|
<<< Back to Sampling and Analytical Methods |
 Printing Instructions
Printing Instructions |
For problems with accessibility in using figures, illustrations and PDFs in this method, please contact
the SLTC at (801) 233-4900. These procedures were designed and tested for internal use by OSHA personnel.
Mention of any company name or commercial product does not constitute endorsement by OSHA. |
Acetic Acid
Related Information: Chemical Sampling -
Acetic Acid
|
| Method no.: |
PV2119 |
|
|
| Control no.: |
T-PV2119-01-03020-M |
|
|
| Target concentration: |
10 ppm (25 mg/m3) |
|
|
| Procedure: |
Samples are collected by drawing
a known volume of air through glass sampling tubes containing coconut shell
charcoal (SKC Anasorb CSC, lot 2000). Samples are extracted with 0.01 N NaOH
and analyzed by IC using a conductivity detector. |
|
|
| Recommended air volume and sampling rate studied: |
240 min at 0.2 L/min (48 L) |
|
|
| Reliable quantitation limit: |
2.9 ppb |
|
|
| Status of method: |
Partially evaluated method. This
method has been subjected to established evaluation procedures of the Methods
Development Team and is presented for information and trial use. |
|
|
| February 2003 |
Mary E. Eide |
|
|
Methods Development Team
Industrial Hygiene Chemistry Division
OSHA Salt Lake Technical Center
Sandy UT 84070-6406 |
1. General Discussion
1.1 Background
1.1.1 History
The previous partially validated method for acetic acid, ID-186SG, called for
collection on charcoal tubes and extraction with 1.5 mM sodium borate.1 An
extraction study of acetic acid using coconut shell charcoal ( SKC Anasorb CSC,
lot 2000) using 1.5 mM sodium borate had non-linear extraction efficiencies,
which ranged from 99.3% for a 2.098 mg loading to 66.8% for a 0.105 mg loading.
Extraction studies using 0.01 N NaOH had an average recovery of 98.6 over the
range of 0.15 to 2.31 mg loading. Retention studies showed no loss of acetic
acid when 48 L of humid air (~80% RH) was drawn through spiked tubes at 0.2
L/min. Storage studies showed little loss when stored for 14 days at either
refrigerated or ambient temperatures.
1.1.2 Toxic effects (This section is for information only and should
not be taken as the basis of OSHA policy.)2
Acetic acid is a severe skin, eye and mucous membrane irritant. As an eye
irritant it can cause burns, lachrymation, and conjunctivitis. Skin exposure
can cause burns. Mucous membrane exposure results in burns and bleeding from
ulcerations, along with nausea, vomiting and diarrhea.
1.1.3 Workplace exposure3,4
Acetic acid is used as a feedstock in the production of acetates, acetyls,
cellulose acetate, acetate rayon, and plastics. It is used as a laundry sour,
in tanning, and printing and dyeing. It is used as an acidulant and
preservative in foods and pharmaceuticals. It is used as a solvent for gums,
resins, volatile oils, and other organic compounds. In 2002 4.8 billion pounds
of acetic acid were produced in the US.
1.1.4 Physical properties and other descriptive information5,6
|
CAS number:
|
64-19-7
|
IMIS:
|
00207
|
| synonyms:
|
glacial acetic acid; methane carboxylic acid;
ethanoic acid; vinegar acid
|
| RTECS number:
|
AF1225000
|
molecular
weight:
|
60.05
|
| melting point:
|
16.7ºC
|
boiling point:
|
118ºC
|
| appearance:
|
clear liquid
|
molecular
formula:
|
C2H4O2
|
|
odor:
|
vinegar
|
flash point:
|
39ºC (103ºF)(cc)
|
|
autoignition |
|
vapor |
11.52 kPa or |
| temperature: |
465ºC (869ºF) |
pressure: |
11.4 mm Hg @ 20ºC
|
| solubility:
|
water, alcohol, ether
|
density:
|
1.049
|
| structural formula:
|
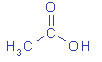 |
|
|
This method was evaluated according to the OSHA SLTC "Evaluation
Guidelines for Air Sampling Methods Utilizing Chromatographic Analysis".8
The Guidelines define analytical parameters, specify required laboratory
tests, statistical calculations and acceptance criteria. The analyte air
concentrations throughout this method are based on the recommended sampling
and analytical parameters.
