
| |  | |  |
Children's Health
|
| Decline of Ambient Air Pollution Levels and Improved Respiratory Health in Swiss Children Lucy Bayer-Oglesby,1 Leticia Grize,1 Markus Gassner,2 Kathy Takken-Sahli,3 Felix H. Sennhauser,4 Urs Neu,5 Christian Schindler,1 and Charlotte Braun-Fahrländer1 1Institute of Social and Preventive Medicine of the University of Basel, Basel, Switzerland; 2School Health Service, Grabs, Switzerland; 3School Health Service, Zürich, Switzerland; 4University Children's Hospital, Zürich, Switzerland; 5Institute of Geography, University of Bern, Bern, Switzerland Abstract
The causality of observed associations between air pollution and respiratory health in children is still subject to debate. If reduced air pollution exposure resulted in improved respiratory health of children, this would argue in favor of a causal relation. We investigated whether a rather moderate decline of air pollution levels in the 1990s in Switzerland was associated with a reduction in respiratory symptoms and diseases in school children. In nine Swiss communities, 9,591 children participated in cross-sectional health assessments between 1992 and 2001. Their parents completed identical questionnaires on health status and covariates. We assigned to each child an estimate of regional particles with an aerodynamic diameter < 10 µg/m3 (PM10) and determined change in PM10 since the first survey. Adjusted for socioeconomic, health-related, and indoor factors, declining PM10 was associated in logistic regression models with declining prevalence of chronic cough [odds ratio (OR) per 10-µg/m3 decline = 0.65, 95% confidence interval (CI) , 0.54-0.79], bronchitis (OR = 0.66 ; 95% CI, 0.55-0.80) , common cold (OR = 0.78 ; 95% CI, 0.68-0.89) , nocturnal dry cough (OR = 0.70 ; 95% CI, 0.60-0.83) , and conjunctivitis symptoms (OR = 0.81 ; 95% CI, 0.70-0.95) . Changes in prevalence of sneezing during pollen season, asthma, and hay fever were not associated with the PM10 reduction. Our findings show that the reduction of air pollution exposures contributes to improved respiratory health in children. No threshold of adverse effects of PM10 was apparent because we observed the beneficial effects for relatively small changes of rather moderate air pollution levels. Current air pollution levels in Switzerland still exceed limit values of the Swiss Clean Air Act ; thus, children's health can be improved further. Key words: air pollution, children, cross-sectional surveys, decline , respiratory health, symptoms. Environ Health Perspect 113:1632-1637 (2005) . doi:10.1289/ehp.8159 available via http://dx.doi.org/ [Online 21 June 2005]
Address correspondence to L. Bayer-Oglesby, Institute of Social and Preventive Medicine, University of Basel, Steinengraben 49, CH-4051 Basel, Switzerland. Telephone: 41-61-267-60-66. Fax: 41-61-267-61-90. E-mail: lucy.oglesby@unibas.ch We thank the School Health Services for organizing the survey ; the children, parents, and teachers for their cooperation ; and J. Schwartz and N. Künzli for valuable comments. This study received financial support from the Swiss National Science Foundation (grant 4026-033109) , the Swiss Federal Office of Public Health, the Swiss Agency for the Environment, Forest and Landscape (FE/SAEFL 2000.I.08, FE/SAEFL 810.98.50) , the Lung Associations Zürich and St. Gallen, and the cantonal health services of Zürich, St. Gallen, Valais, Vaud, Geneva, and Bern. The authors declare they have no competing financial interests. Received 1 April 2005 ; accepted 21 June 2005. |
|
|
 |
The causality of observed associations between air pollution and respiratory health in children is still subject to debate, although numerous studies have reported adverse effects of air pollution on the respiratory health of children, using indicators of general air pollution (Braun-Fahrländer et al. 1997; Chen et al. 1998; Gauderman et al. 2002; Horak et al. 2002; Hruba et al. 2001; McConnell et al. 1999) and of traffic-related air pollution (Brauer et al. 2002; Gehring et al. 2002; Hirsch et al. 1999; Janssen et al. 2003; Nicolai et al. 2003; van Vliet et al. 1997; Venn et al. 2001; Wjst et al. 1993). If it could be shown that reduced air pollution exposures improve the respiratory health of children, this would argue in favor of a causal relation. So far, only a few studies have investigated the expected beneficial effects of air pollution reduction on respiratory health in children. In cross-sectional analysis, the tremendous decline of coal combustion-related air pollution in East Germany after reunification was associated with a decline of respiratory symptoms (Heinrich 2003) and improved lung function (Frye et al. 2003) in children. In a cohort of children, those who moved within California to areas with lower PM10 (particles with an aerodynamic diameter < 10 µg/m3) levels showed increased lung function growth, whereas those moving to more polluted areas had a decreased growth (Avol et al. 2001). McConnell et al. (2003) observed that bronchitis symptoms, assessed yearly for 4 years in a cohort of children with asthma, varied with the yearly variability of PM2.5 (particles with an aerodynamic diameter < 2.5 µg/m3), nitrogen dioxide, and organic carbon.
