You are what you eat.
The Official Mad Cow Disease Home Page
http://www.mad-cow.org/
Getting Rid of Rotavirus
On 12 December 1997, the FDA's Vaccine and Related Biologicals Products Advisory Committee unanimously concluded that RotaShield, the first vaccine for rotavirus--a highly contagious disease that causes life-threatening diarrhea in young children--is safe and effective, and recommended licensing.
In the United States alone, rotavirus causes 3 million cases of childhood diarrhea, resulting in 500,000 doctor visits, 100,000 hospitalizations, and up to 100 deaths annually. Direct costs of the disease run to as much as $400 million and indirect costs (such as lost work time for parents) total $1.4 billion. Worldwide, rotavirus kills 873,000 children each year, largely in underdeveloped countries. The vaccine is intended to be administered in three doses at 2, 4, and 6 months of age when children receive their polio vaccine.
The RotaShield vaccine was created and patented by virologist and physician Albert Kapikian and his colleagues at the National Institute of Allergy and Infectious Diseases in Bethesda, Maryland. Kapikian, head of the epidemiology section of the Laboratory of Infectious Diseases, has devoted nearly 25 years to studying rotavirus. "It's exciting and gratifying as a physician to see that most babies can be protected from severe diarrhea with a product developed in our laboratory," he says.

Last of a dying breed? A new vaccine expected to be approved by the FDA may mean the end of rotavirus outbreaks.
Source: Charles Humphrey/CDC
Australian virologists discovered rotavirus in 1973. Later, several epidemiological studies proved that rotavirus was the most important agent involved in infantile diarrhea worldwide. Kapikian's ingenuity and perseverance helped overcome obstacles in creating the vaccine. For instance, human rotavirus resisted growth in cell cultures, whereas simian rotavirus grew well. The researchers worked to determine whether an animal rotavirus could protect humans, much in the same way that Edward Jenner determined 200 years ago that vaccinating people with cowpox could prevent smallpox. The final vaccine contains a weakened form of rotavirus obtained from rhesus monkeys, bioengineered with three genes from human rotavirus. This combination proved most effective at protecting people against the four most prevalent clinical strains of rotavirus.
The vaccine was tested successfully in numerous clinical trials. Among 2,000 infants in Venezuela, the vaccine reduced severe diarrheal illness by 88% and dehydration by 75%, and decreased hospitalizations by 70%. This study, reported in the 23 October 1997 issue of the New England Journal of Medicine, was the largest and most successful trial of the vaccine in a developing country. A multi-center trial conducted in the United States of 1,200 children showed that the vaccine protected 80% of the children against severe diarrhea and 100% against dehydration, as described in the January 1996 issue of Pediatrics. Another study, which appeared in the 25 October 1997 issue of Lancet, concluded that when tested on 2,400 Finnish children the vaccine reduced severe diarrhea by 91%, clinic visits for dehydration by 97%, and hospitalizations by 100%. In the latest study, published in the October 1997 issue of the Journal of Pediatrics, the vaccine prevented 69% of severe cases of diarrhea on American Indian reservations, where incidence rates of rotavirus disease run higher than for the general population. "The routine use of this vaccine could save lives and prevent illness," says Mathuram Santosham, director of the Johns Hopkins Center for American Indian and Alaskan Native Health in Baltimore, Maryland. In all clinical tests, the vaccine was safe and well-tolerated.
About 90% of all children are infected by rotavirus by age three regardless of hygienic conditions. Diarrhea is considered a normal part of childhood, and few hear about the small number of children who die yearly from rotavirus. Although a current television advertisement for a bathroom cleaner mentions that it kills rotavirus, in reality "parents in the U.S. don't know that rotavirus is a problem," says senior clinical scientist Michael Pastorino of Wyeth-Ayerst Research in Radnor, Pennsylvania, a division of the company seeking licensure for the RotaShield vaccine. Both parents and physicians need to be educated about the new vaccine.
Wyeth-Ayerst is seeking a universal recommendation that all children be vaccinated for rotavirus from the Advisory Committee for Immunization Practices at the CDC, which sets the schedule of childhood immunizations.
Stockpiling Safety?
By spring, the Nuclear Regulatory Commission (NRC) is expected to act, once again, on the 15-year-old issue of whether it makes sense for states to consider stockpiling thousands of tablets of potassium iodide (KI) near nuclear power plants in case of an emergency. No one disputes that KI, a benign substance long approved for over-the-counter use, is an effective thyroid-blocking agent. And no one is arguing that the cost of KI is prohibitive. A 130-mg tablet costs $.07; supplying all residents within five miles of one of the United States' 107 operating nuclear reactors would cost as little as $200,000 (although distributing and disposing of the supply would add costs). Few even question the safety of the tablets. The issue is whether having KI on hand would make some residents trust their fate to a pill rather than to evacuation.
Many states, as well as the nuclear industry, believe evacuation should be the first line of protective action in a nuclear emergency. KI isn't a "silver bullet," says Steve Unglesbee, a spokesman for the Nuclear Energy Institute in Washington, DC, because it can create a false sense of security as well as "ambiguity through choice." Unglesbee likes to quote an April 1997 Chicago Sun-Times editorial that argued against stockpiling: "The idea is akin to putting a dab of sunblock on your nose at the beach; you may end up with a protected prow, but the rest of your body will be scorched."
But supporters of stockpiling, including the American Thyroid Association, contend that it would be negligent of the industry and of state and federal governments not to endorse what they call a simple and cheap insurance policy. "I compare it to lifeboats and life jackets on a ferry," says Peter Crane, a staff attorney for the NRC and a long-time advocate for the tablets' use. "It's better to be evacuated in a lifeboat, but also helps to have on a life jacket."

