Label Review Manual
Chapter 7: Precautionary Statements
Table of Contents
- Purpose of Manual
- What is a Pesticide?
- General Labeling Requirements
- Types of Label Reviews
- Ingredient Statement
- Use Classification
- Precautionary Labeling
- Environmental Hazards
- Physical or Chemical Hazards
- Worker Protection Labeling
- Directions for Use
- Labeling Claims
- Storage and Disposal
- Identification Numbers
- Company Name and Address
- Graphic & Symbols on Labels
- Content/Net Weight Statement
- Unique Product Labeling
- The Consumer Labeling Initiative and Pesticide Labels
Current as of August 2007
On this page
- A. Documents Used to Determine Precautionary Statements on Labels
- B. Acute Toxicity Data
- C. Use of Reregistration Eligibility Decision (RED) Documents
- A. The Signal Word
- B. Poison - Skull and Crossbones Symbol
- C. Child Hazard Warning Statement
- D. Hazards to Humans and Domestic Animals Statements
- E. Personal Protective Equipment (PPE)
- F. First Aid Statements
- G. Note to Physicians
I. Introduction
The precautionary statements are designed to provide the pesticide user with information regarding the toxicity, irritation and sensitization hazards associated with the use of a pesticide, as well as treatment instructions and information to reduce exposure potential. While the Precautionary Statements include Personal Protective Equipment (for uses that trigger worker protection standards), User Safety Requirements, Engineering Controls, User Safety Recommendations, Environmental Hazards, and Physical or Chemical Hazards, for the purposes of this manual, those topics are addressed in other chapters. The remaining sections (Signal Word, Child Hazard Warning, Hazards to Humans and Domestic Animals, First Aid and Personal Protective Equipment [Non-Worker Protection Standard (WPS)] are fully addressed in this chapter. Label reviewers should consult the mandatory/advisory PR Notice 2000-5 for guidance in recommended language for precautionary statements.
II. Background Information
- Documents Used to Determine Precautionary Statements on Labels.
The Code of Federal Regulations specifies both the acute toxicity category endpoints [40 CFR 156.62] and the Hazards to Humans and Domestic Animals statements associated with each toxicity category [40 CFR 156.70]. These toxicity categories and labeling statements however are not currently being used by the Agency, because they are less detailed and provide less protection for pesticide users than other guidance. The 40 CFR 156.70(c) states that specific statements pertaining to the hazards of the product and its uses must be approved by the Agency. The labeling statements provided in the Federal Register Notice issued on 9/26/84, entitled Proposed Rule on Labeling Requirements (Volume 49, Number 188) have been used by the Agency for the past twenty years. The acute toxicity categories listed in the Proposed Rule are also being used with one exception. The acute inhalation toxicity category endpoints currently used are from a 2/1/94 Health Effects Division paper entitled "Interim Policy for Particle Size and Limit Concentration Issues in Inhalation Toxicity Studies.”
- Acute Toxicity Data.
The Signal Word, Hazards to Humans and Domestic Animals, Personal Protective Equipment (non-WPS) and First Aid statements are typically determined by the results of the six acute toxicity studies performed with the product formulation. The acute oral, acute dermal and acute inhalation studies evaluate systemic toxicity via the designated routes of exposure. The primary eye irritation and primary skin irritation studies measure irritation or corrosion, while the dermal sensitization study evaluates the potential for allergic contact dermatitis. With the exception of dermal sensitization, each acute study is assigned to a toxicity category based on the study results (See Table 1 below). The results of these six acute toxicity studies must be known in order for the appropriate labeling language to be determined.
1 4 hr exposureTable 1 - Toxicity Categories Study Category I Category II Category III Category IV Acute Oral Up to and including 50 mg/kg > 50 thru 500 mg/kg > 500 thru 5000 mg/kg > 5000 mg/kg Acute Dermal Up to and including 200 mg/kg > 200 thru 2000 mg/kg > 2000 thru 5000 mg/kg > 5000 mg/kg Acute Inhalation1 Up to and including 0.05 mg/liter > 0.05 thru 0.5 mg/liter > 0.5 thru 2 mg/liter > 2 mg/liter Primary
Eye IrritationCorrosive (irreversible destruction of ocular tissue) or corneal involvement or irritation persisting for more than 21 days Corneal involvement or other eye irritation clearing in 8-21 days Corneal involvement or other eye irritation clearing in 7 days or less Minimal effects clearing in less than 24 hours Primary
Skin IrritationCorrosive (tissue destruction into the dermis and/or scarring) Severe irritation at 72 hours (severe erythema or edema) Moderate irritation at 72 hours (moderate erythema) Mild or slight irritation at 72 hours (no irritation or slight erythema)
- Use of Reregistration Eligibility Decision (RED) Documents.