In the first cross-sectional assessment of the Swiss Surveillance Program of Childhood Allergy and Respiratory Symptoms with Respect to Air Pollution and Climate (SCARPOL) in 1992-1993, Braun-Fahrländer et al. (1997) reported that rates of respiratory symptoms and diseases, adjusted for individual risk factors, were positively associated with PM10, NO2, and sulfur dioxide in children living in 10 urban, suburban, rural, and alpine areas of Switzerland. Since then, air pollution abatement measures (emission limits for industries, introduction of low-sulfur heating oil and catalytic converters) implemented after the Swiss Clean Air Act (1985) have led to declining air pollution levels in Switzerland [Swiss Agency for the Environment, Forest and Landscape(SAEFL) 2003; Kuebler et al. 2001]. In contrast to East Germany, where the tremendous air pollution decline in the 1990s went hand in hand with dramatic political and social changes, the political and social system in Switzerland has been very stable for many decades, which is an asset in our study. We hypothesize that if the health effects observed in SCARPOL in 1993 (Braun-Fahrländer et al. 1997) were causal, a) the observed reduction of PM10 in Switzerland since the first cross-section of SCARPOL would be associated with a reduction of prevalence rates of respiratory symptoms and diseases in the second health assessment phase, and b) the average reduction of symptom prevalence would be more pronounced in areas with stronger reduction of air pollution levels.
Table 1.

|
Table 2.

|
Study population and design.In 10 Swiss communities covering a broad range of urbanization, air pollution levels, and climatic conditions, 10,397 school children (76.1%) ages 6-15 years have participated in cross-sectional, questionnaire-based health assessments between 1992 and 2001. For urban areas, we chose Lugano, Zürich, Bern, and Geneva; for suburban areas, Anières and Biel; for rural areas, Langnau, Payerne, and Rheintal; and for an alpine area, Montana. Because of the absence of PM10 data, we had to exclude children of Rheintal for this analysis, resulting in a sample of 9,591 children. The detailed recruiting procedure for the first cross-sectional health assessment in 1992-1993, which has also been applied for subsequent assessments, has been described previously (Braun-Fahrländer et al. 1997). Children of three school grades (first, fourth, and eighth) were recruited in the first phase in 1992-1993; in the second phase, one grade was enrolled each school year (first grade in 1998-1999, eighth grade in 1999-2000, and fourth grade in 2000-2001) (Table 1). This resulted in two repeated cross-sectional surveys for each age-group that are 6, 7, and 8 years apart for the first, eighth and fourth grade, respectively. The ethics committees of the Universities of Geneva and Bern approved the study protocol.
Health assessment.For all participating children, we collected identical parent-completed questionnaires on health status, family history of disease, spare-time activities, indoor exposures, and residential situation. The questionnaire included the core questions on asthma and allergy of the International Study of Asthma and Allergy in Childhood (ISAAC) (Asher et al. 1995). Definitions of symptoms and diseases examined in this analysis are given in Table 2.
