
NTP Study Reports

NTP Study Reports
Home » Study Results & Research Projects » NTP Study Reports » All Long-Term Reports » Abstract for TR-464 - Sodium Xylenesulfonate (Technical Grade)
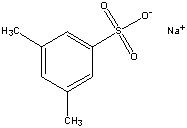
| Chemical Formula: (CH3)2C6H3SO3 - Na + - 3D Structure* | ||
|---|---|---|
| *To view structure, download free Chemscape Chime Plug-in | ||
Sodium xylenesulfonate
is used as a hydrotrope, an organic compound that increases the
ability of water to dissolve other molecules. Sodium xylenesulfonate
is a component in a variety of widely used shampoos and liquid
household detergents where it can constitute up to 10% of the
total solution. Because of its widespread use, the potential for
human exposure to sodium xylenesulfonate is great. Male and female
F344/N rats and B6C3F1 mice were administered sodium
xylenesulfonate in water or 50% ethanol dermally for 17 days,
14 weeks, or 2 years. Genetic toxicology studies were conducted
in Salmonella typhimurium, L5178Y mouse lymphoma cells,
and cultured Chinese hamster ovary cells.
Groups
of five male and five female rats were administered 300 mL of
0, 5, 15, 44, 133, or 400 mg/mL sodium xylenesulfonate in distilled
water by dermal application 5 days per week for 17 days. All rats
survived to the end of the study. Final mean body weights and
body weight gains of dosed rats were similar to those of the control
groups. Dermal applications of 300 mL of 5, 15, 44, 133, and 400
mg/mL delivered average daily doses of approximately 10, 30, 90,
260, and 800 mg sodium xylenesulfonate/kg body weight to males
and 13, 40, 120, 330, and 1,030 mg/kg to females. Clinical findings
generally involved the skin of dosed animals and included tan
or brown skin discoloration and crusty white deposits (presumed
to be dried chemical) at the site of application. Neither of these
observations were considered significant findings. The relative
liver weights of 133 and 400 mg/mL male and female rats were significantly
greater than those of the control groups, but the absolute liver
weights were not increased and the biological significance of
the relative differences in liver weight was unclear. In males
and females, the few lesions observed grossly and microscopically
were generally attributed to repeated clipping and were not considered
related to chemical administration.
Groups
of five male and five female mice were administered 100 mL of
0, 5, 15, 44, 133, or 400 mg/mL sodium xylenesulfonate in distilled
water by dermal application 5 days per week for 17 days. All mice
survived to the end of the study. Final mean body weights and
body weight gains of dosed mice were similar to those of the controls.
Dermal applications of 5, 15, 44, 133, and 400 mg/mL delivered
average daily doses of approximately, 20, 60, 190, 540, and 1,600
mg sodium xylenesulfonate/kg body weight to males and 26, 80,
220, 680, and 2,000 mg/kg to females. Clinical findings included
crusty white deposits (presumed to be dried chemical) at the site
of application in two 133 mg/mL males and in all 400 mg/mL males
and females. The absolute and relative liver weights of 15 and
44 mg/mL males and 400 mg/mL males and females were significantly
greater than those of the control groups, but the biological significance
of these differences was unclear. The few skin lesions observed
grossly and microscopically in males and females were generally
attributed to repeated clipping and were not considered related
to chemical administration.
Groups
of 10 male and 10 female rats were administered 300 mL of 0, 5,
15, 44, 133, or 400 mg/mL sodium xylenesulfonate in 50% ethanol
by dermal application for 14 weeks. For special hematology and
clinical pathology studies, additional groups of 10 male and 10
female rats were administered 0, 5, 15, 44, 133, or 400 mg/mL
sodium xylenesulfonate in 50% ethanol by dermal application for
14 weeks. All rats survived to the end of the study. Final mean
body weights and body weight gains of dosed male and female rats
were similar to those of the control groups. Dermal applications
of 5, 15, 44, 133, and 400 mg/mL delivered average daily doses
of approximately 6, 20, 60, 170, and 500 mg sodium xylenesulfonate/kg
body weight to males and 10, 30, 90, 260, and 800 mg/kg to females.
