Many published studies have demonstrated
statistically significant associations
between ambient air fine particulate
matter (aerodynamic diameter ≤ 2.5 µm,
PM2.5) mass concentrations
and short-term changes in heart rate
(HR) and/or HR variability (HRV) in humans
and laboratory animals (U.S. Environmental
Protection Agency 2004). However, interpretation
of these findings is complicated in that
the effects may go in either direction
and are observed on some days and not
on others. These inconsistencies may
be due to the limitations of PM2.5 mass
as an index of exposure to the biologically
active components of the ambient PM2.5.
It is well known that the composition
of ambient air PM2.5 has considerable
temporal and spatial variability. Studies
in human volunteers and laboratory animals
have been limited in their power to identify
the causal components because they require
both the long-term collection of electrocardiographic
(ECG) data and simultaneous availability
of data on tracers or factors associated
with PM2.5 composition.
In a recent study of the effects of
subchronic (5-6 months of daily
6 hr) exposures of normal (C57) mice
and a mouse model of atherosclerosis
(apolipoprotein deficient, ApoE-/-,
mice) to fine concentrated ambient particles
(CAPs) in Tuxedo, New York, USA, at an
average concentration of 113 µg/m3,
we generated the kinds and amounts of
data needed to address the issue raised
in this article. The results of the overall
subchronic study design and the results
obtained for associations of PM2.5 mass
concentration with progressive changes
in HR and HRV and the changes in atherosclerotic
plaque, gene expression, and brain cell
distribution at the end of the study
are described in a series of articles
(Chen and Hwang 2005; Chen and Nadziejko
2005; Gunnison and Chen 2005; Hwang et
al. 2005; Lippmann et al. 2005a 2005b;
Maciejczyk et al. 2005; Veronesi et al.
2005). In another article (Maciejczyk
and Chen 2005) describing the parallel
study that went on simultaneously with
the inhalation study, we exposed BEAS-2B
cells (an airway epithelial cell line) in
vitro to CAPs and reported that nuclear
factor-kappa B (NF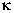 B)
expression was most closely associated
with the residual oil (RO) component,
which was, on average, 1.4% of the PM2.5 mass.
B)
expression was most closely associated
with the residual oil (RO) component,
which was, on average, 1.4% of the PM2.5 mass.
For this article, we used the 5 months
of daily 6-hr source apportionments described
in Maciejczyk and Chen (2005), the continuous
HR data for exposure (weekday) days provided
in Hwang et al. (2005), and the corresponding
HRV data given in Chen and Hwang (2005)
to determine the source-related PM2.5 components’ short-term
associations with HR and HRV.
The methods used to generate the factors
associated with specific major PM source
categories were described by Maciejczyk
and Chen (2005). Briefly, fine CAPs were
collected from a rural area upwind of
New York City for the 0900- to 1500-hr
period on weekdays only, March through
September 2003. Chemical composition
data for CAPs were modeled using factor
analysis with varimax orthogonal rotation
to determine four particle source categories
contributing significant amount of mass
to CAPs at Sterling Forest (Tuxedo, New
York). These source categories are regional
secondary sulfate (SS) characterized
by high sulfur, silicon, and organic
carbon; resuspended soil (RS) characterized
by high concentrations of calcium, iron,
aluminum, and silicon; RO-fired power
plants emissions of the Eastern United
States identified by presence of vanadium,
nickel, and selenium; and motor vehicle
(MV) traffic and unknown other sources.
To estimate the mass contributions of
each individual source category, the
CAP mass concentration was regressed
against the factor scores. Using the
method developed by Thurston and Spengler
(1985), we determined that regional sulfate
was the largest contributor to average
mass (56.1%), followed by soil (11.7%).
The RO combustion accounted for 1.4%,
and the MV traffic and other sources
category contributed 30.9%.