During Reregistration, the RED document may also specify personal protective equipment, engineering controls and user safety recommendations. In cases where RED specifications differ from those determined by the acute toxicity categories, the most protective statements must be employed. The regulations allow use of a higher signal word for human hazard when necessary to prevent unreasonable adverse effects on man and the environment. 40 CFR 156.64(b)(1)
III. Determining the Precautionary Labeling
- The Signal Word
- When Required. A Signal Word is required for all registered pesticide products unless the pesticide product meets the criteria of Toxicity Category IV by all routes of exposure. If a signal word is used in this case, it must be “Caution.”
- Determining the Signal Word. The Signal Word is determined by the most severe toxicity category assigned to the five acute toxicity studies (see table 1) or by the presence of methanol in concentrations of 4% or more. The Signal Words and associated toxicity categories are as follows:
Toxicity Category IDANGERToxicity Category II WARNINGToxicity Category III CAUTIONToxicity Category IV None Required
Refer to the acute toxicity data review to determine the most severe toxicity category. Also check the Confidential Statement of Formula to determine if methanol is present in concentrations of 4% or more. If so, the Signal Word, regardless of the toxicity categories noted in the acute toxicity review, is DANGER.
- Location and Prominence. The Signal Word is required to appear on the front panel of the label, and the Agency requests that it appear on a separate line from the required Child Hazard Warning statement, Keep Out of Reach of Children (KOROC). It is preferred that it appear below the KOROC statement. The Signal Word is also required on any supplemental labeling intended to accompany the product in distribution or sale. The Agency also requests that it appear in the Precautionary Statements section immediately below the subheading "Hazards to Humans and Domestic Animals". In cases where the “First Aid” and “Hazards to Humans and Domestic Animals” statement appear on the front panel, the Agency requests that the Signal Word be placed directly below the Child Hazard Warning statement, but it does not have to be repeated after the "Hazards to Humans and Domestic Animals" statement. All required front panel warning statements shall be grouped together on the label, and shall appear with sufficient prominence relative to other front panel text and graphic material to assure that they will not be overlooked under customary conditions of purchase and use [40 CFR 156.60(b)]. See Chapter 3 for the Minimum Type Size requirements for the Signal Word, Child Hazard Warning statement and the sample label format. The Agency prefers that the Signal Word appear in all capital letters and be oriented in the same direction as other label text.
- Related Information. Because of the potential for confusion, the Agency historically has not approved labels containing the terms "caution," "warning", or "danger,” except as the Signal Word for that label (e.g., "CAUTION: Wash hands before eating, or smoking" on a label with the signal of “Caution”). California's Proposition 65 may require the term "warning" be used on a label. However, registrants should use the term "notice" or "attention" instead, so that it does not conflict with the EPA required Signal Word.
- When Required. A Signal Word is required for all registered pesticide products unless the pesticide product meets the criteria of Toxicity Category IV by all routes of exposure. If a signal word is used in this case, it must be “Caution.”
- Poison - Skull and Crossbones Symbol
- When Required.
The word "POISON" and the skull and crossbones symbol
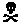 are required for products classified as toxicity category I for acute oral, acute dermal, or acute inhalation toxicity studies [40 CFR 156.64(a)(1)]. It is also required if the inert, methanol, is present at 4% or more in the subject product.
are required for products classified as toxicity category I for acute oral, acute dermal, or acute inhalation toxicity studies [40 CFR 156.64(a)(1)]. It is also required if the inert, methanol, is present at 4% or more in the subject product.
