| In Vitro Estrogenicity of Polybrominated Diphenyl Ethers, Hydroxylated PBDEs, and Polybrominated Bisphenol A Compounds Ilonka A.T.M. Meerts,1 Robert J. Letcher,2* Saske Hoving,1 Göran Marsh,3 Åke Bergman,3 Josephine G. Lemmen,4 Bart van der Burg,4 and Abraham Brouwer1,5 1Toxicology Group, Wageningen University and Research Center, Wageningen, The Netherlands
2Research Institute of Toxicology, Utrecht University, Utrecht, The Netherlands
3Department of Environmental Chemistry, Stockholm University, Stockholm, Sweden
4Hubrecht Laboratory, Netherlands Institute for Developmental Biology, Utrecht, The Netherlands
5Institute of Environmental Studies, Free University of Amsterdam, Amsterdam, The Netherlands Abstract
Polybrominated diphenyl ethers (PBDEs) are used in large quantities as additive flame retardants in plastics and textile materials. PBDEs are persistent compounds and have been detected in wildlife and in human adipose tissue and plasma samples. In this study, we investigated the (anti) estrogenic potencies of several PBDE congeners, three hydroxylated PBDEs (HO-PBDEs) , and differently brominated bisphenol A compounds in three different cell line assays based on estrogen receptor (ER) -dependent luciferase reporter gene expression. In human T47D breast cancer cells stably transfected with an estrogen-responsive luciferase reporter gene construct (pEREtata-Luc) , 11 PBDEs showed estrogenic potencies, with concentrations leading to 50% induction (EC50) varying from 2.5 to 7.3 µM. The luciferase induction of the most potent HO-PBDE [2-bromo-4-(2,4,6-tribromophenoxy) phenol] exceeded that of estradiol (E2) , though at concentrations 50,000 times higher. As expected, brominated bisphenol A compounds with the lowest degree of bromination showed highest estrogenic potencies (EC50 values of 0.5 µM for 3-monobromobisphenol A) . In an ER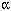 -specific, stably transfected human embryonic kidney cell line (293-ER -specific, stably transfected human embryonic kidney cell line (293-ER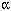 -Luc) , the HO-PBDE 4-(2,4,6-tribromophenoxy) phenol was a highly potent estrogen with an EC50 < 0.1 µM and a maximum 35- to 40-fold induction, which was similar to E2. In an analogous ERß-specific 293-ERßs-Luc cell line, the agonistic potency of the 4-(2,4,6-tribromophenoxy) phenol was much lower (maximum 50% induction compared to E2) , but EC50 values were comparable. These results indicate that several pure PBDE congeners, but especially HO-PBDEs and brominated bisphenol A-analogs, are agonists of both ER -Luc) , the HO-PBDE 4-(2,4,6-tribromophenoxy) phenol was a highly potent estrogen with an EC50 < 0.1 µM and a maximum 35- to 40-fold induction, which was similar to E2. In an analogous ERß-specific 293-ERßs-Luc cell line, the agonistic potency of the 4-(2,4,6-tribromophenoxy) phenol was much lower (maximum 50% induction compared to E2) , but EC50 values were comparable. These results indicate that several pure PBDE congeners, but especially HO-PBDEs and brominated bisphenol A-analogs, are agonists of both ER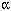 and ERß receptors, thus stimulating ER-mediated luciferase induction in vitro. These data also suggest that in vivo metabolism of PBDEs may produce more potent pseudoestrogens. Key words: ER-CALUX, estrogenicity, flame retardants, hydroxylated compounds, polybrominated diphenyl ethers. Environ Health Perspect 109:399-407 (2001) . [Online 27 March 2001] and ERß receptors, thus stimulating ER-mediated luciferase induction in vitro. These data also suggest that in vivo metabolism of PBDEs may produce more potent pseudoestrogens. Key words: ER-CALUX, estrogenicity, flame retardants, hydroxylated compounds, polybrominated diphenyl ethers. Environ Health Perspect 109:399-407 (2001) . [Online 27 March 2001] http://ehpnet1.niehs.nih.gov/docs/2001/109p399-407meerts/ abstract.html Address correspondence to I.A.T.M. Meerts, NOTOX Safety & Environmental Research B.V., Hambakenwetering 7, 5231 DD's-Hertogenbosch, The Netherlands. Telephone: +31-73-6406700. Fax: +31 73 6406799. E-mail: ilonka.meerts@notox.nl *R.J. Letcher is currently at the Great Lakes Institute for Environmental Research, University of Windsor, 304 Sunset Avenue, Windsor, Ontario, N8B 3P4 Canada. This research was supported financially by the European Commission, Environment and Climate Program (grants ENV-CT96-0170 and ENV-CT96-0204) . Received 11 April 2000 ; accepted 25 October 2000.
The full version of this article is available for free in HTML or PDF formats. |

