|
If you've gathered additional data between the time you submitted and the review, you may be allowed to send it. Contact your scientific review officer (SRO) to find out if this is possible; he or she decides whether to accept any additional information. If the SRO allows this, follow his or her instructions; typically you may email up to two pages and must send it within one month before the review. See the NIH Best Practice Guidelines (MS Word, 57k) for details. Be aware that reviewers are under no obligation to review late materials, and they could reflect either positively or negatively on you.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Your application's most significant test is initial peer review.
Your peers -- successful scientists in your field and related ones -- use the information in your application to assess the merit of the science you've proposed and your ability to get the work done.
A peer review results in numerical value indicating the reviewers' judgment of scientific and technical merit. That number is the most important factor in determining your application's success.
The next section details NIH initial peer review. (For a broad overview, read Perspective on Peer Review in our New Investigator Guide to NIH Funding.)
Find more information online:
- Review Criteria SOP and other Peer Review SOPs
- Peer Review of Applications -- CSR questions and answers
- Peer Review Meetings -- CSR questions and answers
- Peer Review Policies and Practices
- After Peer Review questions and answers
Who Peer Reviews Your Application?
|
NIH peer reviewers are scientists, mostly from academia, who come to NIH three times a year for several days to review applications.
 Our Advice: Know Who's Who in Peer Review
Our Advice: Know Who's Who in Peer Review
The key people involved are:
- Review committee chair -- a committee member who leads the discussions.
- Scientific review officer -- NIH staff member who manages the effort and generally has a Ph.D. in a relevant field of science. SROs recruit reviewers, inform them of policies, create lists of streamlined applications, and write summary statements.
Depending on grant type, initial peer review meetings take place in either CSR or an institute. The process is essentially equivalent in both venues in terms of policy, review criteria, committee composition, conduct of the meetings, and size of the group -- about 20 members.
CSR Review
- Standard research grants, R01s, are reviewed in CSR.
- CSR structures its review committees, also called study sections, into umbrella organizations, the integrated review groups.
- Each IRG houses several study sections, which have a narrower scientific focus.
- See an example of an IRG organization at Infectious Diseases and Microbiology IRG.
- CSR also organizes special panels when special expertise is needed.
- Go to CSR Study Section Roster Index to see members of standing study sections.
NIAID Review
- NIAID has chartered review committees for AIDS; Allergy, Immunology, and Transplantation; and Microbiology and Infectious Diseases.
- They review applications for program projects, cooperative agreements, training and career development awards, and applications responding to requests for applications. Find our standing review committees on Councils and Committees.
- For many RFAs, we set up special review groups that have knowledge relevant to the science.
SROs Assess Completeness, Assign Reviewers
|
Your scientific review officer does an initial check of your application to make sure the key parts are there.
If you're responding to a request for applications, NIAID program staff check to ensure it is responsive to the request for applications.
Before sending your application to reviewers, SROs look at the application more thoroughly to make sure it's complete and may contact you if anything is missing. If this happens, send in the information quickly so reviewers receive it well before the review.
SROs assign primary, secondary, and tertiary reviewers.
- Assigned reviewers read your application thoroughly and write a critique before the meeting. They also assign preliminary scores for each review criterion and an overall impact/priority score.
- SROs may also ask other members to serve as readers.
Four to six weeks before the meeting. SROs send each committee member a copy of all applications to be reviewed. For both paper and electronic applications, CSR and NIAID follow roughly the following procedure:
- Send a password-protected CD of all applications to each reviewer, except those who have a conflict of interest.
- Send paper copies of applications to assigned reviewers.
For example: for a review of 100 applications, a reviewer may receive 95 on CD (five not sent because of conflicts) and paper copies of 15 of the 95 to which he or she is assigned.
Noncompetitive Applications Get a Streamlined Review
|
NIH uses a process called streamlining so reviewers can focus on applications that have a chance of being funded.
