Endotoxin is biologically active lipopolysaccharide,
a component of the outer cell membrane of gram-negative
bacteria. Endotoxin has potent proinflammatory effects
that have been well characterized at high doses among
adults but are less well understood in home settings
and among infants and children (Liu 2002; Reed and Milton
2001). Data suggesting that early-life infections and
exposure to a farming lifestyle decrease the risk of
childhood allergic disease have led to the hypothesis
that early-life household endotoxin exposure may be protective
against asthma and allergy (Braun-Fahrlander et al. 2002;
Celedon et al. 2002; Martinez and Holt 1999; von Mutius
et al. 2000). To test this hypothesis, epidemiologists
have begun to measure endotoxin levels in house dust
samples in population-based studies (Braun-Fahrlander
et al. 2002; Gehring et al. 2001a, 2001b; Park et al.
2001; Rizzo et al. 1997; von Mutius et al. 2000).
In principle, the goal of endotoxin assessment for
use in studies of endotoxin and chronic disease onset
is to estimate subjects’ exposure, appropriately
integrated over time and space. However, the appropriate
sampling strategy is not well defined, and practical
limitations often dictate actual sampling protocols.
Commonly, endotoxin sampling would include collection
of dust at only one point in time from one or at most
a few rooms. Repeated endotoxin sampling within the time
period of interest is seldom attempted. For optimal classification
of chronic exposure, however, the relationship between
point exposure measurements and temporal and spatial
averages is of particular relevance. Nevertheless, few
data are available on the relationship of endotoxin measured
at a specific time and place in the home to endotoxin
measured in other rooms within the home and at other
times.
In the setting of linear regression, the within- to
between-subject variance ratio is an estimate of the
signal-to-noise ratio and has been used to assess the
misclassification bias that occurs when using an imperfectly
measured or surrogate exposure. A large within- to between-subject
variance ratio indicates that a single exposure sample
will provide a less precise estimate of chronic exposure.
Park et al. (2000) applied a variance components model
to estimate within- and between-home variances in endotoxin
measurements in monthly samples over 12 months in a convenience
sample of 20 Boston homes. Within-home variations in
endotoxin levels were greater than between-home variations,
except for endotoxin sampled in bed dust of adult participants.
If generally true, the findings of Park et al. (2000)
indicate that comparisons of exposure between homes based
on a single assessment of endotoxin levels in sites other
than the bed are not particularly useful--even for
assessing average exposure over 1 year. Sites other than
the bed may be relevant for endotoxin exposure, particularly
for infants and toddlers in the United States, who often
have relatively little dust in their plastic-covered
bed mattresses and who spend a great deal of time in
other rooms and crawling on the floor. Two recent reports
of variability within and between homes in larger epidemiologic
studies in Germany suggest that over 1 year, single measurements
may be sufficient to distinguish exposure between homes,
but that more measurements are needed to assess long-term
average exposure (Heinrich et al. 2003; Topp et al. 2003).
In this report, we used a variance components analysis
to reexamine the utility of endotoxin measurements in
dust collected from different rooms in distinguishing
average exposure during the first months to 1 year of
life using a sample of 470 homes of children in Boston,
Massachusetts. We assessed the correlation of endotoxin
sampled in one room with levels in other rooms, and the
correlation of a single endotoxin measurement with measurements
of endotoxin in the same room 5-11 months later.
We estimated room-specific within- to between-home variance
ratios and explored the implications of these variance
estimates for epidemiologic studies of dust endotoxin
and health outcomes.
Cohort. The Epidemiology of Home Allergens
and Asthma study is a longitudinal birth cohort study
of environmental predictors of development of allergy
and asthma among children born to a parent or parents
with a history of allergy and/or asthma (Gold et al.
1999). The study is investigating the relationship between
indoor allergen exposure and the development of allergy
and asthma in early childhood. Between September 1994
and June 1996, women who had given birth at the Brigham
and Women’s Hospital in Boston were asked if she
or the baby’s father had a history of allergy,
hay fever, or asthma. Women answering affirmatively were
asked to complete a screening questionnaire. Inclusion
criteria included history of allergy, hay fever, and/or
asthma in at least one parent, maternal age ≥ 18
years, English or Spanish speaking, residence in the
greater Boston area, and no plans to move in the next
year. Infants were excluded for premature birth (< 36
weeks), birth with major congenital or teratologic abnormalities,
or admission to the neonatal intensive care unit. Of
the 1,405 women who completed the screening questionnaire,
499 mothers (505 children) met inclusion and exclusion
criteria and agreed to participate.