The methods used to process the voluminous
HR and HRV data for the same period were
described by Hwang et al. (2005) and
Chen and Hwang (2005). Briefly, they
used their recently developed nonparametric
method (Nadziejko et al. 2004) to estimate
the daily time periods that mean HR differed
significantly between the CAPs and the
air sham-exposed groups. CAP exposure
most affected HR between 0130 and 0430
hr. With the response variables being
the average HR, they adopted a two-stage
modeling approach to obtain the estimates
of chronic and acute effects on the changes
of this variable. In the first stage,
a time-varying model estimated daily
crude effects. In the second stage the
true mean of the estimated crude effects
was modeled with a polynomial function
of time for chronic effects, a linear
term of daily CAP exposure concentrations
for acute effects, and a random component
for unknown noise. A Bayesian framework
combined these two stages.
For the analyses of HRV, the times
in milliseconds of occurrence of two
consecutive R waves in the ECG waveform
(RR) were calculated on a beat-to-beat
basis. Because of limitation in data
storage capacity, the RR intervals were
recorded consecutively for 5 sec in every
15-min interval for all mice during 10-27
April 2003, and for ApoE-/- mice
in the control group during 22 April
through 20 July 2003. The rest of recordings
were taken consecutively about 10 sec
in every 5-min interval for the mice.
There are about 34-64 and 100 RR
intervals recorded in 15- and 5-min intervals,
respectively. For the analysis, we decided
to work on fluctuations of RR intervals
on an every 15-min basis. To match the
data in the 15-min recordings, we used
only the first 60 RR intervals in the
last of 3 consecutive 5-min intervals.
The two HRV indices that we used were
the standard deviations of the RR intervals
(SDNNs) and the square root of the mean
squared differences (RMSSD) of successive
RR intervals in 5 sec. The nonparametric
method identified the 0000- to 0500-hr
period during which the two groups had
the largest HRV differences within each
day. To match the HR analyses of effects
with the HRV changes, we used the same
period (0130-0430 hr) for calculating
mean log SDNN and log RMSSD to represent
daily HRV responses for this period for
each mouse. In the analysis of effects
on HR, we also calculated daily responses
for the 1100- to 1300-hr period during
exposure for examining acute effects.
However, because the number of normal
RR intervals recorded during the exposure
period was small because of interference
from the perforated metal chamber, we
instead used the 1600- to 1800-hr interval
after exposure as an alternate for calculating
daily HRV response. Daily changes in
HR during this period, which were not
reported in the previous study, were
also calculated for this analysis.
To examine whether variations of concentrations
in major sources are correlated with
short-term changes of cardiac functions
in exposed mice, we adopted the following
approach:
Let Xijkd be the
average cardiac function measurement
for mouse j in the ith
group at a given period on the dth
day of the kth week, where
i = 0 (control), 1 (exposure)
j = { 1~9
when i =
0
1~10
when i =
1
k = 1 (Saturday, 12 April, through
Friday, 18 April), 2 (Saturday, 19 April,
through Friday, 25 April), . . . , 22
(Saturday, 6 September, through Wednesday,
10 September)
d = 1 (Saturday), 2 (Sunday),
3 (Monday), 4 (Tuesday), 5 (Wednesday),
6 (Thursday), 7 (Friday).

Figure 1. Daily average measurements
of HR (bpm) for CAP-exposed and air
sham-exposed (control) ApoE-/- mice
during the daily exposures (1100-1300
hr). |

Figure 2. The difference (D) in HR
(bpm) between CAP-exposed and air
sham-exposed (control) ApoE-/- mice
during the daily exposures (1100-1300
hr). |
Table
1
 |
Table
2
 |
Table
3
 |
Table
4
 |
Table
5
 |
Table
6
 |
We have seen that daily cardiac function
measurements changed over the 5 months.
Such changes may be caused by the cumulative
effects of aging, exposure, and other
unknown environmental factors. To examine
the association between exposed level
and acute cardiac function change on
exposure days, we generated baseline
adjusted measurements for each mouse
on the exposure days by subtracting averaged
measurement on the previous weekend from
each measurement on weekdays. Presumably,
the daily series of these baseline-adjusted
measurements
Yijkd =
Xijkd - (
Xijk1 +
Xijk2)/2
will have little cumulative effect. To
see whether the idea worked or not, we
explored the data. Figure 1 shows two
series of daily averaged baseline adjusted
measurements of HR at the 1100- to 1300-hr
period for mice in the control and exposure
groups. The exposure chamber effects
reduced HR in both groups, which also
corresponded to the quiescent period
of mouse circadian rhythm during the
daytime. The two series also share the
same quadratic shape. Although it is
not clear why this has happened, some
common factors have strong effects on
measurements of mice in both control
and exposure groups. Instead of searching
for a smooth curve for modeling the pattern
caused by common factors, we can simply
use the baseline-adjusted measurements
of the nine mice in the control group
to calculate an average for each exposure
day. That is the darker curve plotted
in Figure 1. If there is no exposure
effect, the darker curve and lighter
curve of averaged measurements for mice
in the exposure group will not differ.