Review committees don't review any application the group unanimously feels is roughly in the bottom half of applications being reviewed at the meeting (that percentage varies by grant type as well as by study section).
Since no institute has a 50 percentile payline, these applications are simply not competitive.
Here is how streamlining works:
- One week before the study section meets, SROs ask members for a list of applications they feel should not be reviewed and prepare a combined list.
- If any reviewer disagrees with a call, the group will review that application.
We describe this in more detail below.
Find more information online:
- Unfunded Application Advice questions and answers
- Are there formal "quotas" for the percentage of grants not given a formal review and a score? in the Peer Review Meetings -- CSR questions and answers
 |
If you already have your review results, was your application streamlined?
|
Basic Layout of Initial Peer Review
|
CSR review committees gather three times a year for a one- to three-day meeting.
Initial peer review meetings take place four to five months after the NIH receipt date for applications other than AIDS, one to two months later for AIDS applications.
At the meeting, the scientific review officers make sure the group adheres to policy and procedure. The group's chairperson, a committee member, facilitates the discussions. To experience a peer review meeting, watch CSR's video of a mock study section Video on Peer Review at NIH.
 Our Advice: Know What Happens During Peer Review
Our Advice: Know What Happens During Peer Review
Though they do not participate, institute program staff may attend the meeting and can become a source of additional insight into the discussion.
- You might want to ask your program officer if he or she plans to attend the meeting.
- If you need to revise later, your program officer's feedback can be a valuable supplement to the summary statement. (We discuss this further in Contact Your Program Officer for Feedback.)
After the SRO opens the meeting, the primary reviewer presents your application to the group. (Peer reviewers with a conflict of interest leave the room beforehand.)
- Reviewers review applications in the order of their preliminary overall impact scores.
- This ordering helps them calibrate final scores.
- Starting with the highest scoring applications also helps them gauge when it is appropriate to stop discussing applications. Generally, review groups do not discuss about half of applications. At that point, they decide if other applications merit discussion.
- The group explores differences of opinion, interacting heavily during the discussion, which generally lasts 10 to 15 minutes.
- Other reviewers ask the assigned reviewers questions and skim the application during the discussion.
- Generally, once the members have found a fatal flaw they all agree to, they stop discussing the application.
- Examples of fatal flaws are not protecting the safety of lab workers or animals, proposing too much work for the award time, not recognizing a key paper in the field, or including a factual inaccuracy.
- Where possible, reviewers evaluate applications from new and early-stage investigators together as a separate group within each study section. Clustering these applications ensures that the group reviews at least half of them and that NIH can meet its targets for funding new investigators.
- Review materials are confidential.
- Reviewers are not allowed to divulge any information outside the meeting.
- At the end of the meeting, NIH staff collect and destroy all materials used in the review.
Most Reviewers Scan Each Application
|
Generally, only assigned reviewers will read your application before the review. Other reviewers mostly read just your Abstract, Background and Significance, and Specific Aims.
Reviewers receive dozens of applications for each meeting, totaling thousands of pieces of paper to read in a few weeks -- and they have full-time jobs! They couldn't possibly read all applications in depth.
Keep in mind that about twenty people will score your application even though only a few will have read it in depth. This is the reason you write and organize your Specific Aims for both audiences (we discussed this topic in Plan and Organize Effectively, and Part 5. Research Plan).
You must make a strong case for your research so the primary reviewer can readily read, understand, and explain your project to the group. In Part 4. Target Your Audience and Part 5. Research Plan, we told you how to turn your assigned reviewers into your advocates.
If you're sending in an application you revised based on reviewers' comments from the last review, the reviewers will evaluate your response. For details, see How to Revise and Resubmit.
Assigning an Overall Impact/Priority Score
|
Reflecting the reviewers' judgment of the technical and scientific merit of your application, the overall impact/priority score is an essential review outcome.
- As we told you above, before the meeting the assigned reviewers score each criterion and determine a preliminary overall impact/priority score.
