
| |  | |  |
Children's Health
|
| Molecular Evidence of an Interaction Between Prenatal Environmental Exposures and Birth Outcomes in a Multiethnic Population Frederica P. Perera,1 Virginia Rauh,1 Robin M. Whyatt,1 Wei-Yann Tsai,1 John T. Bernert,2 Yi-Hsuan Tu,1 Howard Andrews,1 Judyth Ramirez,1 Lirong Qu,1 and Deliang Tang1 1Columbia Center for Children's Environmental Health, Mailman School of Public Health, Columbia University, New York, USA; 2Centers for Disease Control and Prevention, National Center for Environmental Health, Division of Laboratory Sciences, Atlanta, Georgia, USA Abstract
Inner-city, minority populations are high-risk groups for adverse birth outcomes and also are more likely to be exposed to environmental contaminants, including environmental tobacco smoke (ETS) , benzo[a]pyrene (BaP) , and other polycyclic aromatic hydrocarbons (PAHs) found in urban air. In a sample of nonsmoking African-American and Dominican women, we evaluated the effects on birth outcomes of prenatal exposure to ETS, using questionnaire data and plasma cotinine as a biomarker of exposure, and environmental PAHs using BaP-DNA adducts as a molecular dosimeter. We previously reported that among African Americans, high prenatal exposure to PAHs estimated by prenatal personal air monitoring was associated with lower birth weight (p = 0.003) and smaller head circumference (p = 0.01) after adjusting for potential confounders. In the present analysis, self-reported ETS was associated with decreased head circumference (p = 0.04) . BaP-DNA adducts were not correlated with ETS or dietary PAHs. There was no main effect of BaP-DNA adducts on birth outcomes. However, there was a significant interaction between the two pollutants such that the combined exposure to high ETS and high adducts had a significant multiplicative effect on birth weight (p = 0.04) and head circumference (p = 0.01) after adjusting for ethnicity, sex of newborns, maternal body mass index, dietary PAHs, and gestational age. This study provides evidence that combined exposure to environmental pollutants at levels currently encountered in New York City adversely affects fetal development. Key words: adducts, birth outcomes, development, environmental, ETS, PAHs, pollutants, prenatal. Environ Health Perspect 112:626-630 (2004) . doi:10.1289/ehp.6617 available via http://dx.doi.org/ [Online 14 January 2004]
Address correspondence to F.P. Perera, Columbia Center for Children's Environmental Health, Mailman School of Public Health, Columbia University, 60 Haven Ave., #B-109, New York, New York 10032 USA. Telephone: (212) 304-7280. Fax: (212) 544-1943. E-mail: Fpp1@columbia.edu We thank D. Holmes, M. Borjas, A. Reyes, J. Ramirez, L. Cruz, L. Qu, Y. Cosme, S. Illman, L. Needham, R. Jackson, Harlem Hospital, Allen Pavillion, and New York-Presbyterian Hospital. Support was provided by the National Institute of Environmental Health Sciences (grants P50 ES09600, 5 RO1 ES08977, RO1ES111158, RO1 ES012468, RO1 ES10165) , the U.S. Environmental Protection Agency (grants R827027, 8260901, and NCER STAR Program) , Irving General Clinical Research Center (grant RR00645) , Bauman Family Foundation, Gladys & Roland Harriman Foundation, New York Community Trust, Educational Foundation of America, and the Horace W. Goldsmith Foundation. The authors declare they have no competing financial interests. Received 29 July 2003 ; accepted 14 January 2004. |
|
|
 |
The impact of environmental toxicants on children's health is increasingly being recognized as significant [Faustman 2000; Greater Boston Physicians for Social Responsibility (GBPSR) 2000; Landrigan et al. 1999; Perera et al. 1999; U.S. Environmental Protection Agency (EPA) 1996]. Human and experimental studies indicate that the fetus and infant are more sensitive than adults to many environmental toxicants, including environmental tobacco smoke (ETS), and polycyclic aromatic hydrocarbons (PAHs) (Mott et al. 1994; National Academy of Sciences 1993; World Health Organization (WHO) 1986; Whyatt and Perera 1995]. Urban, minority populations represent high-risk groups for adverse birth outcomes [New York City Department of Health (NYCDH) 1998; Perera et al. 2002]. These same populations are likely to be more heavily exposed to ambient air pollution and ETS (Chen and Petitti 1995; Heritage 1992; Metzer et al. 1995; Olden and Poje 1995; Pirkle et al. 1996; Wagenknecht et al. 1993; Wernette and Nieves 1992). However, only limited information is available on the extent and impact of prenatal exposure to these environmental contaminants on fetal growth and development. Etiologic studies have largely been ecologic in nature, lacking individual-level data on exposure.