Dust collection and endotoxin assessment. Within
the first 3 months of the index child’s birth,
an initial exposure assessment was conducted on the 499
homes of participants (Chew et al. 1998). An exposure
assessment was conducted approximately 6 months later
in a subset of homes. House dust was collected on a 19  90
mm cellulose extraction thimble using a modified Eureka
Mighty-Mite vacuum cleaner (Eureka Co., Bloomington,
IL). Separate dust samples were collected from the kitchen
floor, family room, and the floor of the infant’s
bedroom. In the kitchen, the floor under cabinets, around
the refrigerator, and under the sink were vacuumed for
5 min. In the family room, the seat cushion, arms, and
back of the chair most often occupied by the primary
caregiver were vacuumed for 2.5 min. Two square meters
of the floor surrounding this chair was also vacuumed
for 2.5 min. In the bedroom, 2 m2 of floor
surrounding the baby’s crib was vacuumed for 5
min. Collected dust was immediately placed in airtight
bags. Initial sampling of dust to be used for endotoxin
analysis was conducted between November 1994 and October
1996. The second dust sampling was conducted in a subset
of homes between June 1995 and October 1996. Homes were
selected for repeat sampling if the initial sampling
was conducted during winter months. In the laboratory,
dust samples were sifted using a 425-µm mesh sieve
to remove large debris (e.g., breakfast cereal) and provide
a more uniformly mixed, fine dust sample for partition
into aliquots for several assays. The fine dust was then
weighed and aliquoted for future analysis. Dust samples
were stored desiccated at -20°C until extraction.
Samples were analyzed for allergen and fungi and additionally
analyzed for endotoxin only if there was > 200 mg
of dust in the sieved sample. Endotoxin levels were not
determined for 29 (6%) of the 499 participating homes.
Up to six samples (three rooms with up to two samples)
were possible per home. In the 470 homes with at least
one endotoxin sample, we collected a mean of 2.7 and
median of three samples per home.
90
mm cellulose extraction thimble using a modified Eureka
Mighty-Mite vacuum cleaner (Eureka Co., Bloomington,
IL). Separate dust samples were collected from the kitchen
floor, family room, and the floor of the infant’s
bedroom. In the kitchen, the floor under cabinets, around
the refrigerator, and under the sink were vacuumed for
5 min. In the family room, the seat cushion, arms, and
back of the chair most often occupied by the primary
caregiver were vacuumed for 2.5 min. Two square meters
of the floor surrounding this chair was also vacuumed
for 2.5 min. In the bedroom, 2 m2 of floor
surrounding the baby’s crib was vacuumed for 5
min. Collected dust was immediately placed in airtight
bags. Initial sampling of dust to be used for endotoxin
analysis was conducted between November 1994 and October
1996. The second dust sampling was conducted in a subset
of homes between June 1995 and October 1996. Homes were
selected for repeat sampling if the initial sampling
was conducted during winter months. In the laboratory,
dust samples were sifted using a 425-µm mesh sieve
to remove large debris (e.g., breakfast cereal) and provide
a more uniformly mixed, fine dust sample for partition
into aliquots for several assays. The fine dust was then
weighed and aliquoted for future analysis. Dust samples
were stored desiccated at -20°C until extraction.
Samples were analyzed for allergen and fungi and additionally
analyzed for endotoxin only if there was > 200 mg
of dust in the sieved sample. Endotoxin levels were not
determined for 29 (6%) of the 499 participating homes.
Up to six samples (three rooms with up to two samples)
were possible per home. In the 470 homes with at least
one endotoxin sample, we collected a mean of 2.7 and
median of three samples per home.
The endotoxin activity of dust samples was determined
with the kinetic Limulus assay with the resistant-parallel-line
estimation (KLARE) method (Milton et al. 1992, 1997). Limulus amebocyte
lysate was supplied by BioWhittaker (Walkersville, MD),
and control standard endotoxin was obtained from Associates
of Cape Cod (East Falmouth, MA). Endotoxin measurements
were adjusted for lot-to-lot variation in Limulus amebocyte
lysate sensitivity to house dust endotoxin [lot 6L016C
used for assay of 42% of the samples was used as the
standard lot, and nine additional lots each used for
2-11% of samples were adjusted using data from
previously described lot-to-lot comparison assays (Milton
et al. 1997)]. Control standard potency was determined
for each combination of lysate and standard with reference
to the reference standard endotoxins EC5 or EC6 [U.S.
