Polychlorinated biphenyls (PCBs), polychlorinated dibenzo-
p-dioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs) (the latter two termed dioxins) are lipophilic and bioaccumulating environmental pollutants that are known for their neurotoxic effects in animals and humans. These fat-soluble toxicants are present in human beings and cross the placenta during pregnancy, thereby exposing children during the rapid development of the central nervous system (CNS). In the last two decades, several prospective epidemiologic studies in industrialized countries have shown subtle effects of exposure to background levels of PCBs and dioxins on health, growth, and development in children [reviewed in Weisglas-Kuperus (
1) and Brouwer et al. (
2)]. Many systems of the developing CNS may be affected by the neurotoxic effects of prenatal exposure to PCBs and dioxins (
2). One property of PCBs and dioxins and their metabolites is the modulation of the endocrine system, including sex-steroid hormones such as estrogens and androgens (
3,4). Steroid hormones play an important mediating role in the development of the CNS and influence not only reproductive but nonreproductive behaviors that show sex differences (
5,6). In evaluation of steroid hormone disrupting effects of PCBs and dioxins, effects on sexual dimorphic neurobehavior may therefore be important end points. Moreover, prenatal sex difference in sex-steroid hormone metabolism could cause sex differences in endocrine disrupting effects of PCBs and dioxins.
Sex-specific effects of perinatal PCB and dioxin exposure have been reported in animal studies on sexual dimorphic neurobehaviors such as sweet preference (7,8) and spatial learning (9,10). In humans, sex-specific neurobehavioral effects of prenatal exposure to PCBs and dioxins have been described only for the Yu-Cheng accident (11). In this cohort of children born to mothers who were accidentally exposed to high levels of PCBs and PCDFs in rice oil, cognitive (predominantly spatial) abilities were more affected by prenatal exposure to PCBs/PCDFs in boys than in girls.
In animal studies, nonreproductive behaviors that were altered by gonadal steroids include spatial and visual discrimination learning (12,13), open field exploration (14), and rough and tumble play (15). Especially behaviors that show sex differences were altered by gonadal steroids, whereas no such effect has been reported on behaviors that do not show sex differences. In humans, childhood play behavior shows marked sexual dimorphic differences and gives the clearest evidence for prenatal hormonal influence on human behavioral development (16).
In the Netherlands, a cohort of children born healthy has been prospectively followed from birth to school age to address neurotoxic effects of perinatal exposure to PCBs and dioxins. In this cohort, prenatal PCB exposure was related to lower psychomotor scores at 3 months of age (17) and lower cognitive abilities at 42 months (18). At school age, lower parental and home characteristics were associated with negative effects of prenatal PCB exposure on cognitive and motor development, whereas in children raised in relatively more privileged environments these subtle effects of prenatal PCB exposure were not detectable (19).
As part of the follow-up assessment at school age, we measured gender-role play in the Rotterdam cohort, half of the Dutch PCB and dioxin population. Our aim in this study was to evaluate effects of perinatal exposure to PCBs and dioxins on play behavior and whether these effects show sex differences.
Subjects and study design. The study population consisted of 207 healthy Caucasian mother-infant pairs who were recruited from June 1990 to February 1992 in the area of Rotterdam, in the Netherlands. The study design and recruitment process, chemical analysis, and PCB and dioxin concentrations have been described in detail elsewhere (
20). Pregnancy and delivery were uncomplicated. Only first- or second-born children, born healthy at term, were included. Half of the group of children was breast-fed (BF) (
n = 105) for at least 6 weeks; the others were formula-fed (FF) (
n = 102) during infancy. All FF infants received formula from a single batch (Almiron M2; Nutricia NV, Zoetermeer, The Netherlands) from birth until 7 months of age. In this formula, PCBs and dioxins were not detectable. The medical ethics committee of the University Hospital Rotterdam/Sophia Children's Hospital approved the study design, and the parents gave informed consent.
Assessment of exposure variables. Plasma samples were collected from the mothers during the last month of pregnancy, and cord plasma samples were collected directly after birth. These samples were analyzed for four PCB congeners: International Union for Pure and Applied Chemistry (IUPAC) numbers 118, 138, 153 and 180. Two weeks after delivery, a 24-hr representative breast-milk sample was collected from the mothers who were breast-feeding their children. Breast-milk samples were analyzed for 17 dioxins (PCDDs and PCDFs), 6 dioxin-like PCBs (3 planar PCBs and 3 mono-ortho PCBs), and 20 nondioxin-like PCBs. Toxic potency of the mixture of dioxins and dioxin-like PCBs was expressed by using the toxic equivalent (TEQ) approach (21).