The only notable clinical finding was brown discoloration of the
skin at the site of application in dosed animals. Hematology and
clinical chemistry parameters of dosed groups of males and females
were significantly different from those of the controls in several
instances, but these differences were sporadic and did not demonstrate
a treatment relationship. The absolute and relative liver weights
of males receiving 44, 133, or 400 mg/mL were significantly less
than those of the control group, but the biological significance
of these differences was unclear, and there were no treatment-related
histopathologic effects in the liver. There were no significant
differences in liver weights in female rats.
Minimal hyperplasia
of the epidermis at the site of application occurred in both male
and female rats in the control group as well as most dosed groups.
The incidence of epidermal hyperplasia in 400 mg/mL males was
possibly chemical related.
Groups of 10 male and 10 female mice were administered 100 mL of 0, 5, 15, 44, 133, or 400 mg/mL sodium xylenesulfonate in 50% ethanol by dermal application for 14 weeks. There were no chemical-related deaths. The mean body weight gain of the 400 mg/mL males was significantly greater than that of the control group. Dermal applications of 5, 15, 44, 133, and 400 mg/mL delivered average daily doses of approximately 17, 40, 140, 440, and 1,300 mg sodium xylenesulfonate/kg body weight to males and 20, 60, 170, 530, and 1,630 mg/kg to females. There were no clinical findings related to sodium xylenesulfonate administration.
Epidermal hyperplasia occurred in one 44 mg/mL female, two 133 mg/mL males, five 400 mg/mL males, and four 400 mg/kg females. Hyperplasia of the epidermis in 400 mg/mL males and females was probably related to chemical administration.
Chronic inflammation of the skin occurred primarily in the control groups of males and females. These lesions consisted of mononuclear inflammatory cells in the dermis.
Groups of 50 male and 50 female rats were dermally administered 0, 60, 120, or 240 mg sodium xylenesulfonate/kg body weight in 50% ethanol for 104 weeks.
Survival
of dosed males and females was similar to that of the control
groups. Mean body weights of dosed males and females were similar
to those of the controls throughout
the study. In male groups, there were no clinical findings considered
treatment related. In females, clinical findings were limited
to irritation at the site of application in one control female,
four 120 mg/kg females, and two 240 mg/kg females.
There were no neoplasms at any site (including the skin) that were considered treatment related.Low incidences of hyperplasia of the epidermis at the site of application occurred in males in the 60, 120, and 240 mg/kg groups. Low incidences of hyperplasia of the epidermis at the site of application also occurred in females in the 120 and 240 mg/kg groups, and they occurred with a significant positive trend. Low incidences of hyperplasia of the sebaceous gland occurred in control and 60 mg/kg males and in control, 120 mg/kg, and 240 mg/kg females.
Groups
of 50 male and 50 female mice were dermally administered 0, 182,
364, or 727 mg sodium xylenesulfonate/kg body weight in 50% ethanol
for 104 to 105 weeks.
Survival
of dosed males and females was similar to that of the control
groups. Mean body weights of dosed males and females were generally
similar to those of the controls throughout the study; however,
the mean body weights of 727 mg/kg females were greater than those
of the control group from week 85 to week 97. With the exception
of irritation at the site of application in one 364 mg/kg female,
there were no clinical findings related to sodium xylenesulfonate
administration.
There
were no neoplasms at any site (including the skin) that were considered
treatment related.Hyperplasia of the epidermis occurred in control,364
mg/kg, and 727 mg/kg males and in control and dosed females. In
male mice, the incidences occurred with a significant positive
trend. Focal ulceration occurred in one 727 mg/kg male and in
one female in each dose group. In males and females from control
and dosed groups, the incidences of hepatocellular adenoma, hepatocellular
carcinoma, and hepato- cellular adenoma or carcinoma (combined)
were generally higher than those expected by spontaneous occurrence.