Pharmacopoeia, Inc., Rockville, MD; 1 ng EC5 and EC6
= 10 endotoxin units (EU)] available at the time the
assays were performed, by simultaneous assay of the control
with the reference or with a control traceable to assay
with the reference. Results are reported as EU per milligram
of dust sampled. The median coefficient of variation
of the assay of house dust samples, 23%, was previously
reported (Milton et al. 1997). None of the samples was
below the limit of detection.
Statistical analysis. We used SAS
version 8.2 for all statistical analyses (SAS Institute
Inc., Cary, NC) and assessed the normality of endotoxin
distributions using the Shapiro-Wilk normality test.
The dust endotoxin data were log-transformed to normalize
the distribution of residuals in the mixed-effects models.
We compared means in a mixed-effects model to account
for correlation of samples within the same home. The
correlation of endotoxin measured in dust sampled from
different rooms in a home was assessed using Pearson
correlation coefficients (with room-specific averages
for rooms with replicate endotoxin observations) and
using a mixed-effects model.
We fit a mixed-effects model of log endotoxin levels
as a function of room and season adjusting for the correlation
of repeated measurements within the same home. Inclusion
of a random room effect within homes and declaration
of a repeated-measures structure allowed us to characterize
variation within and between homes and over time (Hamlett
et al. 2003; Lyles et al. 1997; Rappaport 1991; Rappaport
et al. 1995; Symanski et al. 1996). We obtained parameter
estimates using restricted maximum likelihood (Diggle
2002). More precisely, the general mixed-effects model
is described by the expression
where Yij is the jth repeated
observation of log-transformed endotoxin for home i.
The terms

are fixed covariates associated with the jth
repeated measure on the ith home. The residual
variance, 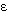 ij,
is modeled to include an appropriate correlation structure
between endotoxin observations. For each room, the model
estimates within-home and between-home variance,
ij,
is modeled to include an appropriate correlation structure
between endotoxin observations. For each room, the model
estimates within-home and between-home variance,  2w and
2w and  2b,
respectively. We then calculated within- to between-home
variance ratios and intrahome correlation coefficients
for endotoxin sampled from the floor of the subject’s
bedroom, the family room, and the kitchen dust samples.
The within- to between-home variance ratio characterizes
the degree to which a single observation of endotoxin
is representative of chronic exposure. The intrahome
correlation coefficient characterizes the reproducibility
(stability) of repeated endotoxin measurements over time
(Rosner 1995). To estimate 95% confidence intervals (CIs),
standard errors of within- to between-home variance ratios,
and intrahome correlation coefficients were estimated
using the delta method, using asymptotic variance and
covariance estimates of the room-specific within-home
and between-home variances estimated by the mixed effects
model.
2b,
respectively. We then calculated within- to between-home
variance ratios and intrahome correlation coefficients
for endotoxin sampled from the floor of the subject’s
bedroom, the family room, and the kitchen dust samples.
The within- to between-home variance ratio characterizes
the degree to which a single observation of endotoxin
is representative of chronic exposure. The intrahome
correlation coefficient characterizes the reproducibility
(stability) of repeated endotoxin measurements over time
(Rosner 1995). To estimate 95% confidence intervals (CIs),
standard errors of within- to between-home variance ratios,
and intrahome correlation coefficients were estimated
using the delta method, using asymptotic variance and
covariance estimates of the room-specific within-home
and between-home variances estimated by the mixed effects
model.
Definition of categorical variables. The
season of dust sampling was categorized as winter (November
through March), spring (April and May), summer (June
through August), or fall (September and October), to
match Boston’s climate (Chew et al. 1999). Presence
of a pet dog was categorized as no dog versus one or
more dogs. The type of house occupied by the family was
grouped into a) single-family or two-family dwellings
or b) homes in apartment buildings with three
or more units.