Assessment of air pollution exposures.We assigned to each child an estimate of regional PM10 for the year preceding the questionnaire date, obtained from one fixed monitoring station in each community. Children were living within a few (3-5) kilometers of the monitors. Monitors were located in the centers of the communities, with the exception of the rural monitors in Payerne and Montana. Röösli et al. (2000, 2001) have demonstrated that in Switzerland, PM10 levels are homogeneously distributed within regions and are not significantly affected by local traffic, justifying the single-monitor approach for the assignment of PM10 exposures. Because PM10 measurements started in four communities not before 1993, we assigned annual means of 1993 to all children participating in the first cross-section (1992-1993 school year). Annual means of PM10 have been estimated for 1993 and for 1997-2000. We converted Harvard Impactor data of 1993 to DIGITEL HiVol values based on collocated measurements of the two monitors for 24 months (Krütli and Monn 1999). Between 1997 and 2000, PM10 was measured in the nine regions with DIGITEL HiVol samplers (DIGITEL 1999). In addition, we obtained temperature measurements from the fixed monitoring stations for calculating the number of cold days (days with the maximum temperature below zero degrees Celsius) for each region and year.
Statistical analysis. To analyze the association between change of air pollution levels and change of respiratory health, we used multivariate logistic regression models. For the children participating in the second health assessment phase (school years 1998-1999, 1999-2000, and 2000-2001), change in PM10 was calculated as the difference between the assigned PM10 estimate and the 1993 baseline values corresponding to their area. For the children participating in the first health assessment phase (school year 1992-1993), change in PM10 was set to zero. In addition to change in PM10, a dummy variable for each region was included in the regression models. To test for community correlation possibly introduced by clustering of uncontrolled covariates, we also evaluated random-effect models.
For the nine health end points, we computed adjusted odds ratios (ORs) associated with a decline of 10 µg/m3 in PM10. A priori, our regression models also included those covariates that had an impact on the effect estimates or were identified as confounders of air pollution effects in the first cross-sectional analysis of 1992-1993 (Braun-Fahrländer et al. 1997). Covariates included
- Socioeconomic factors (age, sex, nationality, parental education, number of siblings, farming status)
- Health-related factors (low birth weight, breastfeeding, child who smokes, family history of asthma, bronchitis, and/or atopy)
- Indoor factors (mother who smokes, humidity, mode of heating and cooking, carpeting, pets allowed in bedroom)
- Avoidance behavior with respect to allergies (carpet or pets removed for health reasons)
- Questionnaire-related factors (person who completed questionnaire).
These covariates proved relevant in the multivariate model also for analyzing the impact of change of PM10 on respiratory symptoms. Age was included as a categoric variable (three groups according to school grades) because preliminary analysis suggested a nonlinear association between age and the evaluated health outcomes. In the first cross-section, all questionnaires were completed during wintertime to avoid confounding by season. The cross-sectional assessments of the second phase had to be spread over the whole school year for logistic reasons. A dummy variable for the month when the questionnaire was completed was included in the multivariate logistic regression models to adjust for possible reporting bias by season of the interview.
We evaluated whether secular trends had occurred between 1992-1993 and 1998-2001 that could be related to changing prevalence of the investigated symptoms and diseases--namely, climatic factors (milder or colder winters), participation rates, and mother's concern about an association between environmental exposure and children's respiratory health.
We further tested the final models for interactions between change of PM10 on the one hand and covariates such as age group, sex, family history of allergic diseases (asthma and/or atopy), asthma ever of child, smoker (child and/or mother), and indoor exposures (heating and/or cooking) on the other. The fit of the final models was evaluated.
To evaluate whether the average reduction of symptom prevalence is more pronounced in areas with stronger reduction of air pollution, we computed covariate-adjusted prevalence by community for the first (1992-1993) and second health assessment phase (1998-2001). To visualize the associations, we plotted the mean region-specific change in adjusted prevalence between the first and second phase against the respective mean change in PM10 levels. Corresponding Pearson correlation coefficients for the associations between these aggregate data were computed.