In the present study, we evaluated the effects of prenatal exposure to common urban pollutants: environmental PAHs estimated by DNA adducts in white blood cells (WBCs) formed by benzo[a]pyrene (BaP), a representative PAH, and ETS estimated by questionnaire data and plasma concentrations of cotinine. In addition to being genotoxic and carcinogenic, PAHs such as BaP are endocrine disruptors (Bostrom et al. 2002; Bui et al. 1986; Davis et al. 1993). Prior laboratory and two human studies in Central Europe indicate that transplacental exposure to PAHs at relatively high concentrations (annual average airborne concentrations of 7-17 ng/m3 BaP in the human studies) is associated with adverse birth outcomes (Barbieri et al. 1986; Bui et al. 1986; Dejmek et al. 2000; Legraverend et al. 1984; Perera et al. 1998). We recently reported that prenatal PAH exposure estimated by personal air monitoring was associated with reduced birth weight and head circumference among African Americans in the present New York City, New York, cohort (Perera at al. 2003). ETS is a complex mixture of > 4,000 chemicals, including PAHs and carbon monoxide (Leikauf et al. 1995). ETS measured by self-report or by biomarkers such as cotinine has been shown in many studies to adversely affect fetal growth as well as child growth and development (reviewed in Eskenazi et al. 1995; Etzel 1997; National Research Council 1986). Adverse effects include deficits in birth weight, birth length, and cognitive functioning at age 3 (Janerich et al. 1990; Martinez et al. 1994; Schuster-Kolbe and Ludwig 1994; Sexton et al. 1990). Likely mechanisms underlying the adverse effects of fetal exposure to ETS include anti-estrogenic effects, induction of P450 enzymes, DNA damage resulting in activation of apoptotic pathways, binding to receptors for placental growth factors resulting in decreased exchange of oxygen and nutrients, and direct effects of carbon monoxide.
Here we tested the hypothesis that prenatal exposure to environmental pollutants alone and/or in combination is negatively associated with birth weight, length, and head circumference, after controlling for the effects of known physical, biologic, and toxic determinants of fetal growth. As reported previously, the study cohort has substantial exposure to multiple contaminants during pregnancy (Perera et al. 2002; Whyatt et al. 2002, 2003). Specifically, analysis of PAHs in air samples from the first 250 subjects showed that all samples had detectable levels of one or more carcinogenic PAHs, ranging across 4 orders of magnitude (Perera et al. 2002). Almost half of the mothers and infants initially enrolled had cotinine levels indicative of ETS exposure ( 0.05-25 ng/mL). Maternal and newborn plasma cotinine levels were significantly higher for mothers who reported smoking by others in the household than for mothers who reported no smoking in the home (p < 0.001). 0.05-25 ng/mL). Maternal and newborn plasma cotinine levels were significantly higher for mothers who reported smoking by others in the household than for mothers who reported no smoking in the home (p < 0.001).
|
Table 1.