* Product C and Product E must also bear additional labeling (Skull & Crossbones symbol in close proximity to the word "POISON" which must appear in red on a contrasting background). Product C must bear the additional labeling as a result of the toxicity category I classification for the acute oral toxicity study. Product E must bear the additional labeling because it contains a special inert (methanol) at greater than 4%, as described in paragraph B.1. above.Table 2 - Examples of Signal Word Determination Type of Study Product A Product B Product C* Product D Product E* Acute Oral III IV I III III Acute Dermal IV III III IV III Acute Inhalation III IV III III III Primary Eye III II I I III Primary Skin IV IV II IV III Special Inert, e.g., methanol No No No No Yes* CORRECT SIGNALWORD CAUTION WARNING DANGER
(Poison with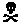 Skull & Crossbones)
Skull & Crossbones)DANGER DANGER
(Poison with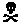 Skull & Crossbones)
Skull & Crossbones)
- Location and Prominence. If required, the word "POISON" and the skull and crossbones symbol must appear in immediate proximity to each other. The word "POISON" must appear in red on a background of a distinctly contrasting color. If the proposed label does not indicate these display requirements, include this requirement in your response to the registrant. In addition, the Agency requests that the “Poison” and the skull and crossbones symbol appear near the Tox.1 signal word “Danger”.
- When Required.
The word "POISON" and the skull and crossbones symbol
- Child Hazard Warning Statement
- When Required. The Child Hazard Warning statement, “Keep Out of Reach of Children” (KOROC) is required on all product labels, unless the requirement is waived. The warning statement requirement may be waived when the registrant adequately demonstrates that the likelihood of contact with children during distribution, storage or use (e.g., an MUP in some situations) is extremely remote or if the pesticide is approved for use on infants or small children.
- Location and Prominence. Location and Prominence. The Child Hazard Warning statement must appear on the front panel [40 CFR 156.66]. It is preferred that the Child Hazard Warning appear on a separate line above the Signal Word. If the Signal Word and Child Hazard Warning were to appear on the same line, a pesticide user could incorrectly assume that the Signal Word is intended primarily for children rather than as a general precaution for all persons. If the label under review has the signal word and Child Hazard Warning on the same line, request that the registrant revise the label. Also make sure that the Child Hazard Warning statement is oriented in the same direction as other label text.
- Additional Information. Based on the FIFRA unreasonable adverse effects standard, the Agency has not allowed the Precautionary Statements or the Directions for Use to contain any statement which implies that the product may be used by children. For example, draft labels of products intended to repel insects should not contain instructions such as "Do not allow use by small children without close adult supervision." Such labeling creates unacceptable risk issues, as it implies that a child can apply the product as long as an adult watches.
A modified Child Hazard Warning statement may be used for products where child contact is expected during normal use. For products requiring a modified statement, make sure that the statement is appropriate for the use pattern. Examples of appropriate statements are as follows:
"Do not allow children to apply product" or "Do not allow children to play with pet collar."
- When Required. The Child Hazard Warning statement, “Keep Out of Reach of Children” (KOROC) is required on all product labels, unless the requirement is waived. The warning statement requirement may be waived when the registrant adequately demonstrates that the likelihood of contact with children during distribution, storage or use (e.g., an MUP in some situations) is extremely remote or if the pesticide is approved for use on infants or small children.
- Hazards to Humans and Domestic Animals Statements
- When Required. Hazards to Humans and Domestic Animals statements are required when any acute toxicity study results in a product classification of toxicity category I, II, or III and/or when the dermal sensitization study result is positive. Hazards to Humans and Domestic Animals statements may specify both mandatory actions and advisory information.
- Required Header. The Hazards to Humans and Domestic Animals statements must appear under the section heading "Precautionary Statements" and below the subheading "Hazard to Humans and Domestic Animals." The Signal Word must appear before the precautionary paragraph. [40 CFR 156.70]
- Location and Prominence. The Hazards to Humans and Domestic Animals section may appear on any panel. Please note, however, that these statements should not be included within the Directions For Use section. These statements should be organized so that the routes of exposure of most concern (severe routes of exposure) as supported by the toxicity category classification are listed first.
- Determining the Hazards to Humans and Domestic Animals
Statements for Fumigant Products. Refer to PR Notice 84-5, Registration Standards or Reregistration Eligibility Decision Documents (REDs) suggested Hazards to Humans and Domestic Animals statements.