All analyses were conducted with Stata Statistical Software, Release 8.0.SE (StataCorp, College Station, TX, USA).
|
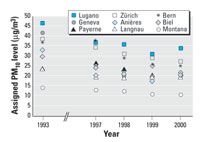
Figure 1. Annual means of PM10 levelsa assigned to children of the first (1993) and second (1997-2000) health assessment phase in nine SCARPOL regions.
aMeasured with DIGITEL HiVol Samplers. 1993 data converted from Harvard Impactor data.
|
Table 3.
|
Table 4.
|
|
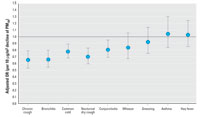
Figure 2. Adjusted ORsa and 95% CIs of symptoms and respiratory diseases in SCARPOL associated with a decline of 10 µg/m3 PM10 levels.
aAdjusted for age, sex, nationality, parental education, number of siblings, farming status, low birth weight, breast-feeding, child who smokes, family history of asthma, bronchitis, and/or atopy, mother who smokes, indoor humidity, mode of heating and cooking, carpeting, pets allowed in bedroom, removal of carpet and/or pets for health reasons, person who completed questionnaire, month when questionnaire was completed, number of days with the maximum temperature < 0°C, belief of mother that there is an association between environmental exposures and children's respiratory health, and region.
|
|

Figure 3. Mean change in adjusted prevalencea (1998-2001 to 1992-1993) versus mean change in regional annual averages of PM10 (1997-2000 to 1993) for nocturnal dry cough, chronic cough, and conjunctivitis symptoms across nine SCARPOL regions. Abbreviations: An, Anières; Be, Bern; Bi, Biel; Ge, Geneva; La, Langnau; Lu, Lugano; Mo, Montana; Pa, Payerne; Zh, Zürich.
aAdjusted for age, sex, nationality, parental education, number of siblings, farming status, low birth weight, breastfeeding, child who smokes, family history of asthma, bronchitis, and/or atopy, mother who smokes, indoor humidity, mode of heating and cooking, carpeting, pets allowed in bedroom, removal of carpet and/or pets for health reasons, person who completed questionnaire, month when questionnaire was completed, number of days with the maximum temperature < 0°C, and belief of mother that there is an association between environmental exposures and children's respiratory health.
|
PM10 levels, adjusted prevalence, and covariates 1992-2001. Figure 1 shows PM10 levels at fixed monitoring sites in nine study regions of SCARPOL in 1993 and between 1997 and 2000. Across the nine study regions, the average decline of PM10 between 1993 and 2000 was 9.8 µg/m3 (29%). The average absolute decline in the urban and suburban areas Anières, Bern, Biel, Geneva, Lugano, and Zürich (12.7 µg/m3) was about three times as strong compared with the rural and alpine areas Langnau, Payerne, and Montana (4.0 µg/m3).
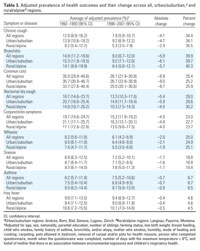
The adjusted prevalence of all investigated health end points declined between 1992-1993 and 1998-2001 (Table 3). Both the absolute and relative declines were stronger for the nonallergic outcomes chronic cough, bronchitis, common cold, nocturnal dry cough, and conjunctivitis symptoms (4.5-8.9% absolute decline of prevalence, on average, across the nine regions) compared with the allergy-associated end points sneezing during pollen season, asthma, and hay fever (0.4-1.7%). A tendency of a stronger absolute decline in suburban areas compared with rural/alpine areas was observed for the nonallergic, but not for the allergy-associated, outcomes.
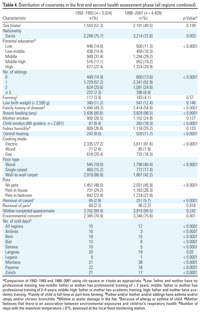
Table 4 shows the distribution in the first (1992-1993) and second (1998-2001) health assessment phase of the covariates included in the multivariate models for analyzing the association between change of air pollution and change of prevalence. Excluded are children with missing data for one or more covariates. The most striking time trend is the increase in self-reported smoking of eighth graders from 6.4 to 16.3% (p < 0.0001). Mothers' environmental concerns had declined on average from 78.9 to 75.6% (p = 0.001). The average annual number of cold days (days with the maximum temperature below zero degrees Celsius) had declined from 15 in 1992-1993 to 12 in 1998-2002 (p < 0.0001) across all study regions, with the strongest decline in Anières (from 10 to 3). An increase in the number of cold days was recorded in the alpine area Montana (from 21 to 38). Because the generally milder winters (with the exception of Montana) and the attenuated environmental concerns would be expected to move in the same direction as declining air pollution levels, that is, toward lower prevalence of reported symptoms and diseases, the logistic regression models were adjusted for the two secular trends. Participation rates in the four cross-sections (69.9, 82.4, 75.3, and 75.0%, respectively) indicated no secular trend.