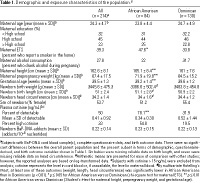
|
Study subjects. Study subjects are Dominican and African-American women residing in Washington Heights, Central Harlem, and the South Bronx, New York, who delivered at New York Presbyterian Medical Center (NYPMC), Harlem Hospital (HH), or their satellite clinics (Perera et al. 2002; Whyatt et al. 2002); Table 1 presents demographic and exposure characteristics of the population. Ethnicity was self-identified. Women were eligible if they were nonsmokers; were 18-35 years of age; were registered at the obstetrics and gynecology clinics at NYPMC and HH by the 20th week of pregnancy; were free of diabetes, hypertension, or known HIV; and had resided in the area for at least 1 year. The mean gestational age at enrollment was 39.5 weeks. Two hundred ninety-eight women were considered to be fully enrolled in the study; that is, they had been monitored prenatally during the third trimester using a personal air monitor and had delivered, and a maternal and/or umbilical cord blood sample had been collected.
The 214 subjects included in the present analysis are those with adduct measurements in umbilical cord blood samples (in some cases the amount of blood collected was inadequate for the assay), and complete questionnaire and medical record data were used as covariates in the multiregression models. Fully enrolled subjects missing any of these data points (n = 84) were excluded from the analysis. Only nonsmokers were included. Nonsmokers were initially defined as having answered "no" to the question "presently, does a household member or regular visitor to your home smoke cigarettes, pipes, marijuana, or cigars in your home" and as having plasma cotinine concentrations 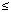 15 ng/mL. Cotinine data were available for approximately 90% of the subjects. Three subjects with plasma cotinine concentrations > 15 ng/mL were excluded to rule out active smoking. There were no significant differences in sociodemographic characteristics or levels of exposure between the present subset and fully enrolled subjects with missing data required in the present analysis. 15 ng/mL. Cotinine data were available for approximately 90% of the subjects. Three subjects with plasma cotinine concentrations > 15 ng/mL were excluded to rule out active smoking. There were no significant differences in sociodemographic characteristics or levels of exposure between the present subset and fully enrolled subjects with missing data required in the present analysis.
Personal interview. A 45-min questionnaire was administered by a trained bilingual interviewer during the last trimester of pregnancy. The questionnaire included demographic information, lifetime residential history (country of birth, location, and duration of residence), travel outside the current area of residence during the past year, history of active and passive smoking (including number of household members who smoke), alcohol use during each trimester of pregnancy, and consumption of PAH-containing meat (frequency of eating fried, broiled, or barbecued meat during the last 2 weeks). Socioeconomic information related to income and education was also collected. The questionnaire was based on that used in a prior study of women and newborns and adapted for the New York City population (Perera et al. 1998).
Biologic sample collection and analysis. Maternal blood (30-35 mL) was collected within 1 day postpartum, and umbilical cord blood (30-60 mL) was collected at delivery. Samples were transported to the laboratory immediately. The buffy coat, packed red blood cells, and plasma samples were separated and stored at -70°C. A portion of each sample was shipped to the Centers for Disease Control and Prevention (CDC) for analysis of cotinine (2 mL) and pesticides (10 mL). Plasma cotinine was analyzed by the CDC using high-performance liquid chromatography atmospheric-pressure ionization tandem mass spectrometry as described previously (Bernert et al. 1997, 2000). The limit of detection for cotinine was 0.05 ng/mL.
DNA adducts. BaP-DNA adducts in extracted WBC DNA from maternal and cord blood were analyzed by the HPLC/fluorescence method of Alexandrov et al. (1992), which uses an HPLC method to detect BaP tetromers. This assay is a sensitive and specific method for measuring BaP-DNA adducts in WBCs from individuals exposed to BaP (Bartsch 1996). The method has a coefficient of variation of 12%. Samples from mother-child pairs were run in the same batch.
Measures relevant to birth outcomes. Information abstracted by the research workers from mothers' and infants' medical records after delivery included date of delivery; gestational age at birth (based on the last menstrual period); infant sex, birth weight, length, head circumference, infant malformations, and Apgar scores; maternal height, prepregnancy weight, and total weight gain; complications of pregnancy and delivery; and medications used during pregnancy.