- Determining the Hazards to Humans and Domestic Animals
Statements for Non-Fumigant Products. Statements from the tables 3-8 can be selected based on the toxicity category assigned to each study. Statements from these tables should be combined to form a concise paragraph. Repetitious sentences should be omitted. In cases where the toxicity categories are not known, the precautionary labeling must be consistent with the signal word.
- Related Information. Hazards to Humans and Domestic Animals statements must be appropriate for all uses on the label. These statements must be consistent with each use pattern listed on the label. No statement should be used that is reasonably beyond the control of the typical applicator. Hazards to Humans and Domestic Animals statements must not require use of specialized equipment which would not be readily available to the typical user of the product.
- Products that contain greater than 4% Methanol. If the product contains 4% or more of methanol, the Agency believes that in order to mitigate potential risk the following statement should be added to the label:
"Methanol may cause blindness."
Table 3 - Typical Statements for Acute Oral Toxicity Toxicity Category Signal Word Statements I DANGER-POISON
Skull & Crossbones required*Fatal if swallowed. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. II WARNING May be fatal if swallowed. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. III CAUTION Harmful if swallowed. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. IV CAUTION (optional) No statements are required. However, the registrant may choose to use category III labeling.
Table 4 - Typical Statements for Acute Dermal Toxicity Toxicity Category Signal Word Statements I DANGER-POISON
Skull & Crossbones requiredFatal if absorbed through skin. Do not get in eyes, on skin, or on clothing. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. Wear [specify appropriate protective clothing]. Remove and wash contaminated clothing before reuse. II WARNING May be fatal if absorbed through skin. Do not get in eyes, on skin, or on clothing. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. Wear [specify appropriate protective clothing]. Remove and wash contaminated clothing before reuse. III
CAUTION Harmful if absorbed through skin. Avoid contact with skin, eyes or clothing. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. Remove and wash contaminated clothing before reuse. Wear [specify any appropriate protective clothing, if appropriate]. IV CAUTION (optional) No statements are required. However, the registrant may choose to use category III labeling.
* Choose the word which appropriately describes the product during use.Table 5 - Typical Statements for Acute Inhalation Toxicity Toxicity Category Signal Word Statements I DANGER-POISON
Skull & Crossbones requiredFatal if inhaled. Do not breathe (dust, vapor, or spray mist).* Wear [specify appropriate respiratory protection from Table 4, Chapter 10]. Remove and wash contaminated clothing before reuse.II WARNING May be fatal if inhaled. Do not breathe (dust, vapor or spray mist).* Wear [specify appropriate respiratory protection from Table 4, Chapter 10]. Remove and wash contaminated clothing before reuse. III CAUTION Harmful if inhaled. Avoid breathing (dust, vapor or spray mist).* Remove and wash contaminated clothing before reuse. IV CAUTION (optional) No statements are required. However, the registrant may choose to use category III labeling.
* The term "corrosive" is not required if corrosive effects were not observed during the study.Table 6 - Typical Statements for Primary Eye Irritation Toxicity Category Signal Word Statements I DANGER Corrosive.* Causes irreversible eye damage. Do not get in eyes or on clothing. Wear [specify appropriate protective eyewear such as goggles, face shield, or safety glasses]. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. Remove and wash contaminated clothing before reuse. II WARNING Causes substantial but temporary eye injury. Do not get in eyes or on clothing. Wear [specify appropriate protective eyewear such as goggles, face shield, or safety glasses]. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. Remove and wash contaminated clothing before reuse. III CAUTION Causes moderate eye irritation. Avoid contact with eyes or clothing. Wear [specify protective eyewear, if appropriate]. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. IV CAUTION (optional) No statements are required. However, the registrant may choose to use category III labeling.
Table 7 - Typical Statements for Primary Skin Irritation Toxicity Category Signal Word Statements I DANGER Corrosive. Causes skin burns. Do not get in eyes, on skin, or on clothing. Wear [specify appropriate protective clothing and gloves]. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. Remove and wash contaminated clothing before reuse. II WARNING Causes skin irritation. Do not get on skin or on clothing. Wear [specify appropriate protective clothing and gloves]. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, using tobacco or using the toilet. Remove and wash contaminated clothing before reuse. III CAUTION Avoid contact with skin or clothing. Wash thoroughly with soap and water after handling and before eating, drinking, chewing gum, or using tobacco. Wear [specify protective clothing and gloves, if appropriate]. IV CAUTION (optional) No statements are required. However, the registrant may choose to use category III labeling.