1.2 Detection limit of the overall procedure (DLOP) and reliable
quantitation limit (RQL)
The DLOP is measured as mass per sample and expressed as equivalent air
concentrations, based on the recommended sampling parameters. Ten samplers
were spiked with equal descending increments of analyte, such that the lowest
sampler loading was 1.02 µg of acetic
acid. This is the amount spiked on a sampler that would produce a peak
approximately 10 times the response for a sample blank. These spiked samplers
were analyzed with the recommended analytical parameters, and the data
obtained used to calculate the required parameters (standard error of estimate
and slope) for the calculation of the DLOP. The slope was 2.29×104
and the SEE was 783.3. The RQL is considered the lower limit for precise
quantitative measurements. It is determined from the regression line
parameters obtained for the calculation of the DLOP, providing 75% to 125% of
the analyte is recovered. The DLOP and RQL were 0.103 µg
and 0.342 µg respectively.
Table 1.2
Detection Limit of the Overall Procedure for Acetic acid |
|
mass per sample
(µg) |
area counts
(µV-s) |
|
| |
0.00 |
|
0 |
|
0.102 |
|
1449 |
|
0.204 |
|
3508 |
|
0.306 |
|
5138 |
|
0.408 |
|
7505 |
|
0.510 |
|
9314 |
|
0.612 |
|
11963 |
|
0.714 |
|
15314 |
|
0.816 |
|
17662 |
|
0.918 |
|
20064 |
|
1.02 |
|
23125 |
|

Figure 1.2.1 Plot of data to determine the DLOP/RQL for acetic acid.
(Y=2.29x104 X - 1240; SEE=783.3) |
|
Below is a chromatogram of the RQL level.
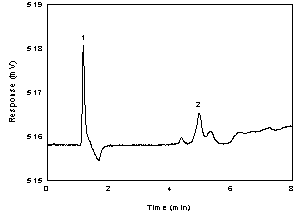
Figure 1.2.2. Chromatogram of the acetic acid standard, as the acetate
ion, near the RQL. (Key: (1) water; (2) acetate ion) |
2. Sampling Procedure
All safety practices that apply to the work area being sampled should be
followed. The sampling equipment should be attached to the worker in such a
manner that it will not interfere with work performance or safety.
2.1 Apparatus
2.1.1 Samples are collected using a personal sampling pump
calibrated, with the sampling device attached, to within ±5% of the
recommended flow rate.
2.1.2 Samples are collected with 7-cm × 4-mm i.d. × 7-mm o.d. glass
sampling tubes packed with two sections (100/50 mg) of coconut shell
charcoal, Anasorb CSC, lot 2000. The sections are held in place and
separated with a glass wool plug and two urethane foam plugs. For this
evaluation, commercially prepared sampling tubes were purchased from SKC,
Inc. (catalog no. 226-01).
2.2 Reagents
None required.
2.3 Technique
2.3.1 Immediately before sampling, break off the ends of the
flame-sealed tube to provide an opening approximately half the
internal diameter of the tube. Wear eye protection when breaking ends.
Use tube holders to minimize the hazard of broken glass. All tubes
should be from the same lot.
2.3.2 The smaller section of the adsorbent tube is used as a back-up and is
positioned nearest the sampling pump. Attach the tube holder to the sampling
pump so that the adsorbent tube is in an approximately vertical position with
the inlet facing down during sampling. Position the sampling pump, tube holder
and tubing so they do not impede work performance or safety.
2.3.3 Draw the air to be sampled directly into the inlet of the
tube holder. The air being sampled is not to be passed through any
hose or tubing before entering the sampling tube.
2.3.4 After sampling for the appropriate time, remove the adsorbent
tube and seal it with plastic end caps. Seal each sample end-to-end
with an OSHA-21 form as soon as possible.
2.3.5 Submit at least one blank sample with each set of samples.
Handle the blank sampler in the same manner as the other samples
except draw no air through it.
2.3.6 Record sample air volumes (liters), sampling time (minutes)
and sampling rate (mL/min) for each sample, along with any potential
interferences on the OSHA-91A form.
2.3.7 Submit the samples to the laboratory for analysis as soon as
possible after sampling. If delay is unavoidable, store the samples in
a refrigerator. Ship any bulk samples separate from the air samples.
2.4 Extraction efficiency
The extraction efficiency was determined by liquid-spiking charcoal tubes,
lot 2000, with acetic acid at 0.1 to 2 times the target concentration. These
samples were stored overnight at ambient temperature and then extracted for 30
minutes on the shaker, then spun down on the centrifuge at 2800 rpm for 5
minutes, and analyzed. The mean extraction efficiency over the studied range
was 98.6%. The wet extraction efficiency was determined at 1 times the target
concentration by liquid spiking the acetic acid onto charcoal tubes which had
10-L humid air (absolute humidity of 15.9 mg/L of water, about 80% relative
humidity at 22.2ºC) drawn through them
immediately before spiking. The mean recovery for the wet samples was 99.0%.