In fact, the difference between two curves
shown in Figure 2 indicates that CAP
exposure had the effect of reducing HR.
The difference series in the plot also
show no trend over the 5 months, indicating
that cumulative effects have been removed.
Hence, we may construct a model to fit
the baseline-adjusted measurements for
examining whether the short-term cardiac
function changes are related to exposure
levels of the identified source factors
F1, F2, F3, and F4. A linear model is
given by
Yijkd= µ
kd+ β
1• (
F4)
kd•
I(
i
= 1)
+ β
2• (
F3)
kd•
I(
i
= 1)
+ β
3 • (
F2)
kd•
I(
i =
1)
+ β
4• (
F1)
kd•
I(
i
= 1) +
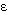 ijkd
ijkd´,
[1]
where 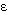 ijkd is
an autoregressive process of order one.
If the estimate of βh differs
significantly from zero, we may claim
that the hth source factor is
associated with the acute changes of
HR and HRV.
ijkd is
an autoregressive process of order one.
If the estimate of βh differs
significantly from zero, we may claim
that the hth source factor is
associated with the acute changes of
HR and HRV.
Associations between sources
and short-term HR changes. Using
the source apportionment factors
from Maciejczyk and Chen (2005),
we have the following four source
classes: SS, RS, RO, and MV. There
were no significant associations
between these four source categories
and HR in the C57 normal mice at
any of the three intervals. However,
as shown in Table 1, there were highly
significant associations between
PM2.5 and the RS source
factor and decreases in HR for the
ApoE-/- mice
during the daily CAP exposures but
no associations with the other source
factors. By contrast, Tables 2 and
3 indicate that there was no residual
association of HR with PM2.5 or
the RS factor later in the afternoon
or late that night.
In the afternoon, there was a significant
association between decreases in HR and
the SS factor for the ApoE-/- mice
that had not been present during exposure
and did not persist into the nighttime
period. It is also of some interest that
the MV traffic and other source category
was not significantly associated with
HR during any of the three time periods.
For the C57 mice, there were no significant
associations of HR with PM2.5 or
any of its component source classes during
any of the three daily time periods.
Associations between sources
and short-term HRV changes. It
is unfortunate that there was too
much signal noise during the exposures
to permit reliable analyses of HRV
changes during the hours of CAP exposure.
We therefore cannot tell whether
the transient effect of PM2.5 or
its RS source component on HR was
also present for HRV. For C57 mice,
the only significant association
was between the MV and other source
factor and a decline in RMSSD during
the afternoons after the exposures
(p = 0.00; data not shown).
For the ApoE-/- mice
(Table 4), there were very strong
associations of HRV with the RO source
factor in the afternoon. These decreases
in HRV did not persist at night (Table
5) and had not been seen for HR at
any time period. Finally, there were
strong associations between HRV during
the nighttime hours and both the
SS source category and the RS source
category that were not seen for HR
at the other intervals, or for HRV
at the other time periods. However,
it must be noted that although the
SS source factor was associated with
decreased HRV, the RS source category
was associated with an increase in
HRV. For PM2.5, there
was a significant (p = 0.03)
decrease in RMSSD and a nearly significant
(p = 0.07) decrease in SDNN
for the 0130- to 0430-hr interval
but no such an association during
the 1600- to 1800-hr period.
Interpretation of the various significant
(p < 0.05) associations between
source factors and the HR and HRV variables
in CAP-exposed mice at this time would
be speculative at best, especially because
three of the source factors showed some
association at one interval or another,
and the fourth (MV traffic and other
category) showed a strong association
(p = 0.00) with RMSSD in the afternoon
after exposure in the C57 mice. The strongest
associations for the ApoE-/- mice
are summarized in Table 6.