Statistical analysis. As described above, to exclude active smokers we removed subjects with cotinine levels > 15 ng/mL. Additional analyses were also done after further removing the nine subjects who reported smoking any amount during pregnancy, and the results were materially unchanged. Adducts were used both as a continuous variable and as a dichotomous one. We defined high adducts as > 0.36 adducts/10-8 nucleotides (the median of the detectable adduct values or the upper 20% percentile). As in prior studies, in the analysis of the relationship between adducts and birth outcomes, cord blood adducts were used as the independent variable (Perera et al. 2000). The maternal and cord plasma concentrations of cotinine were significantly correlated (Spearman's rank, r = 0.887; p < 0.001). Therefore, in the 30 cases where the umbilical cord cotinine levels were not available, the mothers' values were used. High/low cotinine was dichotomized using the median of all samples as the cutpoint (0.0435 ng/mL).
The relationships between the exposure variables and the birth outcomes were analyzed by multiple regression, adjusting for known or potential confounders. In addition to cord blood adducts dichotomized as high/low and self-reported ETS (yes/no smokers in the home), the final regression model included covariates representing known or suspected risk factors that were associated with birth outcomes (p 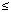 0.1 by linear regression. Birth outcomes were log transformed to provide a better fit to the data and/or to approximate the normal distribution and stabilize the variance. Models 1, 2, and 3 evaluated the main effects of self-reported ETS, high/low cotinine, and high/low BaP-DNA, adjusting for potential confounders, including ethnicity, body mass index, gestational age, dietary PAHs, infant sex, and cesarean delivery (a predictor of head circumference). Income (< $10,000 or 0.1 by linear regression. Birth outcomes were log transformed to provide a better fit to the data and/or to approximate the normal distribution and stabilize the variance. Models 1, 2, and 3 evaluated the main effects of self-reported ETS, high/low cotinine, and high/low BaP-DNA, adjusting for potential confounders, including ethnicity, body mass index, gestational age, dietary PAHs, infant sex, and cesarean delivery (a predictor of head circumference). Income (< $10,000 or  $10,000), parity (0 or $10,000), parity (0 or  1 live birth), social adversity (a composite score based on marital status, income, education, and whether currently on assistance), and alcohol consumption (yes/no) were not significant predictors of outcomes (p > 0.1) and were not included. The other variables, including dietary PAHs, were included as covariates. The final models tested the interaction between adducts and ETS (or cotinine) using appropriate interaction terms, adducts, and ETS (or cotinine). 1 live birth), social adversity (a composite score based on marital status, income, education, and whether currently on assistance), and alcohol consumption (yes/no) were not significant predictors of outcomes (p > 0.1) and were not included. The other variables, including dietary PAHs, were included as covariates. The final models tested the interaction between adducts and ETS (or cotinine) using appropriate interaction terms, adducts, and ETS (or cotinine).
Demographic and exposure characteristics for the subjects included in the present analysis are provided in Table 1 together with summary data on cord blood BaP-DNA and cotinine. The subset did not differ from the overall cohort in terms of demographic variables. Among subjects in the present analysis, 46% of mothers and 49% of newborns had cotinine levels > 0.05 and 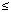 15 ng/mL, indicative of ETS exposure. Self-reported ETS and plasma cotinine differed by ethnicity, with African Americans being significantly more likely to report ETS exposure (p 15 ng/mL, indicative of ETS exposure. Self-reported ETS and plasma cotinine differed by ethnicity, with African Americans being significantly more likely to report ETS exposure (p  0.05) and to have a higher rate of detectable cotinine (78.1 vs. 31.9%; p 0.05) and to have a higher rate of detectable cotinine (78.1 vs. 31.9%; p  0.01). Sixty-two percent of cord and 61% of maternal blood samples had nondetectable levels of BaP-DNA (< 0.25 adducts/10-8 nucleotides). 0.01). Sixty-two percent of cord and 61% of maternal blood samples had nondetectable levels of BaP-DNA (< 0.25 adducts/10-8 nucleotides).