Change in PM10 exposure versus change in prevalence. Figure 2 shows that declining levels of PM10 were associated with declining prevalence of chronic cough, bronchitis, common cold, nocturnal dry cough, and conjunctivitis symptoms. For wheezing, sneezing, asthma, and hay fever, no significant association could be seen with declining PM10 levels. We found no effect modification by age group, sex, family history of allergic diseases, asthma of child, smoking, or indoor exposures. Random effect models did not change the effect estimates.
Mothers' concerns regarding air pollution and children's respiratory health were significant predictors for reported bronchitis, common cold, nocturnal dry cough, conjunctivitis symptoms, wheeze, and asthma, whereas the number of cold days was not significantly associated with reported symptoms and diseases (data not shown). Without adjustment for the temporal trends of mothers' beliefs (on individual level) and number of cold days (on area level), the effect estimates were slightly stronger for chronic cough, common cold, nocturnal dry cough, and conjunctivitis symptoms and reached significance for wheeze (data not shown). Besides change in PM10, the covariates age, family history of bronchitis, child's smoking, indoor humidity, and removal of carpets were the strongest significant predictors for chronic cough and bronchitis, while for asthma and hay fever, this applied to sex, age, family history of asthma and atopy, and removal of carpets and pets (data not shown). Crude estimates were quite similar to adjusted ORs (data not shown). The fit of the models was generally satisfactory according to Hosmer-Lemeshow chi-square (8 d.f.).
Figure 3 illustrates that, on an aggregate level, across regions the mean change in adjusted prevalence of nocturnal dry cough is associated with the mean change in PM10 levels (rPearson = 0.81, p = 0.008). The strongest decline of adjusted prevalence of nocturnal dry cough was observed in Geneva, Lugano, and Anières, where the strongest reduction of PM10 had also been achieved. Similar associations were observed for chronic cough (r = 0.78; p = 0.02) and conjunctivitis symptoms (r = 0.69; p = 0.04) (Figure 3), whereas for common cold (r = 0.48; p = 0.19) and bronchitis (r = 0.10; p = 0.80), the associations across regions were weaker and not significant.
We showed that decreasing levels of PM10 were associated with declining prevalence rates of those respiratory symptoms and diseases associated with air pollution in the first cross-sectional analysis of SCARPOL (Braun-Fahrländer et al. 1997). The reduction in prevalence rates was larger in areas with a stronger decrease in PM10 levels. Decreasing environmental concerns of mothers (Swiss Society for Applied Social Research 2003) over time contributed to the observed decrease in respiratory symptoms and diseases but did not explain the association with air pollution. Adverse effects of PM10 have no apparent threshold, as we observed the beneficial effects for relatively small changes in rather moderate air pollution levels. We therefore conclude that even relatively small reductions in air pollution levels may improve children's respiratory health.
Our findings are consistent with the improvement of nonallergic respiratory morbidity in children along with declining air pollution levels reported for East Germany (Heinrich et al. 2002; Kramer et al. 1999), although baseline levels and decline in Switzerland (SAEFL 2003) were much smaller. They are also in line with the few intervention studies that have investigated the impact of changing air pollution levels on children's lung function growth (Avol et al. 2001; Frye et al. 2003; Neuberger et al. 2002) and bronchial responsiveness (Wong et al. 1998) and on mortality in adults (Clancy et al. 2002; Hedley et al. 2002). All these studies have found improved respiratory health or reduced respiratory and cardiovascular mortality after mitigation of ambient air pollution exposures. The consistency of these findings suggests that the observed associations between air pollution and respiratory health outcomes may be causal.
