
NTP Study Reports

NTP Study Reports
Home » Study Results & Research Projects » NTP Study Reports » All Long-Term Reports » Abstract for TR-474 - Polyvinyl Alcohol
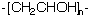
| Chemical Formula: C2H4O | - | 3D Structure* |
|---|---|---|
| *To view structure, download free Chemscape Chime Plug-in | ||
Polyvinyl alcohol is produced primarily for use in textile sizing, adhesives, polymerization aids, and paper coatings. It is also used in surgical drapes, towels, and gauze sponges; protective gloves; cosmetic formulations; topical ophthalmic preparations; plastic sponge implants for reconstructive surgery; and intravaginal contraceptive foam and film. In addition, polyvinyl alcohol is used with magnesium sulfate to dilate the cervix of women prior to induction of labor. It is estimated that hundreds of thousands of women in the United States use an intravaginal product containing polyvinyl alcohol each year. The Food and Drug Administration nominated low-viscosity polyvinyl alcohol for a 2-year study because of concern about the lack of information about the long-term toxic and carcinogenic effects by the intravaginal route. Female B6C3F1 mice received polyvinyl alcohol (approximately 99% pure) in deionized water by intravaginal administration for 30 days or 2 years.
Three
groups of 50 female B6C3F1 mice were used in this intravaginal
study. The vehicle control group received only 20 µL of
a deionized water vehicle. The other two groups each received
20 µL of 25% polyvinyl alcohol in deionized water. Animals
in one dose group were returned to their cages after dosing; animals
in the other dose group were restrained in a vertical nose-down
position in restraint bags for several minutes after dosing. Animals
were dosed daily for 30 consecutive days. All mice survived to
the end of the study. The final mean body weights and body weight
gains of dosed mice were similar to those of the vehicle control
group. Abnormalities noted in the vaginal area after dosing included
vaginal plugs, secretions, and swelling. These vaginal changes
were minimal to mild and occurred in vehicle controls as well
as in dosed mice. Restraint of mice after dosing appeared to eliminate
vaginal secretions but increased both the incidence of vaginal
irritation and the severity of vaginal opening swelling. At necropsy,
mildly enlarged uterine horns were observed in 10 vehicle control
mice, three 25% mice, and seven 25% (restrained) mice. No chemical-related
lesions were observed.
Three
groups of 100 female B6C3F1 mice were used in this
intravaginal study: an untreated control group, a vehicle control
group receiving 20 µL deionized water vehicle only, and
a dosed group receiving 20 µL 25% polyvinyl alcohol in deionized
water. Animals were dosed 5 days per week, excluding holidays,
for 104 to 105 weeks.
Survival,
Body Weights, and Clinical Findings
Survival
of dosed mice was similar to that of the two control groups. The
final mean body weight of vehicle control mice was less than that
of the untreated control group. The mean body weights of the dosed
mice were less than those of the untreated controls from week
17 until the end of the study. The only clinical finding was vaginal
irritation, observed in six mice in the vehicle control group
and 11 mice in the dosed group.
Pathology
Findings
No
neoplasms or nonneoplastic lesions related to chemical treatment
were observed. The incidences of reproductive tract nonneoplastic
lesions in the dosed group did not differ significantly from those
in the vehicle control group; similarly, the incidences of reproductive
tract nonneoplastic lesions in the vehicle control group did not
differ significantly from those in the untreated control group.
Under
the conditions of this 2-year study, there was no evidence
of carcinogenic activity of polyvinyl alcohol (molecular weight
approximately 24,000) in female B6C3F1 mice administered
20 µL of a 25% solution intravaginally. There were no neoplasms
or nonneoplastic lesions considered related to treatment with
polyvinyl alcohol.
Synonyms: Ethenol homopolymer, PVA
Trade names: Akwa Tears, Alcotex, Alvyl, Aracet, Cipoviol, Covol, Elvanol, Ethenol, Gelvatol, Gohsenol, Ivalon, Kuralon, Kurare, Lemol, Liquifilm, Mowiol, Polydesis, Polysizer, Polyvinol, Polyviol, Poval, Resistoflex, Rhodoviol, Sno Tears, Solvar, Sumitex, Vibatex, Vinacol, Vinalak, Vinarol, Vinarole, Vinavilol, Vinol, Vinylon
| Summary of the 2-Year Carcinogenesis Study of Polyvinyl Alcohol in Female B6C3F1 Mice | ||||
|---|---|---|---|---|
| Doses | Untreated control, vehicle control receiving 20 µL deionized water only, and dosed group receiving 20 µL 25% polyvinyl alcohol in deionized water | |||
| Body weights | Vehicle control and dosed groups slightly less than untreated control group | |||
| 2-Year survival rates | 47/100, 51/100, 61/100 | |||
| Nonneoplastic effects | None | |||
| Neoplastic effects | None | |||
| Level of evidence of carcinogenic activity | No evidence | |||
Report Date: May 1998
Pathology Tables, Survival and Growth Curves from NTP 2-year Studies
Target Organs & Incidences from 2-year Studies
You may link to the full technical report in pdf format ( Note: A print ready copy of the document is presented in Portable Document Format (pdf) which requires the Acrobat Reader plug-in -- download a free copy of the reader.)
Web page last updated on October 11, 2007
The National Institute of Environmental Health Sciences is one of the National Institutes of Health within the U.S. Department of Health and Human Services. The National Toxicology Program is headquartered on the NIEHS campus in Research Triangle Park, NC.