The mean birth weight was 3445.6 g (SD = 475.3). Mean head circumference was 34.2 cm (SD = 1.4). Mean birth weight and head circumference were lower, and there was greater variability in these outcomes, among African-American than among Dominican infants. The differences between individual outcomes were not significant by t-test; however, by multivariate Hotelling's t-test, at least one of these outcomes (weight, length, head circumference) was significantly lower in African Americans (p < 0.01). Reflecting the fact that all women had reached their third trimester of pregnancy, only 3% percent of infants were preterm (< 37 weeks of gestation). African-American infants had a significantly lower mean gestational age than did Dominican infants (39.2 vs. 39.6 weeks, p  0.01). 0.01).
|
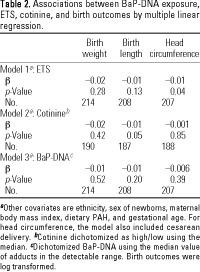
Table 2.
|
|
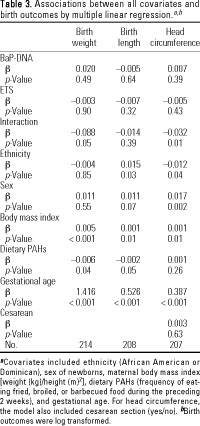
Table 3.
|

Figure 1. Bar graph showing interaction effect of BaP-DNA and ETS on (A) birth weight and (B) head circumference. Abbreviations: ETS-, ETS-negative; ETS+, ETS-positive. The covariates in the graph are BaP-DNA adducts (high/low) and self-reported ETS (yes/no). The graph shows a significant interaction on birth weight (p = 0.05) and on head circumference (p = 0.01). Other covariates in the models are shown in Table 3.
|
As shown in Table 2 by linear regression, ETS exposure was associated with smaller head circumference (ß = -0.01, p = 0.04) after adjusting for potential confounders (model 1). Cotinine was significantly associated with birth length (ß = -0.01, p = 0.05). By Spearman's test, BaP-DNA adducts were not significantly correlated with ETS, cotinine, or dietary PAHs. BaP-DNA alone (either as a continuous or dichotomous variable) was not significantly associated with birth outcomes. However, the interaction between high/low adducts and ETS was significant using either adduct variable. For example, as shown in Table 3, the inter-action effect of BaP-DNA and ETS was significant on birth weight (p = 0.05) and head circumference (p = 0.01). Figure 1 shows the effect of this interaction on birth weight and head circumference. There was a 233-g (6.8%) reduction of birth weight and 1-cm (2.9%) reduction of head circumference in the ETS-positive group with high BaP-DNA levels, compared with the ETS-negative group with low BaP-DNA levels. The interaction effect of BaP-DNA and cotinine was not significant.
In this study we found an association between ETS, with or without BaP-DNA adducts, and two birth outcomes, namely decreased birth weight and smaller head circumference, after adjusting for potential confounders. This association is of potential concern because, although the literature is inconsistent, several studies have reported that reduction in birth weight and head circumference at birth correlates with lower IQ as well as poorer cognitive functioning and school performance in childhood (Chaikind and Corman 1991; Desch et al. 1990; Matte et al. 2001). In a prior study, we found that the levels of PAH-DNA adducts in cord blood of Caucasian newborns in Poland, who were exposed to 10-fold higher levels of PAHs in air pollution than was the New York City cohort, were significantly associated with lower birth weight, reduced length, and reduced head circumference (Perera et al. 1998). In the present study in New York City, at lower levels of exposure, the effect of adducts was seen only in the interaction with ETS. The finding of a significant interactive effect between the two exposures, along with the observed lack of correlation between adducts and ETS, suggests that the effect of ETS is largely caused by other non-PAH constituents of tobacco smoke and that the adducts may largely reflect other environmental sources of PAHs as well as individual susceptibility to them. We note that in this sample, ingestion of dietary PAHs was not correlated with adducts.
Fetal toxicity from ETS and PAHs may be caused by anti-estrogenic effects (Bui et al. 1986), binding of constituents to the human aryl hydrocarbon receptor to induce P450 enzymes (Manchester et al. 1987), DNA damage resulting in activation of apoptotic pathways (Meyn 1995; Nicol et al. 1995; Wood and Youle 1995), binding to receptors for placental growth factors resulting in decreased exchange of oxygen and nutrients, or direct effects of carbon monoxide (Dejmek et al. 2000; National Research Council 1986).