We estimated prenatal exposure to PCBs in the total study population by using the sum of the four PCB congeners in maternal ( PCBmaternal) and in cord plasma (
PCBmaternal) and in cord plasma ( PCBcord). In the BF group additional prenatal exposure measurements were used: the
PCBcord). In the BF group additional prenatal exposure measurements were used: the  PCBmilk (the sum of PCB118, 138, 153, and 180), the dioxin, planar, mono-ortho, and total TEQ value (the sum of the TEQ values of the 17 dioxins and the 6 dioxin-like PCBs), and the
PCBmilk (the sum of PCB118, 138, 153, and 180), the dioxin, planar, mono-ortho, and total TEQ value (the sum of the TEQ values of the 17 dioxins and the 6 dioxin-like PCBs), and the  PCB20 nondioxin PCBs (the sum of 20 nondioxin-like PCBs).
PCB20 nondioxin PCBs (the sum of 20 nondioxin-like PCBs).
Postnatal exposure to PCBs and dioxins through lactation was estimated by multiplying the number of weeks of breast-feeding with, respectively,  PCBmilk; the dioxin, planar, mono-ortho, and total TEQ; and
PCBmilk; the dioxin, planar, mono-ortho, and total TEQ; and  PCB20 nondioxin PCBs concentrations in breast milk.
PCB20 nondioxin PCBs concentrations in breast milk.
Assessment of play behavior. Parents were asked to complete the Dutch version of the Pre-School Activities Inventory (PSAI)(22) (Appendix) when the children reached school age. This questionnaire, along with a questionnaire on problem behavior and a health questionnaire, was sent to the parents in two mailings, depending on the age of the child (in 1998 and 1999), near the end of a school year.
The PSAI is designed to discriminate play behavior both within and between the sexes. It consists of 24 questions addressing three aspects of play behavior: type of toys, activities, and child characteristics. Answers are given on a 5-point scale ranging from never to very often. The questions assess either feminine or masculine play behavior from which 3 scales are derived: a composite scale, integrating both masculine and feminine play behavior, and a masculine and a feminine scale. The composite scale is essentially defined as the difference: feminine scale minus masculine scale. A negative score on the composite scale implies masculine play behavior and a positive score feminine play behavior. A higher score on the feminine scale indicates more feminine play behavior, whereas a higher score on the masculine scale indicates more masculine play behavior.
The questionnaire has been validated in a group of preschool English children (n = 102); additionally, a test-retest reliability for the scores on the PSAI of 0.62 for boys and 0.66 for girls has been found (22). The PSAI has been assessed in various cohorts for standardization and norming purposes. These cohorts include normal preschool children across several samples in the United Kingdom [pilot study (n = 75); validation study (n = 102); and a cohort obtained through the magazine Practical Parenting (n = 1,643)], in the United States (n = 203), and also in the Netherlands, using a Dutch translation of the questionnaire (n = 341) (22).
Assessment of other variables. Variables that may influence child neurodevelopment have been assessed and include birth weight, duration of gestation, fetal exposure to alcohol and cigarette smoking, maternal age at birth of the child, parity, type of feeding during infancy, duration of breast-feeding, sex, and parental education level. The verbal IQ of the parent who spends the most time with the child (usually the mother) was assessed during the follow-up session at 42 months by two subtests, Information and Vocabulary from the Dutch version of the Wechsler Adult Intelligence Scale (WAIS) (23). At 7 years of age, follow-up assessment in this cohort was done at home by a psychologist (H.V.). During this visit, the child's home environment was assessed by the Home Observation for Measurement of the Environment (HOME) (24).