Table 2.4
Extraction Efficiency (%) of Acetic acid |
|
| level |
|
|
sample number |
|
|
|
x target
concn |
mg per
sample |
1 |
2 |
3 |
4 |
5 |
6 |
mean |
|
0.1
0.25
0.5
1.0
1.5
2.0
1.0 (wet)
|
0.115
0.288
0.577
1.15
1.73
2.31
1.15
|
97.4
98.4
98.9
98.0
98.8
96.8
98.7
|
97.6
99.0
99.2
99.7
98.1
97.6
99.1
|
99.5
98.6
99.3
99.2
99.1
98.1
99.1
|
97.3
99.2
99.2
98.5
98.5
97.6
99.4
|
99.4
99.0
99.1
98.8
98.9
97.8
98.5
|
99.8
98.7
99.6
99.1
98.4
98.7
98.9
|
98.5
98.8
99.2
98.9
98.6
97.8
99.0
|
|
2.5 Retention efficiency
Six charcoal tubes, lot 2000 were spiked with 2.31 mg (19.6 ppm) of
acetic acid and allowed to equilibrate for 6 h. The tubes had 48 L humid
air (absolute humidity of 15.9 mg/L of water, about 80% relative
humidity at 22.2ºC) drawn through
them at 0.2 L/min. The samples were extracted and analyzed. The mean
recovery was 97.2%. There was no analyte found on the backup section of
any of the tubes. Results are not corrected for extraction efficiency.
Table 2.5
Retention Efficiency (%) of Acetic acid
|
|
|
|
|
sample number |
|
|
|
| section |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
|
front
rear
total |
96.4
0.0
96.4 |
98.5
0.0
98.5 |
97.4
0.0
97.4 |
96.2
0.0
96.2 |
97.6
0.0
97.6 |
97.1
0.0
97.1 |
97.2
0.0
97.2 |
|
2.6 Sample storage
Fifteen charcoal tubes were each spiked with 1.15 mg (9.76
ppm) of acetic acid. They were allowed to equilibrate for 6 h, then 10 L of
air, with an absolute humidity of 15.7 milligrams of water per liter of air
(about 80% relative humidity at 22.2ºC),
was drawn through them. Three samples were analyzed immediately. Two groups
of six samples were formed with the rest of the samples. One group was
stored at room temperature and the other in a refrigerator. Three from each
group were analyzed after 7 days of storage and the remaining three after 14
days of storage. The amounts recovered, which are not corrected for
extraction efficiency, indicate good storage stability for the time period
studied.
Table 2.6
Storage Test for Acetic acid
|
|
| time (days) |
ambient storage
recovery (%)
|
|
refrigerated storage
recovery (%)
|
|
0
7
14
|
99.4
96.7
94.7
|
97.9
98.5
96.4
|
98.2
97.1
95.8
|
|
99.4
98.6
95.9
|
97.9
97.2
98.2
|
98.2
97.9
96.6
|
|
2.7 Recommended air volume and sampling rate.
Based on the data collected in this evaluation, 48-L air samples should
be collected at a sampling rate of 0.2 L/min for 240 minutes.
2.8 Interferences (sampling)
2.8.1 There are no known compounds which will severely interfere with
the collection of acetic acid.
2.8.2 Suspected interferences should be reported to the laboratory
with submitted samples.
3. Analytical Procedure
Adhere to the rules set down in your Chemical Hygiene Plan. Avoid skin
contact and inhalation of all chemicals and review all appropriate MSDSs.
3.1 Apparatus
3.1.1 Ion chromatograph with a conductivity detector. A Dionex DX500
ion chromatograph with a conductivity detector, and a ASRS anion
suppressor was used in this evaluation.
3.1.2 IC column and guard column which can separate acetate from any
potential interferences. A 250-mm × 4-mm i.d. Dionex IonPac AS4A column and
50-mm × 4-mm i.d. Dionex IonPac AG4A guard column were used in this evaluation.
(Butyric acid is a potential interference on an AS4A column, to obtain a
separation between acetate and butyrate use an AG14A guard column and AS14A
column.)