For the evaluation of the changes on
HR and HRV in the last column of Table
6, we have calculated the changes in
the measured parameters over the interquartile
range of concentrations as is commonly
done in epidemiology. For HR, the changes
are for exposures at the third quartile
to the first quartile of the measured
concentrations. The results show about
3-4 beats/min (bpm) changes. For
HRV, the interquartile change is the
ratio of RMSSDs between the third quartile
and first quartile of the concentrations.
The results show about 2-6% changes.
These are relatively small changes, but
they may have played some role in the
progressive changes in HR that we observed
during the course of the 5 months of
exposure that were described by Hwang
et al. (2005), and the changes in HRV
that were reported by Chen and Hwang
(2005).
It is also interesting that the reduction
in HR during the daily exposures associated
with PM2.5 (-4.1 bpm)
may have been due entirely to the influence
of the RS factor (-4.5 bpm) and
that there was an increase in HR (+2.6
bpm) in the afternoons after the exposures
in the same source factor. This appears
to have been compensated by the decrease
in HR in the afternoon after the exposures
(-2.5 bpm) associated with the
SS factor. Such a compensation would
be consistent with the lack of any association
of HR with PM2.5 in this
interval.
The RO combustion factor, which did
not have any significant association
with HR, appears to have had the effect
of increasing RMSSD by 6.2% during the
afternoons after the exposure but not
at the other intervals. The other observed
statistically significant changes in
RMSSD were associated with opposite effects
during the late night period by the RS
and SS source components, with the SS
factor perhaps accounting for the significant
association in the same direction for
the association of RMSSD with PM2.5 during
the same period.
It is also of interest that the effects
reported here for HR and HRV were occurring
at relatively low concentrations of outdoor
PM2.5 and its component source-related
factors. The average PM2.5 CAPs
during the 6-hr exposures was only 113 µg/m3.
Thus, the 24-hr average exposures were
only 28.3 µg/m3 because
the mice were breathing air that was
filtered of the outdoor air components
during the balance of the day. Outdoor
PM2.5 does not have much diurnal
variation, and it infiltrates indoors
with a high degree of penetration. People
are therefore exposed to concentrations
of PM2.5 of ambient origin
at near ambient concentrations for 24-hr
each day. If indeed the ApoE-/- mouse
is a good model for people with atherosclerosis,
and if the HR and HRV responses to CAPs
in these mice seen in this study are
relevant to them, then such responses
may be occurring in this human subpopulation
at current ambient levels on many days
each year.
There have been no previous reports
that examined such responses at various
periods during and after daily exposures.
The only report of different ambient
air PM source categories having different
lagged effects was that of the PM Source
Apportionment Workshop, in which human
mortality effects were associated with
different days of lag (Thurston et al.
2005).
Although there have been no previous
reports of cardiac function effects that
go in opposite directions after low-level
environmental exposures, there have been
such examples for other physiologic responses.
In previous work in this laboratory,
we reported in both humans (Leikauf et
al. 1981) and rabbits (Schlesinger 1985)
that short-term inhalation of a low concentration
of submicrometer sulfuric acid aerosol
increased the rate of mucociliary particle
clearance from tracheobronchial airways,
whereas a higher concentration (1 mg/m3)
retarded such clearance. Similarly, the
inhalation of the fresh smoke from two
cigarettes accelerated tracheobronchial
particle clearance in both humans and
donkeys, whereas the smoke from 10 or
more cigarettes slowed the particle clearance
in donkeys (Lippmann et al. 1982). In
another study in this laboratory, Schlesinger
(1989) examined the effect of 14 days
of sulfuric acid inhalation on particle
clearance from the pulmonary region of
rabbit lungs and found that low-level
exposures accelerated such macrophage-mediated
clearance, whereas higher levels of exposure
retarded the clearance. In addition,
subchronic inhalation exposures to both
cigarette smoke and sulfuric acid produced
persistent changes in particle clearance
(Lippmann et al. 1987).