Table
1
 |
Table 2
 |
Table 3
 |
Table 4
 |
Table 5
 |
Table 6
 |
A total of 1,287 endotoxin measurements were taken
from 470 of the 499 participating homes. The initial
home assessment included 320 bedroom, 401 family room,
and 245 kitchen dust samples that were assayed for endotoxin
activity. In the follow-up home assessment, endotoxin
was measured in 102 bedroom, 147 family room, and 72
kitchen dust samples. In all, 82 (17%) homes had only
one endotoxin measurement, 147 (31%) had two measurements,
127 (27%) had three measurements, 61 (13%) had four measurements,
32 (7%) had five measurements, and 21 (4%) had all six
measurements. Repeated endotoxin activity measurements
were available for 90 bedrooms, 125 family rooms, and
55 kitchens (180, 250, and 110 samples, respectively;
Table 1).
Distribution of endotoxin levels in house dust. Endotoxin
ranged from 2 to 1,945 EU/mg of dust, with a geometric
mean (GM) of 82 EU/mg of dust, a median of 81 EU/mg,
and a geometric standard deviation of 2.1 (Table 2).
Endotoxin was lowest in the bedroom floor samples, intermediate
in family room samples, and highest in the kitchen floor
samples, and similar in rooms with and without repeat
samples. Endotoxin levels were highest during the summer
and lowest in winter.
Adjusting for season, home assessment (initial or follow-up),
presence of a dog in the home, housing type, and the
correlation between observations made in the same home,
we found that GM endotoxin varied significantly according
to the room in which dust was sampled (p < 0.001)
(Table 3). Endotoxin levels also varied by season in
this model, with highest levels in the summer compared
with fall (p = 0.002), winter (p < 0.001),
and spring (p = 0.054). Endotoxin did not differ
significantly between the initial and repeated samples
(p = 0.494) in the multivariate model, after adjusting
for season. As previously reported, GM endotoxin was
higher (p < 0.001) in the 75 (16%) homes with
dogs relative to homes without dogs, and higher in the
356 (76%) one- and two-family homes compared with homes
in multiunit apartment buildings (p = 0.004).
Correlation of endotoxin levels in house dust. Cross-sectional
correlations between room-specific endotoxin levels were
low to moderate (Table 4). The mixed-model correlations
were similar to the Pearson correlation coefficients.
Relative to the cross-sectional comparison of endotoxin
from different rooms within homes, repeated room-specific
endotoxin levels (5-11 months apart) were more
highly correlated for bedroom floor (r = 0.65;
95% CI, 0.56-0.75), kitchen floor (r = 0.65;
95% CI, 0.53-0.76), and family room (r =
0.54; 95% CI, 0.44-0.63). Thus, the temporal correlation
of endotoxin levels measured over the 5- to 11-month
time was greater than the spatial correlation in those
measurements (Table 4). Correlation coefficients estimated
without adjustment for season were consistently lower,
relative to those estimated while adjusting for fixed
effects of season (Table 5).
We divided measurements of endotoxin sampled at two
different times into quartiles of the time interval between
samples. We did not find any consistent decrease in the
correlations moving from shorter to longer time spans
between sampling. In fact, for samples taken from the
baby’s bedroom floor, the correlation between endotoxin
sampled at two points in time increased with increasing
time between sampling. However, the sample sizes for
each time interval are small and the correlations are
correspondingly less stable.
Endotoxin variance components: variation within
and between homes. We found that season-adjusted
within-home variability was lowest for endotoxin
in dust sampled from the baby’s bedroom floor,
higher in family room samples, and highest for endotoxin
in kitchen floor dust (Table 5). Season-adjusted
between-home variability was lowest in dust sampled
from the family room, higher for bedroom samples,
and again highest for kitchen dust endotoxin (Table
5). In models adjusting for season, the within-home
variance was less than the between-home variance
for all three rooms, suggesting that factors affecting
endotoxin levels within a home over time influence
the total variability less than factors contributing
to home-to-home differences in endotoxin. Within-
to between-home variance ratios were lowest for bedroom
floor and kitchen floor endotoxin and somewhat higher
for family room samples, an observation explained
by the comparatively small between-home variation
in family room endotoxin (Table 5). These findings
suggest that the determinants of endotoxin levels
over time and from home-to-home are room specific.
We divided homes into those without dogs and those
with at least one dog (Table 6), but found no consistent
differences in variance components by presence of a dog.