- The discussion at the meeting may cause assigned reviewers to change what overall impact/priority score they suggest to the group at the end.
- Next, all reviewers vote.
- Assigned reviewers enter their official scores for each criterion and an overall impact/priority score on the vote sheet. The other reviewers can see these scores.
- Other reviewers give an overall impact/priority score (but usually have an option of scoring each criterion).
- Each member marks scores privately on a vote sheet, assigning a whole number from 1 (best) to 9 (worst). For an illustration of the new scores, see the excellent graphic below that NIH created and we adapted.
- At the end of the meeting, the scientific review officer collects vote sheets and adds the scores.
- To create a raw overall impact/priority score, scores are averaged and rounded mathematically to one decimal place, e.g., a 1.34 average yields 1.3. That number is multiplied by 10 to yield an overall impact/priority score, e.g., 13.
- R01 applications also get a percentile. Learn how NIH creates percentiles at Understanding Paylines and Percentiles in Part 10. Funding Decisions of the NIH Grant Cycle: Application to Renewal.
For more information on funding decisions, see How NIAID Determines Which Applications to Fund.
Scoring Table for Research Grants
Impact |
Impact/Priority Score |
Descriptor |
Additional Guidance on Strengths/Weaknesses |
Strengths/Weaknesses |
|---|---|---|---|---|
High |
1 |
Exceptional |
Exceptionally strong with essentially no weaknesses |
|
2 |
Outstanding |
Extremely strong with negligible weaknesses |
||
3 |
Excellent |
Very strong with only some minor weaknesses |
||
Moderate |
4 |
Very Good |
Strong but with numerous minor weaknesses |
|
5 |
Good |
Strong but with at least one moderate weakness |
||
6 |
Satisfactory |
Some strengths but also some moderate weaknesses |
||
Low |
7 |
Fair |
Some strengths but with at least one major weakness |
|
8 |
Marginal |
A few strengths and a few major weaknesses |
||
9 |
Poor |
Very few strengths and numerous major weaknesses |
Definitions | |
| Minor: easily addressable weakness that does not substantially lessen the impact of the project. Moderate: weakness that lessens the impact of the project. Major: weakness that severely limits the impact of the project. |
||||
How NIH Review Criteria Affect Your Score
|
When assessing the scientific and technical merit of an application, all NIH review committees use the same initial peer review criteria -- see the Review Criteria SOP.
If you're responding to a request for applications, check the RFA for any special review criteria.
Role of Review Criteria
Peer reviewers don't score applications strictly by the review criteria. Rather, the criteria are gauges for assessing merit and feasibility.
Your assigned reviewers give your application a score for each criterion as well as the whole application; other reviewers score just the whole application.
Keep in mind that the criteria are unweighted and unrelated to the final overall impact/priority score, which reflects the reviewers' judgment of your application as a whole.
It's important to understand how review criteria relate to your score:
- Overall impact and merit. A final score reflects a judgment of overall potential impact and scientific merit.
- Ideal application. To a large extent, reviewers
judge your application based on their ideal outstanding application in
your field of science.
- This is similar to a dog show, where dogs are judged for "best of breed," and different breeds do not compete with each other.
- So there is not a one-to-one relationship between how your application measures up to the review criteria and your score.
- Usage varies. Adherence to the criteria varies by committee.
- Weight varies. An application does not need to be strong in all review criteria to get a high overall impact/priority score, though all criteria can affect your score. Two example:
- Reviewers may assign an exceptional score to a proposal for important work that is not innovative but is essential to move a field forward.
- An application with high significance may receive an outstanding overall impact/priority score even if reviewers are less enthusiastic about the other criteria.
Other Critical Factors Can Affect Your Score
Your reviewers will consider other items besides the review criteria.
- Special areas. Depending on the experiments you propose, they will make sure you have complied with NIH policies for recombinant DNA research, human subjects, research animals, and select agents.
- Presentation. Your presentation can make or break your application.