Data analysis. To compare groups for a single variable we used the Student's t-test (for continuous variables), the chi-square test (for categoric variables), or the Mann-Whitney U test. Plasma and milk PCB and dioxin values were positively skewed and were therefore normalized by natural logarithmic transformation. We studied the effects of PCB and dioxin exposure on the scores for the play behavior scales using multiple linear regression analyses (SPSS, version 9; SPSS, Chicago, IL, USA). Variables that were likely to affect play behavior were included in the regression model as a fixed set of variables. These variables were: sex (0/1 = boy/girl), highest education level of either parent [0/1/2 = low (primary school, secondary school not finished)/middle (secondary school finished)/high (high school finished, professional and university training)], parental verbal IQ, type of feeding during infancy (0/1 = BF or FF), duration of breast-feeding (0 for FF children), HOME score, and assessment age. Additionally, confounding variables, i.e., variables that correlated (p < 0.2), adjusted for the fixed set of variables, with one of the exposure variables and with scores on one of the three play behavior scales were included in the final regression model. Candidate confounders were alcohol use (0/1 = no/yes) and smoking (0/1 = no/yes) during pregnancy, duration of gestation, birth weight, maternal age at birth, and parity (0/1 = 1st/2nd born). This procedure resulted in the following regression model: sex, parental education level, parental verbal IQ, feeding type, duration of breast-feeding, HOME score, age at assessment, and parity. We studied sex differences in the effects of exposure to PCBs and dioxins by including an interaction term, the product of sex and exposure (sex*exposure), in the regression model. The effect of exposure on the outcome variables in boys and in girls, and the difference between these effects (girls minus boys) are estimated through the interaction term sex*exposure in essentially the same regression model by reparameterizing the sex effect. Results were considered significant if p 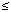 0.05.
0.05.
In the follow-up assessment at school age, 189 of the 207 children in the original cohort were re-examined and 160 of these parents returned the PSAI questionnaire (84% were filled out by mothers, 6% by fathers, and 10% by both parents). Two children were excluded from data analyses due to circumstances other than PCB and dioxin exposure that are likely to influence play behavior: a girl with Turner syndrome and a boy with a pervasive developmental disorder. Four questionnaires had missing data and were therefore excluded from data analyses.
Compared to the nonparticipating children (including both children who did not participate in the follow-up at school age and children whose questionnaires were not returned), prenatal PCB and dioxin exposure levels were comparable with the levels in children whose parents returned the questionnaire. Moreover, the distribution of children over the feeding groups in the participating group was not statistically different from that of the nonparticipating group (BF n = 20; FF n = 22). In regard to the other variables used in the regression model, these groups were also generally comparable except for the parental education levels (p = 0.011), parental verbal IQs (p = 0.009), and HOME scores (p = 0.021), which were higher in the participating group.
The mean age of the children at assessment was 7.5 (± 0.4) years old. The descriptives for the total study group, and for BF and FF boys and girls separately, are presented in Table 1. The characteristics of all boys and girls were not significantly different.
|
Table 1.

|
Comparing characteristics of boys and girls within feeding groups, the HOME score was significantly higher in BF girls than in BF boys. The HOME score and the parental education level were significantly higher in BF girls than in FF girls.
Boys and girls scored significantly different on the three PSAI scales [mean (SD) composite scale: boys -14.6 (5.8), girls 14.0 (5.3); masculine scale: boys 24.2 (5.3), girls 12.6 (4.5); feminine scale: boys 9.6 (3.3), girls 26.4 (6.2); all p-values < 0.001].
Table 2 presents effects of PCBs and dioxins on the PSAI scales for boys and girls including the sex difference in effect, adjusted for all other variables. Effects of prenatal exposure to PCBs on the scores on the composite scale and masculine scale were significantly different for boys and girls. In boys, higher prenatal PCB exposure was related with higher scores on the composite scale and lower scores on the masculine scale, both indicating less masculine play behavior. In girls, effects of prenatal PCB exposure moved in opposite directions on the composite and masculine scales; however, relations were not significant. We saw no sex-specific effects of prenatal PCB exposure on scores on the feminine scale. As an example of the relation between prenatal PCB exposure and play behavior in both sexes, adjusted for confounding variables, the relation between ln PCBcord and scores on the masculine scale are visualized in a partial regression plot (Figure 1).