Unexpectedly, we found that between-home variation in
endotoxin was uniformly higher in homes without dogs
relative to homes with dogs. However, this did not result
in correspondingly uniform changes in the correlation
of endotoxin levels over time.
A similar subgroup analysis was conducted for housing
type (Table 6). We did not find consistent differences
in the variance components comparing single- or two-family
houses with apartments in buildings with three or more
units. Notably, for kitchen endotoxin in multiunit buildings,
we observed a within-home variance 3.7 times that of
the between-home variance, whereas the within-home variance
was smaller than the between-home variance for the other
rooms sampled.
We assessed the distributions of, correlations between,
and components of variation in endotoxin levels in dust
sampled from the homes of subjects participating in an
ongoing birth cohort study. In the homes studied, dust
endotoxin levels were correlated over 5-11 months
(range of intrahome correlations, 0.54-0.65), and
slightly less correlated across rooms within homes (range
of cross-sectional, room-to-room correlations, 0.30-0.42).
Within-home to between-home variance ratios were below
one for all samples: 0.53 and 0.54 for bedroom and kitchen
dust endotoxin, respectively, and 0.85 for family room
endotoxin. Thus, single endotoxin measurements are a
reasonable proxy for average exposure during the first
few months to 1 year of life and capable of distinguishing
among children in metropolitan Boston with regard to
endotoxin exposure in early life.
Park et al. (2000) analyzed the variance components
of endotoxin in dust collected in a one-year longitudinal
study of a convenience sample of 20 homes of students,
faculty, and university staff in Boston. This report
builds on that work by Park and colleagues by characterizing
variability in dust endotoxin using a much larger and
more representative sample of homes in the metropolitan
Boston area with at most two measurements per room in
different seasons. Gereda et al. (2000) made repeated
measurements of house dust endotoxin on 11 homes, 6 months
apart, finding no significant differences in endotoxin
of dust samples from the two assessments. They did not
report the replicate data in their limited number of
homes. Heinrich et al. (2003) reported repeated measurements
of endotoxin in homes over a 1-year period. They found
that endotoxin measurements expressed as units per area
were more consistent and better able to distinguish between
homes than were measurements expressed as units per gram.
However, both methods gave higher between- than within-home
variances and suggested that single measurements could
be used as proxies for average exposure during the year
of sampling. In our study, the protocol for collection
of kitchen and family room dust samples did not use standard
areas because they targeted furnishings or certain architectural
features of the rooms, and therefore precluded estimation
and analysis of units per area.
Endotoxin levels in a given room were only moderately
correlated with those from other rooms in the same home,
suggesting that an endotoxin sample from a single room
may not indicate endotoxin in other rooms or the house
as a whole. In an epidemiologic study, dust sampling
in several rooms, plus a determination of the relative
time spent by the subject in each room may provide a
better estimate of household endotoxin exposure at a
point in time. Room-to-room correlations between endotoxin
levels within homes were assessed using two approaches:
Pearson correlations for simple cross-sectional analysis,
using the mean level for each room if repeated measures
were available; and a mixed-effects model (Heederik et
al. 1991; Rosner 1995). The cross-sectional correlation
coefficients are estimates of the degree to which room-specific
endotoxin is indicative of endotoxin levels in other
rooms within the home. The mixed-effects model estimates
correlations using all of the data accounting for the
correlation in repeated endotoxin levels and thereby
decreasing the uncertainty of these estimates. In this
study, the conventional Pearson correlation coefficients
were for the most part qualitatively similar to the correlation
coefficients estimated from the mixed model.
In our primary analysis, the largest between-home variance
component was observed for kitchen dust endotoxin, followed
by that for bedroom floor samples. The between-home variance
of family room endotoxin was comparatively lower. However,
in multiunit buildings, the between-home variation was
much lower for kitchen floor samples, compared with the
between-home variance for bedroom and family room endotoxin.
Within-home variances were highest for kitchen floor
endotoxin. Room-specific differences in the within-home
variance component were most dramatic for endotoxin sampled
from kitchens in multiunit buildings. The large within-home
variation in endotoxin observed for kitchen dust samples
may be due to water, food products, and vegetable matter
being present to varying degrees in homes and over time.