- Though reviewers assess science, they are also influenced by the writing and appearance of your application. (See the Insider's Guide to Peer Review for Applicants.)
- If your application has lots of typos and internal inconsistencies, your score can suffer. (We covered this topic in Write to Your Audience, Plan and Organize Effectively, and Write, Edit, and Proof Like a Pro.)
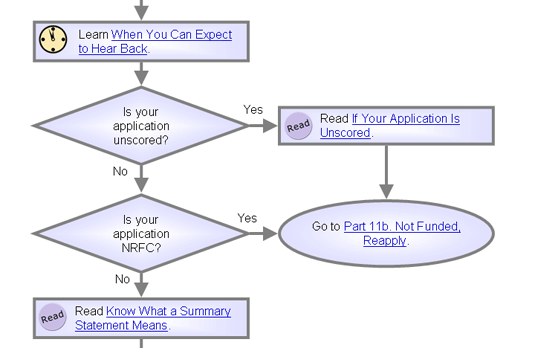
If Your Application Is Unscored
|
Three types of applications are unscored -- they do not receive a full review, overall impact/priority score, or summary statement.
It's important to keep in mind that unscored applications can still be high quality and possibly fundable. To learn more about this now, read below starting at If Your Application Scores Above the Payline or Is Unscored.
Keep in mind the length of time it may take from the time you reapply to the time an award may be received. See R01 Application to Award Timeline for Initial Submission and Resubmission and Timeline by Review Cycle.
1. Streamlined review. Applications that peer reviewers unanimously judge to be roughly in the bottom half of applications being reviewed at the meeting (though the percentage varies by grant type as well as by study section) get a streamlined review.
Streamlined applications are not discussed at the meeting and do not receive an actual overall impact/priority score. Instead, principal investigators get scores for the individual review criteria and critiques from assigned reviewers.
For more information, see Noncompetitive Applications Get a Streamlined Review above and If Your Application Scores Above the Payline or Is Unscored below.
2. Not recommended for further consideration. NRFC is used for applications that lack significant and substantial scientific merit or have serious hazards or ethical issues. Such applications do not warrant a review and are generally not eligible for funding.
3. Deferred. A scientific review group can postpone the review of an application if unable to determine its scientific merit because information is missing. The group may contact the applicant right away or request another review at a later review date.
 |
Do you already know how your application fared in review?
|
 |
Was your application unscored or NRFC?
|
 |
Is your application foreign?
|
Comparing Old and New Peer Review Processes
The table below highlights the differences between both approaches.
Comparison of NIH's Old and New Peer Review Processes |
||
Function |
Old |
New |
|---|---|---|
| Assignment of overall impact/priority scores | Scores reflect reviewer judgment of a whole application: peer review criteria are unweighted and unrelated to the overall impact/priority score. | Unchanged, except assigned reviewers also score each criterion; those scores are also unrelated to the overall score. |
| Each reviewer scores to one decimal place: 1.0 is best, 5.0 worst. | Each reviewer scores in whole numbers: 1 is best, 9 worst. | |
| Determination of overall impact/priority scores | To create a raw score, reviewer scores are averaged and rounded mathematically to two decimal places, e.g., 1.34. The result is multiplied by 100 to give an overall impact/priority score, e.g., 134. The possible scores range from 100 to 500. |
To create a raw score, reviewer scores are averaged and rounded mathematically to one decimal place, e.g., a 1.34 average yields 1.3. The result is multiplied by 10 to give an overall impact/priority score, e.g., 13. The possible scores range from 10 to 90. |
| Streamlined applications | Principal investigators get critiques from assigned reviewers. | Principal investigators get initial scores for individual criteria and critiques from assigned reviewers. |
| Determination of percentiles | Percentiles range from 0.1 (best) to 99.5 (worst). Read How Percentiles Are Determined. | Percentiles range from 1 to 99 in whole numbers. Rounding is always up, e.g., 12.1 percentile becomes 13. |
| With almost 1,000 possible percentile rankings, few applications are ranked the same. | With 99 possible percentile rankings, some applications are ranked the same, making funding decisions more challenging. For tie scores, funding decisions will be based on other important factors such as mission relevance and portfolio balance. |
|
| Percentile base | NIH calculates percentiles using applications submitted for three review cycles. | Unchanged, except for the first year of the transition to the new review processes: |
Summary statements |
See a sample: Annotated Summary Statement (PDF). | Assigned reviewers provide feedback through scores for each criterion and critiques in a structured summary statement. |
Review Criteria |
Five one-word criteria plus descriptive information. | One-word criteria unchanged; descriptions modified. See Review Criteria SOP. |
Foreign Applications Have an Extra Review Step
|
NIH awards grants to foreign applicants if either the expertise or resources are not available here -- for example, access to a unique study population.