PCBcord and scores on the masculine scale are visualized in a partial regression plot (Figure 1).
 |
Figure 1. Relation in boys (A) (r partial = -0.29) and in girls (B) (r partial = +0.17) between scores on the masculine scale and levels of ln PCBcord, adjusted for confounding variables; partial regression plot. PCBcord, adjusted for confounding variables; partial regression plot. |

In the BF group (Table 3), effects of  PCBmilk on the scores on the composite scale were also significantly different for boys and girls (p = 0.020). In girls, higher exposure to these compounds was related to lower scores (p = 0.028), indicating more masculine play behavior, whereas in boys the relation was in the opposite direction, although not significant (p = 0.369). We saw no sex-specific effects of
PCBmilk on the scores on the composite scale were also significantly different for boys and girls (p = 0.020). In girls, higher exposure to these compounds was related to lower scores (p = 0.028), indicating more masculine play behavior, whereas in boys the relation was in the opposite direction, although not significant (p = 0.369). We saw no sex-specific effects of  PCBmilk on scores on the masculine and feminine scale. Effects of prenatal levels of dioxin, planar and mono-ortho TEQs, total TEQ, and the sum of the 20 nondioxin-like PCBs on play behavior were not significantly different for boys and girls. Prenatal dioxin TEQ levels were significantly related with higher scores on the feminine scale in the total group of boys and girls (p = 0.048), indicating more feminized play behavior in both sexes.
PCBmilk on scores on the masculine and feminine scale. Effects of prenatal levels of dioxin, planar and mono-ortho TEQs, total TEQ, and the sum of the 20 nondioxin-like PCBs on play behavior were not significantly different for boys and girls. Prenatal dioxin TEQ levels were significantly related with higher scores on the feminine scale in the total group of boys and girls (p = 0.048), indicating more feminized play behavior in both sexes.
|
Table 3.

|
Postnatal exposure, through lactation, to  PCBmilk, dioxin, planar and mono-ortho TEQs, total TEQ, and
PCBmilk, dioxin, planar and mono-ortho TEQs, total TEQ, and  PCB20 nondioxin PCBs was not related to play behavior in the total BF group nor in boys and girls separately.
PCB20 nondioxin PCBs was not related to play behavior in the total BF group nor in boys and girls separately.
In this study we described sex-specific effects of prenatal exposure to PCBs on play behavior in healthy Dutch children at school age. Higher prenatal exposure to PCBs was associated with less masculinized play behavior in boys and with more masculinized play behavior in girls. Effects of prenatal exposure to dioxins were seen on feminine play behavior. In boys as well as in girls, higher prenatal dioxin levels were associated with more feminized play behavior. Childhood play behavior shows marked sex differences and is likely to be influenced by the prenatal steroid hormone environment. We therefore suggest that these results may indicate behavioral effects of steroid hormone imbalances early in development related to prenatal exposure to PCBs and dioxins, their metabolites, and/or related compounds.
In the Yu-Cheng cohort, researchers observed sex-specific effects of prenatal exposure to high levels of PCBs and PCDFs on the scores on the Raven's Colored Progressive Matrices (CPM) and Standardized Progressive Matrices (SPM) (11). These tests are considered to be tests for general cognitive development that appeal more on spatial rather than verbal capabilities. Spatial abilities form another domain of nonreproductive sex-specific behaviors that provide evidence for prenatal steroid hormone involvement. In the Yu-Cheng cohort, prenatally exposed boys were affected in their scores on the CPM and SPM tests, whereas in exposed girls no effect was seen. Because boys typically develop better spatial abilities than girls (25,26), these results were interpreted as demasculinizing or feminizing effects caused by disturbances in steroid hormones by prenatal exposure to PCBs/PCDFs (11). On the basis of results of play behavior studies in several groups of children that were prenatally exposed to abnormal levels of endogenous or exogenous steroid hormones, it has been hypothesized that there is evidence for prenatal androgen influences on sexual differentiation of childhood play (16). Masculinized or defeminized childhood play behavior was reported in genetic females who were exposed to elevated androgens (27,28), whereas demasculinized or feminized play behavior was associated with prenatal exposure to progestrogenic compounds that are assumed to interfere with androgen action in genetic females and, more subtly, in genetic males (29).
In adults prenatally exposed to diethylstilbestrol (DES)--a group that might be seen as a model group in studying potential estrogenic effects of prenatal PCB and dioxin exposure--childhood play behavior has been studied retrospectively. Males prenatally exposed to DES recalled slightly more masculinized play behavior than nonexposed controls, assessed by an interview covering childhood play behavior (30). In DES females no difference in childhood play, retrospectively assessed by questionnaires filled out by the DES subjects and their mothers, has been reported (31,32). The effects of prenatal exposure to PCBs and dioxins on childhood play behavior we reported in this study are opposite to the results of these DES studies. This difference in effect can be related to the retrospective nature of these DES studies and to differences in timing and duration of exposure to these chemicals in these groups. Moreover, differences in behavioral effects can be related to the level of exposure, which is likely to be higher in DES-exposed children. Many studies have reported that effects of exposure to hormones and hormone-mimicking chemicals show nonmonotonic dose-response curves, such as U-shaped or inverted U-shaped (33-37).