The incidences of hepatocellular neoplasms in some groups of males
and females exceeded the NTP historical control range. Male mice
had a pattern of nonneoplastic liver lesions along with silver
stained positive helical organisms within the liver which suggests
an infection with Helicobacter hepaticus. The findings
in this study of sodium xylenesulfonate were not considered to
have been significantly impacted by the infection with H. hepaticus
or its associated hepatitis.
Sodium
xylenesulfonate was not mutagenic in Salmonella typhimurium
strain TA98, TA100, TA1535, or TA1537 with or without induced
liver S9. Equivocal results were obtained in a mutation assay
with mouse lymphoma cells in the presence of induced S9; no evidence
of mutagenicity was noted without S9 in this assay. In cytogenetic
tests with sodium xylenesulfonate in cultured Chinese hamster
ovary cells, significant increases in sister chromatid exchanges
were observed in the absence of S9 only, and no increases in chromosomal
aberrations were observed with or without S9.
Under the conditions of these 2-year dermal studies, there was no evidence of carcinogenic activity of sodium xylenesulfonate in male or female F344/N rats administered 60, 120, or 240 mg/kg or in male or female B6C3F1 mice administered 182, 364, or 727 mg/kg.
Increased incidences of epidermal hyperplasia in female rats and male mice may have been related to exposure to sodium xylenesulfonate.
Synonyms: Benzenesulfonic acid,
dimethyl-, sodium salt; xylenesulfonic acid, sodium salt; sodium
dimethylbenzenesulfonate; xylenesulfonic acid, sodium salt
Trade names: Conco SXS; Cyclophil;
SXS 30; Eletesol SX 30; Naxonate; Naxonate G; Richonate SXS; Stepanate
SXS; Stepanate X; SXS 40; Ultrawet 40SX
|
| ||||
| Male
F344/N Rats |
Female
F344/N Rats |
Male
B6C3F1 Mice |
Female
B6C3F1 Mice | |
|---|---|---|---|---|
| Doses | 0, 60, 120, or 240 mg/kg in 50% ethanol applied dermally | 0, 60, 120, or 240 mg/kg in 50% ethanol applied dermally | 0, 182, 364, or 727 mg/kg in 50% ethanol applied dermally | 0, 182, 364, or 727 mg/kg in 50% ethanol applied dermally |
| Body weights | Dosed groups similar to control group | Dosed groups similar to control group | Dosed groups similar to control group | Dosed groups similar to control group |
| 2-Year survival rates | 7/50, 17/50, 9/50, 10/50 | 22/50, 16/50, 17/50, 16/50 | 32/50, 37/50, 39/50, 35/50 | 31/50, 32/49, 32/50, 36/50 |
| Nonneoplastic effects | None | None | None | None |
| Neoplastic effects | None | None | None | None |
| Uncertain findings | None | Skin (site of application): epidermal hyperplasia (1/50, 0/50, 4/50, 5/50) | Skin (site of application): epidermal hyperplasia (1/50, 0/50, 4/50, 5/50) | None |
| Level of evidence of carcinogenic activity | No evidence | No evidence | No evidence | No evidence |
| Salmonella typhimuriumgene mutations: | Negative with and without S9 in strains TA98, TA100, TA1535, and TA1537 | |
| Mouse lymphoma mutagenicity | Equivocal with S9; negative without S9 | |
| Sister chromatid exchanges | ||
| Cultured Chinese hamster ovary cells in vitro: | Negative with S9; positive without S9 | |
| Chromosomal aberrations | ||
| Cultured Chinese hamster ovary cells in vitro: | Negative with and without S9 | |
Report Date: June 1998
Pathology Tables, Survival and Growth Curves from NTP 2-year Studies
Target Organs & Incidences from 2-year Studies
You may link to the full technical report in pdf format ( Note: A print ready copy of the document is presented in Portable Document Format (pdf) which requires the Acrobat Reader plug-in -- download a free copy of the reader.)
Web page last updated on October 11, 2007
The National Institute of Environmental Health Sciences is one of the National Institutes of Health within the U.S. Department of Health and Human Services. The National Toxicology Program is headquartered on the NIEHS campus in Research Triangle Park, NC.