- Foreign institutions have an extra review step: reviewers assess whether comparable work is being done in the U.S. If it is, the grant will not likely be funded.
- Reviewers also consider the relevance of the proposed research to NIAID's mission and check whether there is a need for the research. If similar work is being done in the U.S., the application will suffer in review.
- Foreign investigators researching select agents must follow the NIAID Select Agent Research Review and Approval Procedure for New and Continuing Grants that Include Foreign Institutions.
- If using select agents, read NIAID Select Agent Policy for Foreign Institutions questions and answers.
Find more information online:
- Grants Policy and Management Training for Foreign Investigators
- Does NIH Fund Foreign Institutions and Investigators?
- NIAID International Awards
- Foreign and International Grants and Components SOP
- Foreign Workers on NIH Awards SOP
- International Awards questions and answers
Reviewers Are Fair But Not Always Right
|
Bias is extremely rare.
- Peer reviewers themselves go through the same process you're going through. If they aren't fair to you, how could they expect to be treated fairly themselves?
- If anything, reviewers have tried to capture more funds for their field by giving applications increasingly better scores.
- Reviewers and scientific review officers are alert to bias and will argue vigorously against it if they perceive a competitor is not being fair.
- Though reviewers generally are fair, they are not always right.
- They do their best based on the knowledge they have but could miss a point or misunderstand what you've written.
- For this reason, you'll need to do an outstanding job of writing and organizing your application.
- Any reviewer who has a conflict of interest with an application is not allowed to review it. Reviewers sign conflict of interest statements stating they don't have a financial or other interest in your work.
For details on this subject, see the Conflict of Interest in Peer Review SOP and the Privacy, Conduct, Conflict of Interest, and Clinical Research Ethics questions and answers.
|
When You Can Expect to Hear Back
After peer review, all reviewed applications receive an overall impact/priority score and summary statement.
NIH releases summary statements to you and your program officer roughly six to eight weeks after the review meeting, earlier for new investigators. You can retrieve this information from the Commons:
- Overall impact/priority score -- about five working days after the review meeting.
- Summary statement -- about eight weeks after a review meeting, earlier for new investigators.
At that time, it's a good idea to call your program officer if you haven't done so already.
Ask whether your application is likely to be funded or whether he or she can give you more feedback from the review if funding is not on the horizon.
Go to R01 Application to Award Timeline for Initial Submission and Resubmission and Timeline by Review Cycle.
If Your Application Scores Above the Payline or Is Unscored
 Our Advice: If Problems Are Fixable, Start Revising Quickly
Our Advice: If Problems Are Fixable, Start Revising Quickly
If your application missed the payline or is unscored and its faults are fixable, start revising as soon as you can since you may not have much time to revise after you get the summary statement. See R01 Application to Award Timeline for Initial Submission and Resubmission and Timeline by Review Cycle.
First, determine whether the problems are fixable -- read Part 11b. Not Funded, Reapply.
 |
Are you thinking of appealing the review?
|
Know What a Summary Statement Means
|
Scientific review officers prepare summary statements for applications considered to be competitive for funding -- i.e., those given a full review and an overall impact/priority score by the review committee.