Table 8 - Typical Statements for Dermal Sensitization Study Results Statement Product is a sensitizer or is positive for sensitization Prolonged or frequently repeated skin contact may cause allergic reactions in some individuals. Product is not a sensitizer or is negative for sensitization No labeling is required for this result.
- When Required. Hazards to Humans and Domestic Animals statements are required when any acute toxicity study results in a product classification of toxicity category I, II, or III and/or when the dermal sensitization study result is positive. Hazards to Humans and Domestic Animals statements may specify both mandatory actions and advisory information.
- Personal Protective Equipment (PPE)
PPE requirements are specified for uses covered under the Worker Protection Standard (WPS), but there are no regulatory requirements for non-WPS products, products used by residents, or products intended only for manufacturing use. However, to protect human health, the following guidance is offered.
- For Non-WPS (Industrial/Commercial) Uses
While there are no regulatory requirements that demand PPE for non-WPS products, applicators are tested on their comprehension regarding what types of PPE are used and how to use them correctly. This testing implies that PPE is expected to be worn by commercial applicators. In order to protect human health, label reviewers are encouraged to review the toxicity data and the potential exposures from the product’s uses to determine whether PPE would be necessary to meet the standards for registration. In cases where the reviewers determine PPE would be necessary, the various PPE tables in Chapter 10 provide information about which PPE is protective in specific circumstances. If there is an applicable regulatory document which specifies PPE requirements based on concerns specific to the active ingredient then those PPE requirements must be placed on the label.
- For Products used by Residents/Consumers.
In order to protect human health, label reviewers are encouraged to review the toxicity data and the product’s uses to determine whether PPE would be necessary to meet the standards for registration. In cases where the reviewers determine PPE would be necessary, the PPE tables in Chapter 10 provide information about which PPE is protective in specific circumstances. In some cases, the PPE indicated in these tables may need to be modified; for example, to fit the consumer’s ability to acquire it. For example, “shoes” may need to be substituted for “chemical resistant footwear” or “safety glasses” may need to be substituted for “protective eyewear.” If there is an applicable regulatory document which specifies PPE requirements based on concerns specific to the active ingredient then those PPE requirements must be placed on the label.
- For Non-WPS (Industrial/Commercial) Uses
- First Aid Statements
- When Required. A First Aid statement is required when any acute toxicity study result is classified as category I, II, or III. It is acceptable for the registrant to include First Aid statements for study results that are classified as category IV.
- Appropriate Headers. The first aid statements appear under either of the following headings: “First Aid” or “Statements of Practical Treatment.” [PR Notice 2001-1]. The heading “First Aid” is preferred by the Agency. In addition, EPA historically has not allowed the heading "Antidote" in conjunction with the first aid statements unless a specific antidote is necessary.
- Location and Prominence. First Aid statements shall appear on the front panel of the label for all products classified as toxicity category I [40 CFR 156.68]. The Agency may, however, permit reasonable variations in the placement of the First Aid statement as long as the reference statement, “See First Aid (or Statement of Practical Treatment) on [identify appropriate panel]” appears on the front panel, preferably near “Poison” and the skull and crossbones. First Aid statements for toxicity categories II and III classification may appear on any panel of the label. However, any time First Aid statements appear other than on the front panel, a referral statement such as,” See side/back panel for First Aid" should appear on the front panel in close proximity to the Signal Word. Furthermore, First Aid statements on the side or back panel should be grouped near the other precautionary labeling text, yet set apart or distinguishable from the other label text. First Aid statements should be organized so that the most severe routes of exposure, as demonstrated by the toxicity classification, are listed first.
- Determining the First Aid Statements for Fumigant Products.
Refer to PR Notice 84-5 and Registration Standards/REDs.
- Determining the First Aid Statements for Non-Fumigant
Products. Review Table 9 to determine the preferred First Aid statements for each route of exposure. Registrants should support alternative First Aid statements with medical evaluations of the product. Approval of alternative First Aid statements is guided by considerations such as those set out in the "Content and Clarity" section below. The Agency has not approved the use of salt water for emesis as a first aid technique. [PR Notice 80-2].