In our study, declining PM10 levels were not associated with changes in prevalence of asthma, hay fever, and sneezing during pollen season. No adverse effects of PM10 were observed for these allergy-associated health outcomes in cross-sectional analyses of SCARPOL (Braun-Fahrländer et al. 1997), and they have shown only a very small average decline in our study population and stable prevalence over the last decade in Swiss adolescents (Braun-Fahrländer et al. 2004). A similar contrast between nonallergic and allergy-associated health outcomes in children and declining air pollution levels has been reported by Kramer et al. (1999). Hirsch et al. (1999) reported significant associations of NO2, carbon monoxide, and benzene with bronchitis and morning cough but not with allergy-associated end points. A few studies using traffic counts or proximity to street as exposure proxy found positive associations with sensitization and allergy-related symptoms (Nicolai et al. 2003; van Vliet et al. 1997; Venn et al. 2001; Wjst et al. 1993). We cannot exclude such effects, but for our analysis we had no such data available.
Adjustment for the observed time trends of declining environmental concerns of mothers and reduced number of cold days over the study period did not markedly change the effect estimates. The monitoring of influenza epidemics by the Swiss Federal Office of Public Health (SFOPH) does not suggest a decrease in influenza between 1992 and 2001, which might have confounded our findings, but indicates random fluctuations between years (SFOPH 2001). The same is true for the number of hourly ozone concentrations exceeding 120 µg/m3 [Federal Commission for Air Hygiene (EKL) 2004]. For evaluation of the impact of other possible secular trends such as changes in health habits or medication use, we had no data available. Confounding of our cross-sectional findings by political or social time trends is very unlikely. In Switzerland, the system has been very stable throughout the study period (and was for many decades before), in contrast to the social changes that went hand in hand with air pollution reduction in East Germany (Frye et al. 2003; Heinrich 2003). Thus, uncontrolled confounding or secular trends are unlikely to explain our finings.
Our study is limited in that the comparison for each school grade is based on two points in time only, which are 6, 7, and 8 years apart for the first, eighth, and fourth graders, respectively. The difference in absolute change between the three age groups has been taken into account by design in the multivariate logistic regression models. However, we cannot evaluate whether the relevant time frame for the observed associations between air pollution reduction and improved respiratory health is long term (several years) or rather the year-to-year variability of air pollution levels, as recent Californian findings suggest (McConnell et al. 2003). For lifetime prevalence of asthma and hay fever, the relevance of the investigated change of exposure over a few years could be questioned, particularly for teenage children (eighth graders) who were exposed to higher air pollution levels in their early years of life, compared with first graders. Zmirou et al. (2004) report that exposure to traffic exhausts before the age of 3 years is associated with asthma in school children, but not lifelong exposures. In our data, no effect modification by age could be observed for asthma and hay fever, and their lifetime prevalence has been stable over the last decade in Swiss adolescents (Braun-Fahrländer et al. 2004).
We conclude that air pollution abatement measures implemented in Switzerland in the 1990s that resulted in moderately reduced air pollution exposures (SAEFL 2003; Kuebler et al. 2001) have successfully contributed to improved respiratory health in Swiss schoolchildren. Thus, not only dramatic changes (Heinrich 2003), but also modest improvements of ambient air pollution seem to be beneficial for children's respiratory health. The larger reduction in symptom rates in areas with a stronger decrease in PM10 levels supports the causality of observed associations between air pollution and respiratory health in children. Our findings do not suggest a threshold for adverse effects of PM10, because we observed beneficial effects of rather small PM10 reductions in a moderately polluted environment. In urban regions and in the proximity of streets with high traffic volume, current PM10 levels still exceed limit values of the Swiss Clean Air Act (SAEFL 2003). Therefore, it can be assumed that there is still a potential for further improvement of both ambient air pollution and children's health in Switzerland.
|
|
 |
| [References Listed in PubMed]
References
Asher MI, Keil U, Anderson HR, Beasley R, Crane J, Martinez F, et al. 1995. International Study of Asthma and Allergies in Childhood (ISAAC): rationale and methods. Eur Respir J 8(3):483-491.
Avol EL, Gauderman WJ, Tan SM, London SJ, Peters JM. 2001. Respiratory effects of relocating to areas of differing air pollution levels. Am J Respir Crit Care Med 164(11):2067-2072.
Brauer M, Hoek G, Van Vliet P, Meliefste K, Fischer PH, Wijga A, et al. 2002. Air pollution from traffic and the development of respiratory infections and asthmatic and allergic symptoms in children. Am J Respir Crit Care Med 166(8): 1092-1098.