- See an Annotated Summary Statement Sample.
- Your summary statement holds a wealth of information: reviewer critiques, summary of the discussion, overall impact/priority score, percentile, recommended budget, human and animal subjects codes, and any administrative comments.
- Find the overall impact/priority scores and criterion scores from assigned reviewers.
- If your summary statement has a code for a bar to award, we can't give you an award until you resolve the issue.
- Contact your program officer immediately. You may want to read more in the Bars to Grant Awards SOP.
- Bars reflect concerns about human subjects, animals, or biohazards.
- To find out what the codes mean, see Research Animals Involvement Codes, Human Subjects Involvement Codes, and Human Subjects Inclusion Codes.
- A summary statement is not an exhaustive critique.
- It is not a teaching tool containing every point reviewers found to be problematic. Instead, it recaps the highlights of the review discussion as far as it progressed, providing general feedback.
- You'll use this information to revise a fixable application, if necessary.
- Read Summary Statements Have Their Limitations.
 Our advice. After you read the summary statement, contact your NIAID program officer. Ask whether your application
is likely to be funded and whether he or she can give you more
feedback
from the review if funding is not on the horizon.
Our advice. After you read the summary statement, contact your NIAID program officer. Ask whether your application
is likely to be funded and whether he or she can give you more
feedback
from the review if funding is not on the horizon. - Other items in your summary statement:
- In addition to scientific merit, the committee recommends the number of years to be funded and the amount of money it feels is appropriate.
- Reviewers also comment on items that generally do not affect your score, e.g., the appropriateness of your proposed budget, and may recommend changes.
- If you have any questions about your summary statement, contact your program officer.
 |
Should you revise?
|
 |
Are you thinking of appealing the review?
|
Prepare Your Just-in-Time Information
|
Just-in-time means you send information to NIAID when we request it. To see how we ask you for this information, go to our Sample Just-in-Time Email and How and when will I find out if I need to send information just-in-time? in the Just-in-Time questions and answers.
We request this information if your application scored roughly within the top 20 percent. Though you may not get funded, you should prepare your just-in-time information anyway. See If I receive a JIT notification, does that mean I'll get an award? in the Just-in-Time questions and answers.
Other support information is always just-in-time. We also request any of the following documentation relevant to your research that you did not include in your application:
- Human
subjects
- Assurance number.
- Certification of institutional review board approval of Research Plan.
- Certification of human subjects education.
- Animals
- Animal welfare assurance number.
- Certification of institutional animal care and use committee approval.
End-of-year warning. We may skip over your application if it comes up for funding at the very end of the fiscal year and your just-in-time submission is not ready. While we're waiting for you, we may fund other applications, and you could lose your chance of funding if we run out of money or time.
When it's due. Your institution's business official should submit other support and human subjects training information within two weeks of receiving a just-in-time notice. You don't need to sign this information because you have a signature assurance on file with your institution.
Since institutional review board and institutional animal care and use committee certifications may take more than two weeks, your business official may submit these approvals at the earliest date possible. Whether you send the certifications with your application or just-in-time, they should be sent together, not separately.
NIH prefers that your institution submit the documentation through the Commons. If you wish, you may fax it to your grants management specialist.
Read the next sections for details on the bullets above.
Find more information online:
- Just-in-Time questions and answers
- Just-in-Time SOP
 |
Will you conduct human subjects or animal research?
|
If You Have Human Subjects Documentation
|
If you're conducting human subjects research, your institution's business official needs to submit the following documents.
Wait until you get a just-in-time request if you didn't send this with your application. NIH prefers that your institution submit the documentation through the Commons. If you wish, you may fax it to your grants management specialist.
Human Subjects Assurance
Your institution needs to file a human subjects assurance with the Office for Human Research Protections (OHRP). Typically, it takes the Office for Human Research Protections (OHRP) two weeks or less to approve your FWA application. However, if OHRP spots a problem with your FWA application, it will probably take longer.