In this study, there were no significant differences in exposure-effect relationships between African Americans and Dominicans. The combination of high BaP-DNA adducts and ETS exposure was associated with a 7% reduction of birth weight and a 3% reduction of head circumference.
PAHs are only one class of chemicals among many found in particulate matter from combustion sources. Nevertheless, our results are consistent with ecologic studies showing associations between ambient levels of air pollutants (including total suspended particulate matter) and low birth weight (Bobak 2000; Chen and Omaye 2001; Ha et al. 2001). A study in Northern Bohemia found that estimated exposure to carcinogenic PAHs in early gestation (based on ambient air monitoring data) was associated with reduced fetal growth (Dejmek et al. 2000).
This study has the advantage of being based on individual prenatal exposure data from biomarkers as well as extensive medical records and questionnaire data. However, it is limited by the modest sample of subjects (214) for whom data from all relevant domains are currently available. The study had insufficient power to examine size for gestational age, and the study design precluded hypotheses related to preterm delivery. Relationships observed in low-income, minority women might be different in women of other races or ethnic, cultural, or socioeconomic backgrounds. However, we reported previously that DNA damage from PAHs was associated with worse birth outcomes among Caucasians in Central Europe who were exposed to air pollution from coal burning (Perera et al. 1998). Finally, we were able to measure biomarkers only at a single time point. In cases of chronic exposure, a single measure can be a representative internal dosimeter; however, this may not be true if exposures are sporadic and biomarkers have short half-lives. This may explain why we saw a stronger association between self-reported ETS exposure and birth outcomes than between cotinine and birth outcomes. Because cotinine is a short-lived biomarker, cotinine measurement at delivery may not be a good indicator of levels throughout the pregnancy.
This study provides new molecular epidemiologic evidence that exposure to common environmental pollutants (PAHs and ETS) at levels currently encountered in New York City can act in combination to adversely affect fetal development.
|
|
 |
| [References Listed in PubMed]
References
Alexandrov K, Rojas M, Geneste O, Castegnaro M, Camus AM, Pesruzzelli S, et al. 1992. An improved fluorometric assay for dosimetry of benzo(a)pyrene diol-epoxide-DNA adducts in smokers' lung: comparisons with total bulky adducts and aryl hydrocarbon hydroxylase activity. Cancer Res 52:6248-6253.
Barbieri O, Ognio E, Rossi O, Astigiano S, Rossi L. 1986. Embryotoxicity of benzo(a)pyrene and some of its synthetic derivatives in Swiss mice. Cancer Res 46:94-98.
Bartsch H. 1996. DNA adducts in human carcinogenesis: etiological relevance and structure-activity relationship. Mutat Res 340:67-69.
Bernert JT, McGuffey JE, Morrison MA, Pirkle JL. 2000. Comparison of serum and salivary cotinine measurements by a sensitive high-performance liquid chromatography-tandem mass spectrometry method as an indicator of exposure to tobacco smoke among smokers and nonsmokers. J Anal Toxicol 24:333-339.
Bernert JT, Turner WE, Pirkle JL, Sosnoff CS, Akins JR, Waldrep MK, et al. 1997. Development and validation of sensitive method for determination of serum cotinine in smokers and nonsmokers by liquid chromatography/atmospheric pressure ionization tandem mass spectrometry. Clin Chem 43:2281-2291.
Bobak M. 2000. Outdoor air pollution, low birth weight, and prematurity. Environ Health Perspect 108:173-176.
Boström CE, Gerde P, Hanberg A, Jernström B, Johansson C, Kyrklund T, et al. 2002. Cancer risk assessment, indicators, and guidelines for polycyclic aromatic hydrocarbons in the ambient air. Environ Health Perspect 110(suppl 3):451-488.
Bui QQ, Tran MB, West WL. 1986. A comparative study of the reproductive effects of methadone and benzo(a)pyrene in the pregnant and pseudopregnant rat. Toxicology 42:195-204.
Chaikind S, Corman H. 1991. The impact of low birth weight on special education costs. J Health Econ 10:291-311.