- Content and Clarity. First Aid statements should be brief, clear, simple and in straightforward language so that the average person in an emergency can easily and quickly understand the instructions. First Aid statements should apply to all ages or when necessary, should include distinctions between the treatments for different ages (e.g., children vs. adults). Any reasonably competent individual should be able to perform the First Aid statements. These statements should not include procedures which must be performed by medical personnel or require specialized equipment. Such procedures belong under the Note to Physician heading (see section G below).
- Acute Dermal and Primary Skin Irritation. Because both of these studies focus on the dermal route of exposure, any first aid statements required by the results of these two studies can be combined. Use the first aid statement required for the acute dermal toxicity study if the results of both studies place the product in the same acute toxicity category. Use the statements for the more severe acute toxicity category if the results of the studies would place the product in different acute toxicity categories.
- Eye and Skin Irritation. If the product is corrosive and is in toxicity category I or II for eye or dermal irritation, then a first aid statement for ingestion may also be included.
- Content and Clarity. First Aid statements should be brief, clear, simple and in straightforward language so that the average person in an emergency can easily and quickly understand the instructions. First Aid statements should apply to all ages or when necessary, should include distinctions between the treatments for different ages (e.g., children vs. adults). Any reasonably competent individual should be able to perform the First Aid statements. These statements should not include procedures which must be performed by medical personnel or require specialized equipment. Such procedures belong under the Note to Physician heading (see section G below).
- If the product contains an organophosphate (i.e.,
an organophosphorus ester that inhibits cholinesterase) or an N-methyl
carbamate (i.e., an N-methyl carbamic acid ester that inhibits cholinesterase).
If the product contains either chemical, the following phrase should be included in the First Aid statement [PRN 2001-1]:
“CONTAINS AN __________________ (either organophosphate or N-methyl carbamate) THAT INHIBITS CHOLINESTERASE.“
- If the product contains zinc phosphide. If the product contains zinc phosphide, If the product contains zinc phosphide, the following First Aid statement is recommended [PRN 2001-1]:
-
Products that contain petroleum distillates. If the product contains ≥10% petroleum distillates, the following First Aid statement should be used [PRN 2001-1]:
“If swallowed: Immediately call a poison control center or doctor. Do not induce vomiting unless told to do so by a poison control center or doctor. Do not give any liquid to the person. Do not give anything by mouth to an unconscious person.”
However, if registrants have data to show there is benefit in drinking water or milk after ingesting their product(s), they may submit alternate wording via amendment.
-
Telephone numbers. EPA encourages registrants to include a company telephone number or toll-free hotline number for emergency information in the first aid section. If a number is included, confusion can be avoided by placing emergency numbers with the “First Aid” or “Hot Line Number” text. If a number is included, it should include a phrase or statement indicating the kinds of information the number should be used for and it may include hours of service. For example:
“Have the product container or label with you when calling a poison control center or doctor, or going for treatment. For emergency information on [product, usage, etc.], call [1-XXX-XXX-XXXX], Monday through Friday, 9 a.m. to 5 p.m. After 5 p.m. call your poison control center at 1-800-222-1222.”
If a registrant does not have its own number, the registrant may use the National Pesticides Information Center (NPIC) 800 number (see below).
-
National Pesticides Information Center. Regarding the General Information provided under the First Aid statements section (Table 9), the emergency phone number could be the National Pesticides Information Center’s 800 number. The section could read:
“Have the product container or label with you when calling a poison control center, doctor, or going for treatment. For emergency information concerning this product, call the National Pesticides Information Center (NPIC) at 1-800-858-7378 seven days a week, 6:30 am to 4:30 pm Pacific Time (NPIC Web site: www.npic.orst.edu).”
Table 9 - First Aid Statements Route of Exposure and Toxicity CategoryFirst Aid Statements Ingestion treatment for acute oral toxicity categories 1, 2, and 3If swallowed: - Call a poison control center or doctor immediately for treatment advice.
- Have person sip a glass of water if able to swallow.
- Do not induce vomiting unless told to by a poison control center or doctor.
- Do not give anything to an unconscious person.