The current knowledge on the mechanisms of action of PCBs and dioxins and their metabolites, such as hydroxylated PCBs, on prenatal steroid hormone metabolism is still limited. Complex interactions with various steroid hormone systems are suggested, including estrogen and androgen hormone systems (3). These systems can be affected on various levels and estrogenic (38,39), antiestrogenic (8,40-42), and antiandrogenic (7) effects have been described in in vivo and in vitro studies, possibly depending on congener type or metabolites. In this study we lack information on prenatal steroid hormone levels, and although play behavior studies suggest that childhood play behavior is mediated predominantly by prenatal androgen action, our data are insufficient to show that multiple endocrine effects are not involved in the mechanism of action of prenatal exposure to PCBs and dioxins.
In the environment, PCBs and dioxins are present as complex mixtures of various congeners that may vary in metabolism, toxicity, and endocrine-disrupting properties. In this study we measured PCBs 118, 138, 153, and 180 in maternal and cord plasma samples. The sum of these four most abundant congeners constitutes 46% of the total PCBs (43). In the BF group various PCB and dioxin congeners were measured in breast milk. Prenatal levels of  PCBmilk were associated with masculine play behavior, similar to what was seen using maternal and cord
PCBmilk were associated with masculine play behavior, similar to what was seen using maternal and cord  PCB levels as prenatal exposure levels. Dioxin exposure was related with more feminine play behavior. Nondioxin-like PCB levels and dioxin-like PCB and total TEQ levels were not significantly associated with play behavior. Whether these results reflect effects that are specific to PCB or dioxin congeners or the limited power of analyses in this subgroup of BF children cannot be concluded from these results. Moreover, total TEQ levels, the sum of the nondioxin-like PCBs, and the four PCBs in breast milk and maternal and cord plasma correlated highly with each other (20).
PCB levels as prenatal exposure levels. Dioxin exposure was related with more feminine play behavior. Nondioxin-like PCB levels and dioxin-like PCB and total TEQ levels were not significantly associated with play behavior. Whether these results reflect effects that are specific to PCB or dioxin congeners or the limited power of analyses in this subgroup of BF children cannot be concluded from these results. Moreover, total TEQ levels, the sum of the nondioxin-like PCBs, and the four PCBs in breast milk and maternal and cord plasma correlated highly with each other (20).
Play behavior in our study was not associated with postnatal exposure to PCBs and dioxins through breast-feeding. We therefore suggest that childhood play behavior is sensitive to endocrine-disrupting behavioral effects of exposure to PCBs and dioxins early in development, as is supported in females with congenital adrenal hyperplasia (28) and by studies in other mammals (44,45).
In conclusion, this is the first behavioral study in humans to show effects of prenatal exposure to environmental levels of PCBs and dioxins on behavior that shows marked sex differences. Moreover, sex-specific effects of background prenatal exposure to PCBs have not been reported previously in human PCB studies. The results of this exploratory study give evidence for steroid hormone involvement in the neurotoxic mechanism of action of prenatal exposure to environmental levels of PCBs, dioxins, and other related organochlorine compounds. Evaluation of the relation between prenatal steroid hormone status and PCB and dioxin exposure is needed to further confirm these findings; in addition, follow-up of this cohort will be necessary to assess potential implications of these results on later development.
|
Pre-School Activity Inventory
copyright Susan Golombok and John Rust (22)
Name:
Age:
Sex: M/F (delete as appropriate)
Instructions
This inventory is about everyday activities of preschool children. It is in three sections: toy preferences, activities, and characteristics. Each question asks how frequently the child plays with particular toys, engages in particular activities or shows particular characteristics. There are five possible answers: (N) Never, (HE) Hardly Ever, (S) Sometimes, (O) Often, or (VO) Very Often. Answer each question by circling the response which best describes the child.
e.g., N HE S O VO
Please answer all of the questions. If you are unsure about which response best describes the child for any of the questions then please answer according to the response which seems most appropriate.