The reproducibility of repeated endotoxin measurements
in dust from the kitchen and bedroom floors, as indicated
by intrahome correlation coefficients of 0.65 for both,
was greater than reported by Park et al. (2000) The intrahome
correlation for endotoxin sampled from the family room
(r = 0.54) was lower than observed for the other
rooms in this analysis but higher than those reported
by Park et al. (2000) for kitchen and bedroom floor dust.
Park and colleagues did not sample family room dust.
Possibly, rooms with more usage and foot traffic have
more variability in endotoxin levels over time. The moderate
temporal stability of endotoxin levels observed in this
assessment suggests that a single exposure assessment
provides a reasonable, although not an optimal indication
of endotoxin levels over time.
Within-home variability in endotoxin was less than
between-home variability for all three rooms, suggesting
that factors affecting endotoxin levels within a home
over time influence the total variability less than factors
contributing to home-to-home differences in endotoxin.
We observed smaller within-home variances, larger between-home
variances, and correspondingly smaller within- to between-home
variance ratios than those observed by Park et al. (2000).
In contrast to Park et al. (2000), we sampled dust in
a far larger number of homes that were likely more representative
of metropolitan Boston area households (e.g., Park and
colleagues did not include homes with dogs) and thus
also expected to have a larger between-home variance.
The present study was limited, however, by having fewer
repeated measurements and insufficient repeated bed dust
and air samples for analysis. Another limitation was
that we could not compute endotoxin loading per unit
area because dust was collected from family room furnishings
as well as floors and around the perimeters of the kitchen.
The ratio of within- to between-home variance may be
used to better interpret reports of associations between
endotoxin levels and disease outcomes and inform endotoxin
exposure assessment strategies for future studies. In
an optimal study of chronic exposure to house dust endotoxin,
all variability would be observed between homes and endotoxin
levels would not vary over time in the same sampling
area. In that case, the within-home to between-home variance
ratio would be zero, and provided there are no other
sources of bias, a single endotoxin measurement would
provide an unbiased estimate of the effects of chronic
exposure on an outcome. In practice, a single measurement
of endotoxin taken in one room of a home is likely to
be an imperfect surrogate for chronic endotoxin exposure
in that home. If the within-home variance is nonzero,
the observed room-specific endotoxin level will deviate
from the true room-specific mean level. If we assume
that the observed measure is an imperfect measure of
the true mean endotoxin for a room but that the error
in measurement is uncorrelated with the true endotoxin
level, the association between single samples of endotoxin
in homes and health effects is likely to be attenuated
relative to the true effect of chronic exposure (Heederik
et al. 1991; Zeger et al. 2000).
The relationship between the effect estimate obtained
using an observed, misclassified exposure and the true
effect estimate has been derived in the univariate setting
with one exposure variable and no covariables. The attenuation
of the effect estimate is given by

where β* is the
observed linear effect estimate, β is
the true effect estimate,  2w is
the within-home variance,
2w is
the within-home variance,  2b is
the between-home variance, and n is the number
of repeated samples per sampling unit. The magnitude
of attenuation increases as the within-home to between-home
variance ratio increases. Because the magnitude of misclassification
depends not on the value of either within- or between-home
variance but on the ratio of the two, there are several
theoretical approaches to reduce or avoid the bias of
the exposure-outcome relationship. Namely, one
could maximize variability of endotoxin across subjects,
thereby increasing
2b is
the between-home variance, and n is the number
of repeated samples per sampling unit. The magnitude
of attenuation increases as the within-home to between-home
variance ratio increases. Because the magnitude of misclassification
depends not on the value of either within- or between-home
variance but on the ratio of the two, there are several
theoretical approaches to reduce or avoid the bias of
the exposure-outcome relationship. Namely, one
could maximize variability of endotoxin across subjects,
thereby increasing  2b,
or sample endotoxin repeatedly to better estimate true
room-specific mean levels within homes.
2b,
or sample endotoxin repeatedly to better estimate true
room-specific mean levels within homes.
Applying this theory to our findings, epidemiologic
studies using a single house-dust endotoxin observation
as an index of chronic exposure may underestimate the
effect of endotoxin on an outcome, given that such an
effect exists and no other bias or misclassification
is present. In the present study, if dust from the three
rooms were equally good proxies for actual exposure,
using family room endotoxin as the exposure measure,
which has the highest within-home to between-home variance
ratio, would result in the largest degree of attenuation
of effects, relative to using endotoxin from the other
rooms.