Acute oral toxicity category 4Statement is not required. Registrants may use toxicity category 1-3 statements if they choose. Skin exposure treatment for acute dermal toxicity, and irritation categories 1, 2, and 3If on skin: - Take off contaminated clothing.
- Rinse skin immediately with plenty of water for 15-20 minutes.
- Call a poison control center or doctor for treatment advice.
Dermal and skin irritation toxicity category 4Statement is not required. Registrants may use toxicity category 1-3 statements if they choose.Inhalation treatment for acute toxicity categories 1, 2, and 3If inhaled:- Move person to fresh air.
- If person is not breathing, call 911 or an ambulance, then give artificial respiration, preferably mouth-to-mouth if possible.
- Call a poison control center or doctor for further treatment advice.
Inhalation toxicity category 4Statement is not required. Registrants may use toxicity category 1-3 statements if they choose.Eye exposure treatment for eye irritation categories 1, 2, and 3If in eyes: - Hold eye open and rinse slowly and gently with water for 15-20 minutes. Remove contact lenses, if present, after the first 5 minutes, then continue rinsing.
- Call a poison control center or doctor for treatment advice.
Eye irritation toxicity category 4Statement is not required. Registrants may use toxicity category 1-3 statements if they choose.General information to include either near the first aid statement or emergency phone number- Have the product container or label with you when calling a poison control center or doctor or going for treatment.
- For emergency information on [product, use, etc.], call the National Pesticides Information Center at 1-800-858-7378, 6:30 AM to 4:30 PM Pacific time (PT), seven days a week. During other times, call the poison control center 1-800-222-1222.
“If swallowed: Immediately call a Poison Control Center or doctor or transport the person to the nearest hospital. DO NOT DRINK WATER. Do not administer anything by mouth or make the person vomit unless advised to do so by a doctor.”
- When Required. A First Aid statement is required when any acute toxicity study result is classified as category I, II, or III. It is acceptable for the registrant to include First Aid statements for study results that are classified as category IV.
- Note to Physicians
- When Used.The Note to Physician is not required nor mentioned in the 40 CFR. If the label under review is for a product which is a fumigant, refer to PR Notice 84-5 or relevant Registration Standards or REDS for the appropriate Note to Physician. For all other products, EPA currently uses a Note to Physician as specified in the 1984 proposed rule for the following types of products:
- All products that are classified as toxicity category I.
- Products which are corrosive or classified as toxicity category I for eye or skin. These products must include the following Note to Physician: “Probable mucosal damage may contraindicate the use of gastric lavage.”
- Products which contain > 10% petroleum distillate should include the following Note to Physician: “Contains petroleum distillate. Vomiting may cause aspiration pneumonia.”
- Products which produce physiological effects requiring specific antidotal or medical treatment such as: Cholinesterase Inhibitors (e.g., carbamates and phosphorothioates, and organophosphates); Metabolic Stimulants (e.g., dichlorphenols); Anticoagulants (e.g., warfarin).
- All products that are classified as toxicity category I.
- Location and Prominence. The Note to Physician should be located in close proximity to the First Aid statement, but should be clearly distinguished from it. In other words, it should not be placed within the First Aid statement, but should appear below the last First Aid statement.
- Contents of Note. The Agency does not provide specific Notes to Physicians except for toxicity category I eye and skin irritants. However, the Agency does provide the following guidance concerning the content of Notes to Physicians. Check the label under review to make certain that it addresses the following information:
- technical information on symptomatology;
- use of supportive treatments to maintain life functions;
- medicine that will counteract the specific physiological effects of the pesticide;
- company telephone number to specific medical personnel who can provide specialized medical advice.
- When Used.The Note to Physician is not required nor mentioned in the 40 CFR. If the label under review is for a product which is a fumigant, refer to PR Notice 84-5 or relevant Registration Standards or REDS for the appropriate Note to Physician. For all other products, EPA currently uses a Note to Physician as specified in the 1984 proposed rule for the following types of products:
IV. Labeling Options
- Use Dilutions (Aqueous Solutions only)
- When Used. Additional Hazards to Humans and Domestic Animals and First Aid statements which correspond with the toxicity categories associated with a product’s use dilution may be allowed on product labels provided the conditions below are satisfactorily addressed. Following is guidance for the submission and review of such data and for the content and placement of associated labeling.