[Key: (N) Never, (HE) Hardly Ever, (S) Sometimes, (O) Often, or (VO) Very Often]
Part 1: TOYS: Please answer the questions according to how often the child played with the following toys during the past months.
|
1. Guns (or used objects as guns) | N | HE | S | O | VO |
|
2. Jewelry | N | HE | S | O | VO |
|
3. Tool set | N | HE | S | O | VO |
|
4. Dolls, doll's clothes, or doll's carriages | N | HE | S | O | VO |
|
5. Trains, cars, or airplanes | N | HE | S | O | VO |
|
6. Swords (or used objects as swords) | N | HE | S | O | VO |
|
7. Tea set N HE S O VO |
PART 2: ACTIVITIES: Please answer these questions according to how often the child engaged in the following activities during the past month.
|
1. Playing house (e.g., cleaning, cooking) | N | HE | S | O | VO |
|
2. Playing with girls | N | HE | S | O | VO |
|
3. Pretending to be a female character (e.g., princess) | N | HE | S | O | VO |
|
4. Playing at having a male occupation (e.g., soldier) | N | HE | S | O | VO |
|
5. Fighting | N | HE | S | O | VO |
|
6. Pretending to be a family character | N | HE | S | O | VO |
|
7. Sports and ball games | N | HE | S | O | VO |
|
8. Climbing (e.g., fences, trees, gym equipment) | N | HE | S | O | VO |
|
9. Playing at taking care of babies | N | HE | S | O | VO |
|
10. Showing all interest in real cars, trains, and equipment | N | HE | S | O | VO
|
|
11. Dressing up in girlish clothes | N | HE | S | O | VO |
PART 3: CHARACTERISTICS: Please answer questions according to how often the child shows the following characteristics:
|
1. Likes to explore new surroundings | N | HE | S | O | VO
|
|
2. Enjoys rough and tumble play | N | HE | S | O | VO
|
|
3. Shows interest in snakes, spiders, or insects | N | HE | S | O | VO
|
|
4. Avoids getting dirty | N | HE | S | O | VO
|
|
5. Likes pretty things | N | HE | S | O | VO
|
|
6. Avoids taking risks | N | HE | S | O | VO
|
NOW PLEASE CHECK THAT YOU HAVE ANSWERED ALL THE QUESTIONS
|
References and Notes
1. Weisglas-Kuperus N. Neurodevelopmental, immunological, and endocrinological indices of perinatal human exposure to PCBs and dioxins. Chemosphere 37:1845-1853 (1998).
2. Brouwer A, Ahlborg UG, Van den Berg M, Birnbaum LS, Boersma ER, Bosveld B, Denison MS, Gray LE, Hagmar L, Holene E, et al. Functional aspects of developmental toxicity of polyhalogenated aromatic hydrocarbons in experimental animals and human infants. Eur J Pharmacol 293:1-40 (1995).
3. Golden RJ, Noller KL, Titus-Ernstoff L, Kaufman RH, Mittendorf R, Stillman R, Reese EA. Environmental endocrine modulators and human health: an assessment of the biological evidence. Crit Rev Toxicol 28:109-227 (1998).
4. Vincent DR, Bradshaw WS, Booth GM, Seegmiller RE, Allen SD. Effect of PCB and DES on rat monoamine oxidase, acetylcholinesterase, testosterone, and estradiol ontogeny. Bull Environ Contam Toxicol 48:884-893 (1992).
5. Matsumoto A. Synaptogenic action of sex steroids in developing and adult neuroendocrine brain. Psychoneuroendocrinology 16:25-40 (1991).
6. Fitch RH, Denenberg VH. A role for ovarian hormones in sexual differentiation of the brain. Behav Brain Sci 21:311-327 (1998).
7. Hany J, Lilienthal H, Sarasin A, Roth-Harer A, Fastabend A, Dunemann L, Lichtensteiger W, Winneke G. Developmental exposure of rats to a reconstituted PCB mixture or aroclor 1254: effects on organ weights, aromatase activity, sex hormone levels, and sweet preference behavior. Toxicol Appl Pharmacol 158:231-243 (1999).
8. Amin S, Moore RW, Peterson RE, Schantz SL. Gestational and lactational exposure to TCDD or coplanar PCBs alters adult expression of saccharin preference behavior in female rats. Neurotoxicol Teratol 22:675-682 (2000).