Chen L, Omaye ST. 2001. Air pollution and health effects in northern Nevada. Rev Environ Health 16:133-149.
Chen L, Petitti DB. 1995. Case-control study of passive smoking and the risk of small-for-gestational-age at term. Am J Epidemiol 142:158-165.
Davis DL, Bradlow HL, Wolff M, Woodruff T, Hoel DG, Anton-Culver H. 1993. Medical hypothesis: xenoestrogens as preventable causes of breast cancer. Environ Health Perspect 101:372-377.
Dejmek J, Solansky I, Benes I, Lenicek J, Srám RJ. 2000. The impact of polycyclic aromatic hydrocarbons and fine particles on pregnancy outcome. Environ Health Perspect 108:1159-1164.
Desch LW, Anderson SK, Snow JH. 1990. Relationship of head circumference to measures of school performance. Clin Pediatr (Phila) 29:389-392.
Eskenazi B, Prehn AW, Christianson RE. 1995. Passive and active maternal smoking as measured by serum cotinine: the effect on birthweight. Am J Public Health 85:395-398.
Etzel RA. 1997. Active and passive smoking: hazards for children. Cent Eur J Public Health 5:54-56.
Faustman EM. 2000. Mechanisms underlying children's susceptibility to environmental toxicants. Environ Health Perspect 108(suppl 1):13-21.
GBPSR (Greater Boston Physicians for Social Responsibility). 2000. In Harm's Way: Toxic Threats to Child Development. Cambridge, MA:Greater Boston Physicians for Social Responsibility.
Ha EH, Hong YC, Lee BE, Woo BH, Schwartz J, Christiani DC. 2001. Is air pollution a risk factor for low birth weight in Seoul? Epidemiology 12:643-648.
Heritage J. 1992. Environmental protection--has it been fair? EPA J 18(1):1-64.
Janerich DT, Thompson WD, Varela LR, Greenwald P, Chorost S, Tucci C, et al. 1990. Lung cancer and exposure to tobacco smoke in the household. N Engl J Med 323:632-636.
Landrigan PJ, Claudio L, Markowitz SB, Berkowitz GS, Brenner BL, Romero H, et al. 1999. Pesticides and inner-city children: exposures, risks, and prevention. Environ Health Perspect 107(suppl 3):431-437.
Legraverend C, Guenthner TM, Nebert DW. 1984. Importance of the route of administration for genetic differences in benzo(a)pyrene-induced in utero toxicity and teratogenicity. Teratology 29:35-47.
Leikauf GD, Kline S, Albert R, Baxter CS, Bernstein DI, Bernstein J, et al. 1995. Evaluation of a possible association of urban air toxics and asthma. Environ Health Perspect 103(suppl 6):253-271.
Manchester DK, Gordon SK, Golas CL, Roberts EA, Okey AB. 1987. Ah receptor in human placenta: stabilization by molybdate and characterization of binding of 2,3,7,8-tetrachlorodibenzo-p-dioxin, 3-methylcholanthrene, and benzo(a)pyrene. Cancer Res 47:4861-4868.
Martinez FD, Wright AL, Taussig LM. 1994. The effect of paternal smoking on the birth weight of newborns whose mothers did not smoke. Am J Public Health 84:1489-1491.
Matte TD, Bresnahan M, Begg MD, Susser E. 2001. Influence of variation in birth weight within normal range and within sibships on IQ at age 7 years: cohort study. Br Med J 323:310-314.
Metzger R, Delgado JL, Herrell R. 1995. Environmental health and Hispanic children. Environ Health Perspect 103(suppl 6):25-32.
Meyn MS. 1995. Ataxia-telangiectasia and cellular responses to DNA damage. Cancer Res 55:5991-6001.
Mott L, Vance F, Curtis J. 1994. Handle with Care: Children and Environmental Carcinogens. New York:Natural Resources Defense Council.
National Academy of Sciences. 1993. Pesticides in the Diets of Infants and Children. Washington, DC:National Academy Press.
National Research Council. 1986. Environmental tobacco smoke: measuring exposures and assessing health effects. Washington, DC:National Academy Press.
