Contact the FDA at the address provided in Exhibit
5.11: FDA Address, for information about the National Drug Code.
Food and Drug Administration
Bureau of Drugs, Drug Listing Staff
5600 Fishers Lane
Center for Drug Evaluation & Research (CDER), HFD-95
Rockville, Maryland 20857
Telephone: (301) 594-1086
Hours: Monday-Friday 8:00am - 4:30pm EST/EDT
Exhibit 5.11: FDA Address
5.10.5 Submitting Labels
Pursuant to 21
CFR 1308.04, firms holding a DEA registration as a manufacturer must
provide DEA (ARCOS) with information about each new product, new dosage form,
or other unit form containing any quantity of controlled
substance. This information must be submitted within 30 days after
manufacturing begins. However, DEA (ARCOS) will also accept this information prior
to the beginning of manufacturing. Two labels or other documents
(e.g., Drug Listing Form, FDA 2657) which reflect the following information must
be submitted:
-
The trade name, brand name, or other
commercial name of the product;
-
The generic or chemical name and
quantity of each active ingredient, including both controlled and
non-controlled substances (indicate what information is a proprietary
trade secret);
-
The National Drug Code assigned to the
product, if any; and
-
The weight of controlled substance as
follows:
(1) Finished Dosage Unit Products:
Grams or milligrams per dosage unit
(2) Bulk Products:
Grams or milligrams per gram of powder
Grams or milligrams per milliliter of liquid
Send this information to DEA (ARCOS). The
address is listed on the contact information page at the front of this
handbook. The Data Systems Unit (ARCOS) strongly advises each manufacturer to
send this information before submitting transaction records
for their new products. A transaction record that does not have a
matching NDC in the ARCOS NDC Dictionary is rejected as erroneous and must be
resubmitted. Call the Data Systems Unit (ARCOS) to find out if the NDC
information for your firm’s new product has been added to the Dictionary.
All transactions for the new product that
have occurred before the current reporting period, must
be submitted as Late Transactions. Otherwise,
these transactions will be rejected as errors because their transaction
dates are not within the current reporting period. See
Section 7, Edit Listings, for Late Transaction instructions.
5.10.6 Pseudo NDC’s
The pseudo NDC is a number developed by DEA
(ARCOS) in consultation with the ARCOS registrant. A pseudo NDC may be
requested from the DATA Systems Unit (ARCOS) when the NDC does not exist or is
unavailable. This number enables ARCOS registrants to report transactions
involving products for which an NDC is either unavailable or does not exist.
Pseudo NDC’s are not listed in the Food and Drug
Administration’s National Drug Code Directory.
5.10.7 Inner and Outer NDC Packages
A controlled substance product may have one
NDC on an outer, larger package and a different NDC on the inner, smaller,
individually-packaged units contained within the larger package. See
Exhibit 5.12: Inner and Outer Packaging for an illustration. Either NDC
may be used when reporting transactions and inventories. Care must be taken
that the NDC corresponds to the product and package size being
reported. A single NDC must never be used to identify two
different package sizes of a product. The quantity reported will indicate the
number of packages for that particular NDC.

Exhibit 5.12: Inner and Outer
Packaging
Previous |
Table of Contents |
Next
|