The fact that three different source
factors showed some indication of a strong
association with either HR or HRV in
ApoE-/- mice in
this study, with the SS source component
having an effect in the opposite direction
to that of the RS source component, illustrates
the complexity facing researchers when
designing studies to identify the causal
factors for the PM-associated adverse
health effects reported in the epidemiologic
literature. It may well be that most,
if not all, PM source categories have
some, if various, effects on cardiac
physiology, with various lag structures,
and that some components mitigate the
effects produced by other components.
Also, we do not know at this time about
the short-term effects, and their temporality,
of inhaled PM2.5 on other
organ systems. However, if most of the
major components of PM2.5 produce
some short-term biologic responses, then
the commonly used integral measure of
PM2.5 mass concentration,
that is, 24-hr average PM2.5,
may be serving as a reasonable integrating
index for at least some of the short-term
health risks. In any case, the results
reported in this article provide us and
others with additional factors to consider
in the planning of our future laboratory
and field studies of PM health effects.
As noted above, Maciejczyk and Chen
(2005) reported that in vitro NF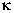 B
expression of BEAS-2B cells exposed to
CAPs collected during the daily 6 hr in
vivo exposures was significantly
increased in association with the RO,
but not with the other source categories
of the CAPs. The NF
B
expression of BEAS-2B cells exposed to
CAPs collected during the daily 6 hr in
vivo exposures was significantly
increased in association with the RO,
but not with the other source categories
of the CAPs. The NF B
expression is an index of cellular oxidative
stress and the release from the cells
of mediators affecting systemic inflammation.
This mechanism for biologic response
is consistent with short lag times between
respiratory tract particle deposition
and cardiac function changes. However,
because the NF
B
expression is an index of cellular oxidative
stress and the release from the cells
of mediators affecting systemic inflammation.
This mechanism for biologic response
is consistent with short lag times between
respiratory tract particle deposition
and cardiac function changes. However,
because the NF B
index of biologic response to CAP exposure
provides no within-day temporality, it
is not possible to make a direct comparison
with the lagged HR and HRV responses
reported in this article. The different
lag structures of the responses reported
in this article may be related to the
solubility of the biologically active
components in each source category.
B
index of biologic response to CAP exposure
provides no within-day temporality, it
is not possible to make a direct comparison
with the lagged HR and HRV responses
reported in this article. The different
lag structures of the responses reported
in this article may be related to the
solubility of the biologically active
components in each source category.
We plan to pursue the issues raised
by the results reported here in our future
subchronic exposure studies in mice.
In terms of comparable investigations
in humans, a study would need access
to a population that is being continuously
monitored for cardiac function as well
as time-resolved PM2.5 compositional
data. The only study we are aware of
to date looking for cardiac responses
to ambient air PM was by Sullivan et
al. (2005), in which they examined the
relation between PM2.5 exposure
(measured by nephthelometry) and the
number of hours preceding the onset of
myocardial infarction (MI). They found
no significant associations between MI
and the nephthelometry data. It is possible
that nephthelometry measurements may
not be representative of the active components
of ambient PM mix or that the nephthelometry
measurements correlate with outcomes
other than MI.
The availability of data on HR and
HRV over a 5-month period during subchronic
exposures of mice to the regional anthropogenic
CAPs at New York University’s Sterling
Forest laboratory in Tuxedo, New York,
and during the afternoon and nighttime
periods after the daily exposures, as
well as elemental composition data for
each day’s exposure, enabled us
to examine daily source apportionments
of the major source categories during
the exposures and their association with
HR and HRV during each of the three time
periods. The RS component was strongly
associated with a transient decrease
in HR during exposure, comparable with
that of the whole PM2.5. The
SS component was strongly associated
with a transient HR decrease in the afternoon
after the day’s exposure. The RO
component was strongly associated with
increases in HRV in the afternoon after
the day’s exposure. The SS and
RS components were strongly associated
with HRV in the nighttime period, with
decreased HRV for the SS component and
increased HRV for the RS component. These
effects were occurring after exposures
at daily average PM2.5 concentrations
occurring frequently in the United States
and may be relevant to the subpopulation
with atherosclerotic disease.
The biologic bases for these various
associations and their temporal lags
are not known at this time but may relate
to the differential solubilities of the
PM components at the respiratory epithelia
and their access to cells that release
mediators that reach the cardiovascular
system. Further research that can elucidate
the underlying processes is clearly needed.