Nicol CJ, Harrison ML, Laposa RR, Gimelshtein IL, Wells PG. 1995. A teratologic suppressor role for p53 in benzo[a]pyrene-treated transgenic p53-deficient mice. Nat Genet 10:181-187.
NYCDH (New York City Department of Health). 1998-1999. Vital Statistics. New York City:New York City Department of Health.
Olden K, Poje J. 1995. Environmental justice and environmental health. Bull Soc Occup Environ Health 4:3-4.
Perera FP, Illman SM, Kinney PL, Whyatt RM, Kelvin EA, Shepard P, et al. 2002. The challenge of preventing environmentally related disease in young children: community-based research in New York City. Environ Health Perspect 110:197-204.
Perera FP, Jedrychowski W, Rauh V, Whyatt RM. 1999. Molecular epidemiologic research on the effects of environmental pollutants on the fetus. Environ Health Perspect 107(suppl 3):451-460.
Perera FP, O'Neill JP, Hemminiki K, Jedrychowski W, Bawle U, Albertini RJ, et al. 2000. A molecular epidemiologic study of the association between environmental exposures to carcinogens and DNA damage and mutation in mothers and newborns [Abstract]. In: Proceedings of the Conference of AACR 91st Annual Meeting: 1-5 April 2000, The Moscone Center, San Francisco, CA. San Francisco, CA:American Association for Cancer Research, 436.
Perera FP, Rauh V, Tsai WY, Kinney P, Camann D, Barr D, et al. 2003. Effects of transplacental exposure to environmental pollutants on birth outcomes in a multi-ethnic population. Environ Health Perspect 111:201-205.
Perera FP, Whyatt RM, Jedrychowski W, Rauh V, Manchester D, Santella RM. 1998. Recent developments in molecular epidemiology: a study of the effects of environmental polycylic aromatic hydrocarbons on birth outcomes in Poland. Am J Epidemiol 147:309-314.
Pirkle JL, Flegal KM, Bernert JT, Brody DJ, Etzel RA, Maurer KR. 1996. Exposure of the US population to environmental tobacco smoke: the Third National Health and Nutrition Examination Survey, 1988 to 1991. JAMA 275:1233-1240.
Schuster-Kolbe J, Ludwig H. 1994. Smoking and the risk of cancer. Wien Med Wochenschr 144:540-544.
Sexton M, Fox NL, Hebel JR. 1990. Prenatal exposure to tobacco: II. Effects on cognitive functioning at age three. Int J Epidemiol 19:72-77.
U.S. EPA. 1996. Environmental Health Threats to Children. Washington, DC:U.S. Environmental Protection Agency.
Wagenknecht LE, Manolio TA, Sidney S, Burke GL, Haley NJ. 1993. Environmental tobacco smoke exposure as determined by cotinine in black and white young adults: the CARDIA study. Environ Res 63:39-46.
Wernette DR, Nieves LA. 1992. Breathing polluted air: minority disproportionately exposed. EPA J 18:16-17.
WHO (World Health Organization). 1986. Principles for evaluating health risks from chemicals during infancy and early childhood: the need for a special approach. In: Environmental Health Criteria 59. Geneva:World Health Organization.
Whyatt RM, Barr DB, Camann DE, Kinney PL, Barr JR, Andrews HF, et al. 2003. Contemporary-use pesticides in personal air samples during pregnancy and blood samples at delivery among urban minority mothers and newborns. Environ Health Perspect 111:749-756.
Whyatt RM, Camann DE, Kinney PL, Reyes A, Ramirez J, Dietrich J, et al. 2002. Residential pesticide use during pregnancy among a cohort of urban minority women. Environ Health Perspect 110:507-514.
Whyatt RM, Perera FP. 1995. Application of biologic markers to studies of environmental risks in children and the developing fetus. Environ Health Perspect 103(suppl 6):105-110.
Wood KA, Youle RJ. 1995. The role of free radicals and p53 in neuron apoptosis in vivo. J Neurosci 15:5851-5857.
Last Updated: March 12, 2004
|
|
 |
|
| |