Braun-Fahrländer C, Gassner M, Grize L, Takken-Sahli K, Neu U, Stricker T, et al. 2004. No further increase in asthma, hay fever and atopic sensitisation in adolescents living in Switzerland. Eur Respir J 23(3): 407-413.
Braun-Fahrländer C, Vuille JC, Sennhauser FH, Neu U, Künzle T, Grize L, et al. 1997. Respiratory health and long-term exposure to air pollutants in Swiss schoolchildren. SCARPOL Team. Swiss Study on Childhood Allergy and Respiratory Symptoms with Respect to Air Pollution, Climate and Pollen. Am J Respir Crit Care Med 155(3): 1042-1049.
Chen PC, Lai YM, Wang JD, Yang CY, Hwang JS, Kuo HW, et al. 1998. Adverse effect of air pollution on respiratory health of primary school children in Taiwan. Environ Health Perspect 106:331-335.
Clancy L, Goodman P, Sinclair H, Dockery DW. 2002. Effect of air-pollution control on death rates in Dublin, Ireland: an intervention study. Lancet 360(9341): 1210-1214.
DIGITEL. 1999. Operation Manual and User's Handbook for Automated Particle Sampler, Type DIGITEL DHA-80 and DIGITEL HVS-HDI2 Operation Module [in German]. Hegnau, Switzerland:DIGITEL AG.
EKL (Federal Commission for Air Hygiene). 2004. Summer smog [in German]. Available: http://www.umwelt-schweiz.ch/buwal/de/fachgebiete/fg_luft/themen/sommersmog/ [accessed 28 September 2005].
Frye C, Hoelscher B, Cyrys J, Wjst M, Wichmann HE, Heinrich J. 2003. Association of lung function with declining ambient air pollution. Environ Health Perspect 111:383-387.
Gauderman WJ, Gilliland GF, Vora H, Avol E, Stram D, McConnell R, et al. 2002. Association between air pollution and lung function growth in southern California children: results from a second cohort. Am J Respir Crit Care Med 166(1):76-84.
Gehring U, Cyrys J, Sedlmeir G, Brunekreef B, Bellander T, Fischer P, et al. 2002. Traffic-related air pollution and respiratory health during the first 2 yrs of life. Eur Respir J 19(4):690-698.
Hedley AJ, Wong CM, Thach TQ, Ma S, Lam TH, Anderson HR. 2002. Cardiorespiratory and all-cause mortality after restrictions on sulphur content of fuel in Hong Kong: an intervention study. Lancet 360(9346):1646-1652.
Heinrich J. 2003. Nonallergic respiratory morbidity improved along with a decline of traditional air pollution levels: a review. Eur Respir J(Suppl 40): 64s-69s.
Heinrich J, Hoelscher B, Frye C, Meyer I, Pitz M, Cyrys J, et al. 2002. Improved air quality in reunified Germany and decreases in respiratory symptoms. Epidemiology 13(4):394-401.
Hirsch T, Weiland SK, von Mutius E, Safeca AF, Grafe H, Csaplovics E, et al. 1999. Inner city air pollution and respiratory health and atopy in children. Eur Respir J 14(3):669-677.
Horak F Jr, Studnicka M, Gartner C, Spengler JD, Tauber E, Urbanek R, et al. 2002. Particulate matter and lung function growth in children: a 3-yr follow-up study in Austrian schoolchildren. Eur Respir J 19(5): 838-845.
Hruba F, Fabianova E, Koppova K, Vandenberg JJ. 2001. Childhood respiratory symptoms, hospital admissions, and long-term exposure to airborne particulate matter. J Expo Anal Environ Epidemiol 11(1):33-40.
Janssen NA, Brunekreef B, van Vliet P, Aarts F, Meliefste K, Harssema H, et al. 2003. The relationship between air pollution from heavy traffic and allergic sensitization, bronchial hyperresponsiveness, and respiratory symptoms in Dutch schoolchildren. Environ Health Perspect 111: 1512-1518.
Kramer U, Behrendt H, Dolgner R, Ranft U, Ring J, Willer H, et al. 1999. Airway diseases and allergies in East and West German children during the first 5 years after reunification: time trends and the impact of sulphur dioxide and total suspended particles. Int J Epidemiol 28(5): 865-873.