Variance components provide a statistical basis for
sampling but should not be the only determinant of a
home sampling strategy. To properly assess exposure,
one must consider other determinants of exposure, including
where the subjects spend their time. We found that at
the time of the first dust sampling, 64% of the children
were reported to spend most of their time in the family
room, whereas 12 and 6% reported spending most of their
time in the kitchen and bedroom, respectively. Eighty-five
percent of participants classified the child’s
time spent in the family room as more than in other rooms.
Thus, use of family room dust samples may provide a better
indicator of exposure compared with using only bedroom
or only kitchen dust samples.
The true window in which endotoxin exposure may act
to modify allergen sensitivity is not known. There is
experimental evidence that endotoxin effects are both
time and dose dependent (Eisenbarth et al. 2002; Tulic
et al. 2000; Wan et al. 2000). It is possible that exposures
in a specific perinatal period may be protective of allergic
disease development, whereas similar exposures occurring
at less relevant periods or at different doses may be
innocuous or even promote allergic disease. Thus, the
timing of endotoxin exposure sampling with respect to
the development of the child may be more important in
defining risk than the season in which the sample was
collected.
The initial motivation for conducting the repeated-measures
dust sampling was to assess the effects of season on
indoor allergens focusing on homes initially sampled
during winter months. Thus, the second home assessment
was conducted in a complementary season relative to the
first home visit. The first home assessments were conducted
during all seasons, although the samples from homes selected
for repeated sampling were collected during the winter
and spring months. In contrast, the repeated measurements
all were taken from dust collected during the summer
and fall months. Because of seasonal variability in endotoxin
levels, GM endotoxin was higher for the second home sampling
compared with the first assessment. The variability and
correlation in endotoxin over time were assessed using
both models that adjusted for season of sampling and
those that did not. Adjusting for a fixed-effect of season
decreased the variability observed within homes and increases
variation between homes. As a result, correlations increased
and the ratio of within- to between-home variance decreased
after controlling for season of sampling.
The endotoxin levels we observed (maximum < 2,000
EU/mg dust) are comparable with those seen in studies
of house dust endotoxin in other urban settings (Gereda
et al. 2000; Park et al. 2000; Rizzo et al. 1997) but
lower than in studies including rural or farm homes (Braun-Fahrlander
et al. 2002; von Mutius et al. 2000). We observed lowest
endotoxin levels in dust sampled during the winter months
and highest levels in dust sampled during the summer.
Park et al. (2000) found similar seasonal patterns for
outdoor samples of endotoxin but suggested that endotoxin
samples from indoor house dust may not follow this pattern
consistently. Our finding of seasonal variability is
consistent with the findings of Rizzo et al. (1997) in
a case-control study of endotoxin and asthma in
children 6-16 years of age living in São
Paulo, Brazil, who reported endotoxin levels to be generally
lower in the winter months and higher in summer months.
Study-to-study comparisons of endotoxin are often limited
by interlab differences in endotoxin assay protocols.
However, the samples described here were assayed by the
same laboratory using the same protocols and Limulus lysates
as the data reported by Park et al. (2000). Because this
sample included only urban and suburban homes, our results
may generalize only to other metropolitan regions of
developed countries in temperate climates. Although we
observed variation in endotoxin levels within and between
homes in our study, the degree of heterogeneity is likely
small relative to industrial or agricultural settings,
where sources of endotoxin exist in particular locations
and not in others. Similarly, one might expect a larger
degree of between-home variability, and perhaps also
different patterns of variability in endotoxin if we
included both nonfarm and farm households, as has been
done in Europe (Braun-Fahrlander et al. 2002).
Our sampling design was not balanced with respect to
season, but this poses no problem for estimation of the
variance components using the mixed-effects model. The
precision of the temporal variance component estimates
(within-home variance) was limited by the fact that we
sampled endotoxin at most two times from a given room.
On the other hand, this sample included a large number
of homes compared with previous studies.
All else being equal, bedroom and kitchen floor samples
provided slightly more stable estimates of endotoxin
over time. Within-home variation in endotoxin levels
was smaller than between-home variation for the three
sampling locations. The correlation over time and the
ratio of within-home to between-home variance observed
in this study support the use of a single endotoxin measurement
as a marker for chronic endotoxin exposure in association
studies.