3.1.3 A means to integrate the chromatograms. The Dionex AI450
software, and a Millennium32 data system were used in this
evaluation.
3.1.4 Automatic sampler. A Dionex model AS40, and sample vials,
0.5-mL, with filter caps was used in this evaluation.
3.1.5 Volumetric flasks, pipets, and calibrated micropipets.
3.1.6 A pipettor capable of dispensing 10-mL of the extracting
solvent to prepare standards and samples. If a dispenser is not
available, a 10-mL volumetric pipet may be used.
3.1.7 Volumetric flasks - 10-mL and other convenient sizes for
preparing standards.
3.1.8 Calibrated 10-µL
syringe for preparing standards if using acetic acid to make analytical
standards.
3.1.9 Micro-analytical balance capable of weighing at least 0.01 mg.
3.1.10 Scintillation vials, glass, 20-mL.
3.1.11 Equipment for eluent degassing. A vacuum pump and ultrasonic
bath were used for this evaluation.
3.1.12 Optional: Centrifuge for spinning down the precipitate in
samples.
3.2 Reagents
3.2.1 Acetic acid, glacial, Reagent grade. Fisher 99.9% (lot
971803) was used in this evaluation. Alternately, sodium acetate may
be used to make analytical standards.
3.2.2 Sodium acetate, Reagent grade. Aldrich 99%+ (lot 16530HS) was
used for this evaluation.
3.2.3 Sodium hydroxide, Reagent grade. Aldrich 97% (lot 09701DQ)
was used in this evaluation.
3.2.4 Sodium borate decahydrate, Reagent grade. Mallinckrodt 99%
(lot KJEZ) was used in this evaluation.
3.2.5 Deionized water, 18 megaohm. A Barnstead NANOpure Diamond
water deionizer was used in this evaluation.
3.2.6 Eluent was prepared by dissolving 1.25 g sodium borate (Na2B4O7·10H2O)
in 2 liters of deionized water, resulting in a 1.5 mM solution.
3.2.7 Extraction solvent is prepared by dissolving 0.4 g NaOH in 1
liter of deionized water, resulting in a 0.01 N NaOH solution.
3.2.8 A 1000 ppm or 1000 µg/mL
acetate ion stock solution is prepared by dissolving 0.6947 g sodium
acetate in 500 mL deionized water.
3.3 Standard preparation
3.3.1 Prepare stock analytical standards by injecting microliter amounts of
acetic acid into volumetric flasks containing 0.01 N NaOH. An analytical
standard at a concentration of 1 µL/10
mL (104.7 ppm solution or 104.7 µg/mL)
is equivalent to 8.88 ppm based on a 48-L air volume. Alternately, a stock solution of sodium acetate (1000 ppm
solution acetate ion or 1000 µg/mL
acetate ion) may be prepared by placing 0.6947 g sodium acetate in 500 mL
deionized water.
3.3.2 Bracket sample concentrations with working standard
concentrations. If sample concentrations are higher than the
concentration range of prepared standards, either analyze higher
standards, or dilute the sample. The higher standards should be at
least as high in concentration as the highest sample. Diluted samples
should be prepared with extracting solvent to obtain a concentration
within the existing standard range. Prepare dilutions of the stock
standards in the concentration range of 0.1 to 200 µg/mL
for analysis with the 0.01 N NaOH solution used for extracting the
samples. The same reagent solution should be used to prepare samples
and standards, matrix matching, as the retention time of the acetate
peak is affected by the matrix concentration.
3.4 Sample preparation
3.4.1 Remove the plastic end caps from the sample tubes and
carefully transfer the adsorbent sections to separate 20-mL vials.
Discard the glass tube, urethane foam plug and glass wool plug.
3.4.2 Add 10 mL of 0.01 N NaOH to each vial using a pipettor or
volumetric pipet.
3.4.3 Immediately seal the vials with caps.
3.4.4 Shake the vials on shaker for 30 minutes. Spin down the
charcoal on a centrifuge for 5 min at about 2800 rpm, or allow to
settle for at least two hours.
3.4.5 Transfer supernatant to autosampler vials for analysis, being
careful not to transfer any particles of charcoal, as the particles
may clog the autosampler or instrument.