9. Schantz SL, Moshtaghian J, Ness DK. Spatial learning deficits in adult rats exposed to ortho-substituted PCB congeners during gestation and lactation. Fundam Appl Toxicol 26:117-126 (1995).
10. Roegge CS, Seo BW, Crofton KM, Schantz SL. Gestational-lactational exposure to aroclor 1254 impairs radial-arm maze performance in male rats. Toxicol Sci 57:121-130 (2000).
11. Guo YL, Lai TJ, Chen SJ, Hsu CC. Gender-related decrease in Raven's progressive matrices scores in children prenatally exposed to polychlorinated biphenyls and related contaminants. Bull Environ Contam Toxicol 55:8-13 (1995).
12. Williams CL, Meck WH. The organizational effects of gonadal steroids on sexually dimorphic spatial ability. Psychoneuroendocrinology 16:155-176 (1991).
13. Bachevalier J, Hagger C. Sex differences in the development of learning abilities in primates. Psychoneuroendocrinology 16:177-188 (1991).
14. Steward J, Cygan D. Ovarian hormones act early in development to feminize adult open-field behavior in the rat. Horm Behav 14:20-32 (1980).
15. Ward IL, Stehm KE. Prenatal stress feminizes juvenile play patterns in male rats. Physiol Behav 50:601-605 (1991).
16. Collaer ML, Hines M. Human behavioral sex differences: a role for gonadal hormones during early development? Psychol Bull 118:55-107 (1995).
17. Koopman-Esseboom C, Weisglas-Kuperus N, de Ridder MA, Van der Paauw CG, Tuinstra LG, Sauer PJ. Effects of polychlorinated biphenyl/dioxin exposure and feeding type on infants' mental and psychomotor development. Pediatrics 97:700-706 (1996).
18. Patandin S, Lanting CI, Mulder PG, Boersma ER, Sauer PJ, Weisglas-Kuperus N. Effects of environmental exposure to polychlorinated biphenyls and dioxins on cognitive abilities in Dutch children at 42 months of age. J Pediatr 134:33-41 (1999).
19. Vreugdenhil HJ, Lanting CI, Mulder PG, Boersma ER, Weisglas-Kuperus N. Effects of prenatal PCB and dioxin background exposure on cognitive and motor abilities in Dutch children at school age. J Pediatr 140:48-56 (2002).
20. Koopman-Esseboom C, Huisman M, Weisglas-Kuperus N, Van der Pauw CG, Tuinstra LGMT, Boersma ER, Sauer PJ. PCB and dioxin levels in plasma and human milk of 418 Dutch women and their infant: predictive value of PCB congener levels in maternal plasma for fetal and infant's exposure to PCBs and dioxins. Chemosphere 28:1721-1732 (1994).
21. Van den Berg M, Birnbaum L, Bosveld ATC, Brunstrom B, Cook P, Feeley M, Giesy JP, Hanberg A, Hasegawa R, Kennedy SW, et al. Toxic equivalency factors (TEFs) for PCBs, PCDDs, PCDFs for humans and wildlife. Environ Health Perspect 106:775-792 (1998).
22. Golombok S, Rust J. The Pre-School Activities Inventory: a standardized assessment of gender role in children. Psychological Assessment 5:131-136 (1993).
23. Stinissen J, Willems PJ, Coetsier P, Hulsman WLL. Manual, Wechsler Adult Intelligence Scale (WAIS) [in Dutch]. Amsterdam:Swets & Zeitlinger NV, 1970.
24. Caldwell B, Bradley R. Home Observation of the Environment. Administration Manual. Little Rock, Arkansas: University of Arkansas, 1984.
25. Voyer D, Voyer S, Bryden MP. Magnitude of sex differences in spatial abilities: a meta-analysis and consideration of critical variables. Psychol Bull 117:250-270 (1995).
26. Linn MC, Petersen AC. Emergence and characterization of sex differences in spatial ability: a meta-analysis. Child Dev 56:1479-1498 (1985).
27. Dittmann RW, Kappes MH, Kappes ME, Borger D, Stegner H, Willig RH, Wallis H. Congenital adrenal hyperplasia. I. Gender-related behavior and attitudes in female patients and sisters. Psychoneuroendocrinology 15:401-420 (1990).