You can Search the OHRP Database, or ask your institution to see if it already has an approved assurance. Make sure the new assurance number is on file if it has changed since you submitted your application.
If you have a subaward agreement, check that the subaward organization has a human subjects assurance and IRB approval.
IRB Certification
You also need to obtain and send certification of your institutional review board's approval of your Research Plan. Unlike the assurance, this certification needs to be re-approved every year of your project.
Training Certification
If you haven't submitted it with your application, send us your certification of human subjects education letter stating that each person identified under key personnel has completed an educational program in the protection of human subjects.
Once your grant is under way, you'll need to send the training letter only for new key personnel. Use our Sample Letter to Document Training in the Protection of Human Subjects, and get detailed information on NIH's FAQ.
Find more information online:
- How to Write a Human Subjects Grant Application Tutorial
- Part 11a. Managing Your Grant.
- Human Subjects, Clinical Research
If You Have Animal Research Documentation
|
If you're working with research animals, your institution's business official needs to submit the following documents.
If you didn't send this with your application, wait until you get a just-in-time request.
Animal Welfare Assurance
Your institution needs to file an animal welfare assurance with the Office of Laboratory Animal Welfare (OLAW).
If you have a subaward agreement, make sure that the subaward organization has an animal welfare assurance and IACUC approval. If the subaward organization has an assurance but your institution doesn't, get an inter-institutional assurance. See Is Your Institution Assured by OLAW? for details.
Your institution can submit the documentation through the Commons or email the signed assurance to olawdoa@mail.nih.gov as a PDF.
IACUC Certification
You will provide certification of your institutional animal care and use committee approval and get re-approved at least every three years. For more information about getting certification of IACUC approval, go to Institutional Animal Care and Use Committee.
NIH prefers that your institution submit the certification date through the Commons. If you wish, you may fax the documentation of approval to your grants management specialist.
Find more information online:
- OLAW -- see a sample animal welfare assurance and the NIH Institutional Animal Care and Use Committee tutorial.
- How to Write an Application Involving Research Animals
- Animals in Research SOPs
- Part 11a. Managing Your Grant.
Prepare Your Other Support Submission
|
Just-in-time, you will send NIAID a list of other support -- existing support you have and support you may gain from the current application.
If you have no other support, we will need a letter stating that fact from your institution's business office.
 Our Advice: Get
Other
Support Information Ready Well Before We Make the Award
Our Advice: Get
Other
Support Information Ready Well Before We Make the Award
Your other support information shows the following:
- No other organization is supporting the research you outlined in your Research Plan -- scientific overlap.
- Your time is not committed more than 100 percent -- commitment overlap.
- You have not requested funding for items paid for by another source -- budgetary overlap.
Overlap and Bridge Awards
- Bridge awards. If you have received an R56-Bridge award from us, include it in your other support information.
- Overlap. Beware of overlap issues, which require us to reduce your award, e.g.,
if you list Howard Hughes Medical
Institute (HHMI) as other support, we will adjust your funding level since
HHMI pays 100 percent of salary and fringe benefits.
- You may submit your application simultaneously to a private foundation or federal agency outside PHS with the same application, but NIH will not fund you if you accept that award.
- You cannot send the same application to more than one PHS agency at the same time with few exceptions -- contact your business office for details.
- If you are applying for more than one grant to a PHS agency, point out in your application and in your cover letter that there's no overlap, and make sure the Specific Aims differ.
Find more information online:
- You Will Send Some Materials Just-in-Time
- Other Support format table in the Grant Application Guide
- Consultants or Collaborators -- How They Differ
- Where to Add Consortium and Contractual Information
If You Have Consortium or Contractual Agreements
If you have consortium or contractual agreements, send through the Commons just-in-time.
 |
|
| The next part of the NIH Grant Cycle: Application to Renewal is Part 9. Second-Level Review. |
Help us improve our outreach to you by emailing deaweb@niaid.nih.gov.
Look It Up
See the Glossary for more terms. |