Krütli P, Monn C. 1999. PM10-Comparison Measurements between Harvard Low-Flow-Rate-Sharp-Cut-Impactor and DIGITEL DHA-80. Institute for Hygiene and Applied Physiology, ETHZ. Report No UH-99-10. Zurich:Swiss Federal Institute of Technology Zurich.
Kuebler J, van den Bergh H, Russell AG. 2001. Long-term trends of primary and secondary pollutant concentrations in Switzerland and their response to emission controls and economic changes. Atmos Environ 35: 1351-1363.
McConnell R, Berhane K, Gilliland F, London SJ, Vora H, Avol E, et al. 1999. Air pollution and bronchitic symptoms in Southern California children with asthma. Environ Health Perspect 107:757-760.
McConnell R, Berhane K, Gilliland F, Molitor J, Thomas D, Lurmann F, et al. 2003. Prospective study of air pollution and bronchitic symptoms in children with asthma. Am J Respir Crit Care Med 168(7):790-797.
Neuberger M, Moshammer H, Kundi M. 2002. Declining ambient air pollution and lung function improvement in Austrian children. Atmos Environ 36:1733-1736.
Nicolai T, Carr D, Weiland SK, Duhme H, von Ehrenstein O, Wagner C, et al. 2003. Urban traffic and pollutant exposure related to respiratory outcomes and atopy in a large sample of children. Eur Respir J 21(6):956-963.
Röösli M, Braun-Fahrländer C, Künzli N, Oglesby L, Theis G, Camenzind M, et al. 2000. Spatial variability of different fractions of particulate matter within an urban environment and between urban and rural sites. J Air Waste Manag Assoc 50(7):1115-1124.
Röösli M, Theis G, Künzli N, Staehelin J, Mathys P, Oglesby L, et al. 2001. Temporal and spatial variation of the chemical composition of PM10 at urban and rural sites in the Basel area, Switzerland. Atmos Environ 35(21):3701-3713.
SAEFL. 2003. NABEL--Air Pollution Levels 2002 [in German]. Schriftenreihe Umwelt. No. 360. Bern: Swiss Agency for the Environment, Forest and Landscape.
SFOPH (Swiss Federal Office of Public Health). 2001. Infectious Disease, Switzerland, 2001: Virus Influenza. Available: http://www.bag.admin.ch/infreporting/gs01/p21.pdf [accessed 31 May 2005].
Swiss Clean Air Act. 1985. Luftreinhalte-Verordnung vom 16. Dezember 1985 (LRV) [in German]. SR-Number 814.318.142.1.
Swiss Society for Applied Social Research. 2003. The worry barometer 2003 of Credit Suisse [in German]. Bern:GfS-Research Institute. Available: http://emagazine.credit-suisse.com/_product_documents/_articles/41997/Sorgenbarometer-2003-final.pdf [accessed 31 May 2005].
van Vliet P, Knape M, de Hartog J, Janssen N, Harssema H, Brunekreef B. 1997. Motor vehicle exhaust and chronic respiratory symptoms in children living near freeways. Environ Res 74(2):122-132.
Venn AJ, Lewis SA, Cooper M, Hubbard R, Britton J. 2001. Living near a main road and the risk of wheezing illness in children. Am J Respir Crit Care Med 164(12): 2177-2180.
Wjst M, Reitmeir P, Dold S, Wulff A, Nicolai T, von Loeffelholz-Colberg EF, et al. 1993. Road traffic and adverse effects on respiratory health in children. BMJ 307(6904):596-600.
Wong CM, Lam TH, Peters J, Hedley AJ, Ong SG, Tam AY, et al. 1998. Comparison between two districts of the effects of an air pollution intervention on bronchial responsiveness in primary school children in Hong Kong. J Epidemiol Community Health 52(9):571-578.
Zmirou D, Gauvin S, Pin I, Momas I, Sahraoui F, Just J, et al. 2004. Traffic related air pollution and incidence of childhood asthma: results of the Vesta case-control study. J Epidemiol Community Health 58(1): 18-23.
Last Updated: October 13, 2005
|
|
 |
|
| |

