 |
|
Molecular & Cellular Biology Group
Considerable sex differences exist in the risk, incidence and pathogenesis of lung diseases in humans (Figure 1). Since researchers use mouse models to study basic pathogenic mechanisms and potential therapies for a variety of human lung diseases, it is important to recognize factors that might influence experimental endpoints of interest. Therefore, the roles of sex hormones have received significant attention, particularly with respect to their effects on lung function and the response of the lung to a variety of environmental agents. 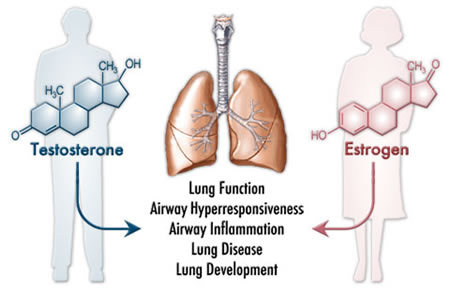 Sex hormones and basal lung functionThe group has documented that basal lung function in naïve male and female mice does not differ substantially, but that male mice have greater airway responsiveness to methacholine aerosol than female mice (Card et al, J Immunol, 2006 (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=pubmed&cmd=Retrieve&dopt=AbstractPlus&list_uids=16785560&query_hl=51&itool=pubmed_docsum) Sex hormones and non-allergic airway inflammationMale mice demonstrated greater airway inflammation and hyperresponsiveness than females following exposure to bacterial lipopolysaccharide (Card et al., J. Immunol., 2006 (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=pubmed&cmd=Retrieve&dopt=AbstractPlus&list_uids=16785560&query_hl=51&itool=pubmed_docsum) Sex hormones and allergic airway inflammationIn a model of allergic airway inflammation, estrogen receptor-α knockout mice had comparable inflammation, but considerably greater airway responsiveness to methacholine than wild-type mice. This data suggests that estrogen receptor-α plays an important regulatory effect on airway responsiveness in the allergic lung (Carey et al., Am. J. Respir. Crit. Care Med., 2006 (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=pubmed&cmd=Retrieve&dopt=AbstractPlus&list_uids=17095746&query_hl=51&itool=pubmed_docsum) Future studiesThe group has identified considerable sex differences in murine airway responsiveness and lung disease progression and will continue to investigate sex hormone-mediated regulation of lung function in naïve mice and the influence of sex hormones on disease pathogenesis. The analyses will use a variety of murine models of lung disease including non-allergic and allergic inflammation, pulmonary fibrosis and others and will provide important information regarding the influence of sex hormones in relevant murine models of lung disease and dysfunction. In addition, the identification of sex differences in lung function or disease processes in experimental models may lead to improved clinical treatment of lung diseases. The group will focus on the following goals:
|
|