28. Berenbaum SA, Duck SC, Bryk K. Behavioral effects of prenatal versus postnatal androgen excess in children with 21-hydroxylase-deficient congenital adrenal hyperplasia. J Clin Endocrinol Metab 85:727-733 (2000).
29. Meyer-Bahlburg HF, Feldman JF, Cohen P, Ehrhardt AA. Perinatal factors in the development of gender-related play behavior: sex hormones versus pregnancy complications. Psychiatry 51:260-271 (1988).
30. Kester P, Green R, Finch SJ, Williams K. Prenatal "female hormone" administration and psychosexual development in human males. Psychoneuroendocrinology 5:269-285 (1980).
31. Lish JD, Ehrhardt AA, Meyer-Bahlburg HF, Rosen LR, Gruen RS, Veridiano NP. Gender-related behavior development in females exposed to diethylstilbestrol (DES) in utero: an attempted replication. J Am Acad Child Adolesc Psychiatry 30:29-37 (1991).
32. Lish JD, Meyer-Bahlburg HF, Ehrhardt AA, Travis BG, Veridiano NP. Prenatal exposure to diethylstilbestrol (DES): childhood play behavior and adult gender-role behavior in women. Arch Sex Behav 21:423-441 (1992).
33. Calabrese EJ, Baldwin LA. The frequency of U-shaped dose responses in the toxicological literature. Toxicol Sci 62:330-338 (2001).
34. Calabrese EJ, Baldwin LA. U-shaped dose-responses in biology, toxicology, and public health. Annu Rev Public Health 22:15-33 (2001).
35. vom Saal FS, Timms BG, Montano MM, Palanza P, Thayer KA, Nagel SC, Dhar MD, Ganjam VK, Parmigiani S, Welshons WV. Prostate enlargement in mice due to fetal exposure to low doses of estradiol or diethylstilbestrol and opposite effects at high doses. Proc Natl Acad Sci USA 94:2056-2061 (1997).
36. Palanza P, Parmigiani S, Liu H, vom Saal FS. Prenatal exposure to low doses of the estrogenic chemicals diethylstilbestrol and o,p'-DDT alters aggressive behavior of male and female house mice. Pharmacol Biochem Behav 64:665-672 (1999).
37. Palanza P, Parmigiani S, vom Saal FS. Effects of prenatal exposure to low doses of diethylstilbestrol, o,p'DDT, and methoxychlor on postnatal growth and neurobehavioral development in male and female mice. Horm Behav 40:252-265 (2001).
38. Bitman J, Cecil HC. Estrogenic activity of DDT analogs and polychlorinated biphenyls. J Agric Food Chem 18:1108-1112 (1970).
39. Korach KS, Sarver P, Chae K, McLachlan JA, McKinney JD. Estrogen receptor-binding activity of polychlorinated hydroxybiphenyls: conformationally restricted structural probes. Mol Pharmacol 33:120-126 (1988).
40. Moore M, Mustain M, Daniel K, Chen I, Safe S, Zacharewski T, Gillesby B, Joyeux A, Balaguer P. Antiestrogenic activity of hydroxylated polychlorinated biphenyl congeners identified in human serum. Toxicol Appl Pharmacol 142:160-168 (1997).
41. Kramer VJ, Helferich WG, Bergman A, Klasson-Wehler E, Giesy JP. Hydroxylated polychlorinated biphenyl metabolites are anti-estrogenic in a stably transfected human breast adenocarcinoma (MCF7) cell line. Toxicol Appl Pharmacol 144:363-376 (1997).
42. Jansen HT, Cooke PS, Porcelli J, Liu TC, Hansen LG. Estrogenic and antiestrogenic actions of PCBs in the female rat: in vitro and in vivo studies. Reprod Toxicol 7:237-248 (1993).
43. Jacobson JL, Humphrey HE, Jacobson SW, Schantz SL, Mullin MD, Welch R. Determinants of polychlorinated biphenyls (PCBs), polybrominated biphenyls (PBBs), and dichlorodiphenyl trichloroethane (DDT) levels in the sera of young children. Am J Public Health 79:1401-1404 (1989).
44. Goy RW, McEwen BS. Sexual Differentiation of the Brain. Cambridge, MA:MIT Press, 1980.
45. Wallen K. Nature needs nurture: the interaction of hormonal and social influences on the development of behavioral sex differences in rhesus monkeys. Horm Behav 30:364-378 (1996).
Last Updated: September 16, 2002