- Data Requirements. All data and draft labeling for use dilution Hazards to Humans and Domestic Animals statements should be sent to the appropriate Product Manager with a request for pesticide amendment. In some cases, use dilution labeling statements triggered by systemic toxicity (acute oral, dermal or inhalation toxicity) may be supported by extrapolation from the LD50/LC50 for the concentrate. At a minimum the following is required to even consider extrapolating toxicity categories. This information must be submitted by the Registrant with the extrapolation request.
- A slope calculated from at least three, and preferably more, dose levels having partial responses (i.e., a well characterized dose-response);
- Dose groups sufficiently large (>5 per group) to allow for the calculation of confidence limits that fall within the defined Toxicity Category boundaries;
- Extrapolation to higher toxicity categories will only be applied to water dilutions. It should also be determined that there are no other factors affecting the toxicity of the EP (e.g., inerts that enhance the absorption of the active ingredient, promote the active ingredient’s toxicity, etc.). Other types of extrapolations will be done on a case by case basis.
- Use dilution Hazards to Humans and Domestic Animals statements triggered by skin or eye irritation must be supported by new or cited studies. If another registered diluted product (such as a ready-to-use formulation) has acceptable data and is found similar to the concentrated product after it has been diluted, those data may also be used to support revised labeling.
- A slope calculated from at least three, and preferably more, dose levels having partial responses (i.e., a well characterized dose-response);
- Labeling Requirements.
It is not EPA’s intent to allow dual sets of Hazards to Humans and Domestic Animals statements and/or First Aid statements on the label. Rather, EPA will allow certain modified statements to be added that are applicable to the most concentrated use dilution only. [40 CFR 156.68(b)] These additional statements (triggered by the toxicity category of the most concentrated use dilution) must be placed directly after the required statements for the concentrate. The following are some examples (in italics) of how use dilution labeling could appear on product labeling:
Hazards to Humans and Domestic Animals:
"Causes substantial but temporary eye injury. Do not get in eyes or on clothing. Wear goggles or face shield. After product is diluted in accordance with the directions for use, goggles or face shield are not required."
First Aid:
"If on skin: Wash with plenty of soap and water. Get medical attention. If product, diluted in accordance with the directions for use, gets on skin, medical attention is not required.”
- When Used. Additional Hazards to Humans and Domestic Animals and First Aid statements which correspond with the toxicity categories associated with a product’s use dilution may be allowed on product labels provided the conditions below are satisfactorily addressed. Following is guidance for the submission and review of such data and for the content and placement of associated labeling.
- Optional Labeling/Deviations
- Optional Hazards to Humans and Domestic Animals Statements. Registrants may submit Hazards to Humans and Domestic Animals statements which reflect specific hazards. [40 CFR 156.70(c)] Such requests must be supported by data (or substantive justification), and should be routed to label reviewers or the Chemistry and Exposure Branch (for PPE). For example, the statements "Do not remove contact lenses, if worn. Get immediate medical attention." would not be approved by the Agency without supporting data or rationale.
- Toxicity Category IV Precautionary Labeling. If the product is all toxicity category IV (non-sensitizer), precautionary labeling statements are normally not required. However, if registrants desire to place precautionary labeling on such a product they may do so. To promote labeling consistency it is recommended that the registrant use precautionary statements triggered by toxicity category III. Registrants may propose alternate labeling which should be reviewed by precautionary labeling reviewers.
- Me-Too Deviations. If a me-too product is citing a product that has optional use dilution hazard statements on the label, those statements are not required on the me-too if the acute toxicity data results are available. Questions about the availability of the acute studies should be referred to the precautionary labeling reviewers.
- Optional Hazards to Humans and Domestic Animals Statements. Registrants may submit Hazards to Humans and Domestic Animals statements which reflect specific hazards. [40 CFR 156.70(c)] Such requests must be supported by data (or substantive justification), and should be routed to label reviewers or the Chemistry and Exposure Branch (for PPE). For example, the statements "Do not remove contact lenses, if worn. Get immediate medical attention." would not be approved by the Agency without supporting data or rationale.
![[logo] US EPA](https://webarchive.library.unt.edu/eot2008/20090512144050im_/http://www.epa.gov/epafiles/images/logo_epaseal.gif)