3.5 Analysis
3.5.1 Ion chromatograph conditions.
|
IC conditions columns: |
IonPac AS4A column 250-mm × 4-mm i.d. and IonPac
AG-4A guard column 50-mm × 4-mm i.d. at 30ºC |
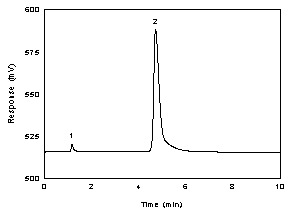
Figure 3.5.1 A chromatogram of 203 µg/mL acetic acid (200 µg/mL
acetate ion) in 0.01 N NaOH. (Key: (1) water; (2) acetate ion.) |
| flow rate: |
1.4 mL/min |
| eluent: |
1.5 mM Na2B4O7
|
| pump pressure: |
1200psi |
| injection size: |
50 µL
|
| retention time: |
4.8 min acetate ion |
3.5.2 Peak areas are measured by an integrator or other suitable
means.
3.5.3 An external standard (ESTD) calibration method is used. A
calibration curve can be constructed by plotting response of standard
injections versus milligrams of analyte per sample. Bracket the samples
with freshly prepared analytical standards over a range of
concentrations.

Figure 3.5.3. Calibration curve of acetic acid.
(Y=8.32x105 x+ 2.20x104). |
3.6 Interferences (analytical)
3.6.1 Any compound that produces a IC response and has a similar
retention time as the analyte is a potential interference. If any
potential interferences were reported, they should be considered before
samples are extracted. Generally, chromatographic conditions can be
altered to separate an interference from the analyte.
Butyric acid, as
the butyrate ion, is an interference on the AS4A analytical column,
therefore an AS14A analytical column should be used to analyze samples
from workplaces where butyric acid is present.
3.6.2 When necessary, the identity or purity of an analyte peak may
be confirmed by mass spectrometry or by another analytical procedure.
The sample must be acidified with sulfuric acid or phosphoric acid to a
pH of 3 or less, to reform the acetic acid from the acetate ion, before
it can be confirmed by GC mass spec. The mass spectrum in Figure 3.6.2.
Confirmation of the acetate ion is also possible by analysis by
capillary electrophoresis or by a second column on IC.
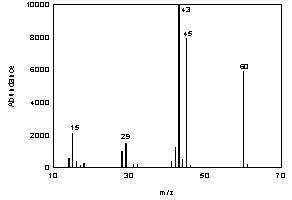
Figure 3.6.2. The mass spectrum of acetic acid. |
3.7 Calculations
The amount of analyte per sampler is obtained from the appropriate
calibration curve in terms of micrograms per sample, uncorrected for
extraction efficiency. If the instrument was calibrated on the concentration
of the acetate ion, the results need to be converted to acetic acid
concentration by multiplying the acetate ion concentration by a ratio of
their molecular weights (60.05/59.04). This total amount is then corrected
by subtracting the total amount (if any) found on the blank. The air
concentration is calculated using the following formulas.
|
|
where: |
CM is concentration by weight (mg/m³) |
|
M is micrograms per sample |
|
EE is extraction efficiency, in decimal form |
|
V is liters of air sampled
|
|
|
|
|
|
where: |
where CV is concentration by volume
(ppm)
|
|
VM is molar volume at 25º C
and 1 atm = 24.46
|
|
CM is concentration by weight
|
|
MR is molecular weight = 60.05
|
4. Recommendations for Further Study
Collection, reproducibility, and other detection limit studies need to be
performed to make this a validated method.
1 OSHA Sampling and Analytical
Methods. http://www.osha.gov
(accessed 6/21/02).
2 Lewis, R., Sax’s Dangerous Properties of Industrial Materials,
Tenth Ed., Vol. 2, Van Nostrand Reinhold: New York, 2000, p15.
3 O’Neil, M.J., Ed, The Merck index, Merck & Co. Inc.
Whitehouse Station, NJ, 2001, p 56.
4 ChemExpo Chemical Profiles. http: www.englib.cornell.edu
(accessed 6/21/02).
5 O’Neil, M.J., Ed, The Merck index, Merck & Co. Inc.
Whitehouse Station, NJ, 2001, p 56.
6 Lewis, R., Sax’s Dangerous Properties of Industrial Materials,
Tenth Ed., Vol. 2, Van Nostrand Reinhold: New York, 2000, p15.
7 OSHA Chemical Sampling Information. http://www.osha.gov (accessed
6/21/02).
8 Burright, D.; Chan, Y.; Eide, M.; Elskamp, C.; Hendricks, W.; Rose,
M. C. Evaluation Guidelines for Air Sampling Methods Utilizing
Chromatographic Analysis; OSHA Salt Lake Technical Center, U.S. Department
of Labor: Salt Lake City, UT, 1999.
|
| |
|
|