|
Medical insurance? A current debate considers whether stockpiling potassium iodide near nuclear reactors offers a safeguard or a false sense of security. |
The tablet works by saturating the thyroid with iodide, which the organ needs to make hormones needed by the nervous system and brain. A saturated thyroid will not, therefore, absorb cancer-promoting radioactive iodide that may escape into the air from a nuclear power plant's containment facility during a nuclear accident. Crane has been leading the effort to stockpile KI as a private citizen since 1989. He says his position comes from experience; Crane was diagnosed with thyroid cancer at age 26, more than 20 years after having his tonsils irradiated as a child in a Chicago hospital. He has battled the disease, which kills about 1,000 U.S. citizens annually, on and off for decades.
In 1989, when the NRC refused to change its opinion not to require KI stockpiling, Crane filed a professional opinion challenging that action. That was three years after the Chernobyl nuclear power plant accident, which resulted in about 900 cases of thyroid cancer, leaving many Soviet children with the "Belarussian necklace," a surgical scar that goes from ear to ear, says Crane. But in Poland, upwind of the accident, children were given KI pills, and there has been no increase in thyroid cancer. Also, in 1989 the World Health Organization recommended preventive distribution of KI, and, to date, France and Switzerland have distributed it widely.
But with one-fourth of the world's reactors, the United States has taken a conservative attitude. Despite a federal recommendation following the Three Mile Island accident in 1979 to stockpile KI, the NRC did not endorse the recommendation in 1985, and deadlocked on the issue again in 1994 after Crane urged reconsideration. One survey considered by NRC commissioners showed that 33 of 43 responding states opposed stockpiling, in part because they would have to develop plans to distribute the pills.
Nevertheless, in October 1996 the federal government agreed to buy KI for any state that wanted it. Additionally, plans were made to stockpile KI in 27 metropolitan areas and three national stockpiles. To date, three city stockpiles exist but plans to move the medicine to accident sites do not.
Now Crane wants the NRC to change its language to say stockpiling is "reasonable and prudent" and that states should consider it. His petition was published in the Federal Register in December for public comment and is expected to be voted on by the NRC after February. Unglesbee says the industry will oppose the measure, as it always has. He says that requiring states to consider KI constitutes "an unfunded federal mandate" on states that have already decided that KI pills "work against more effective evacuation."
But not all states feel that way. Tennessee and Alabama already stockpile KI at emergency centers, and in December, Maine's radiation advisory committee recommended stockpiling. Their endorsement came after strong testimony that portions of the evacuation plan for the area surrounding the Maine Yankee nuclear reactor in Wiscasset, Maine, failed during a test. This reactor, the state's only nuclear plant, has since been closed.
Crane feels that the NRC will eventually endorse stockpiling. "It's a matter of people finding out about it just as they did in Maine," he says. And what if the NRC votes his petition down? Well, he says, he can always ask for a judicial review.
Gamma Groceries
Highly publicized incidents last year, such as the recall by Hudson Foods, a Nebraska meat processor, of millions of pounds of hamburger contaminated with the potentially deadly bacteria Escherichia coli O157:H7, have made food safety a major concern in the United States. According to the Council for Agricultural Science and Technology, a nonprofit organization composed of 30 scientific societies, harmful bacteria carried in food can cause as many as 9,000 deaths annually.
In December, the FDA approved the use of ionizing radiation to kill harmful microorganisms in red meat as one more tool to protect the food supply. (The federal government had previously approved irradiation for poultry and fresh fruits and vegetables.) The move, which allows but does not require meat processors to irradiate meat, pleased food scientists and public health officials but worried some public interest and activist groups.

|
Zapped for safety. Advocates say that irradiating food offers real protection against microbes, but some still worry about the safety of the procedure. |
A prime irradiation target is ground beef, which can be laden with pathogens when contaminated carcasses are ground to make hamburger, says Michael Doyle, director of the Center for Food Safety and Quality Enhancement at the University of Georgia in Athens. Irradiating meat is a fairly simple process in which packaged meat is subjected to ionizing radiation--either gamma rays from radioactive materials such as cobalt 60 or cesium 137 or high energy electrons.
Though the U.S. Department of Agriculture has not drawn up rules to describe in detail how the process will work--it's estimated that rule development will take about a year--food scientists envision it occurring at the end of the packing process, just before the meat is shipped from the plant to retail stores. "It will make meat at the point of purchase at least 90% safer than it is today," says Curtis Melton, a professor of food science at the University of Tennessee in Knoxville.
Dennis Buege, a professor of animal science at the University of Wisconsin in Madison, says irradiation will increase ground beef's shelf life. "Rather than your package of ground beef staying in your refrigerator in good condition for three days, it may stay in good condition for six days," he says. Irradiation may also be particularly important in nursing homes and other institutional settings, where food safety is a major concern because food is prepared for relatively large numbers of people, says Buege.
Irradiation will pasteurize, not sterilize, meat, say food safety specialists. The growth of harmful bacteria, such as Salmonella, Campylobacter, and E. coli O157:H7, and other microorganisms that spoil food but pose no harm will be sharply retarded because there will be fewer of them.
Irradiation is by no means the final answer to food safety concerns, however, experts say. "It's a complement to the other practices. The more safety barriers you have, the less likely you're going to have an unsafe product," says George Pauli, director of the division of product policy at the FDA's Center for Food Safety and Applied Nutrition in Washington, DC. Pauli and other authorities emphasize that irradiation doesn't relieve consumers from their responsibility in handling food safely. While he cannot cite exact figures, Morris Potter, an epidemiologist with the CDC says irradiation should "vastly improve the level of food safety."
Critics, however, claim that food irradiation is not necessary and may even harm food. Michael Jacobson, executive director of the Center for Science in the Public Interest, a nonproft education and advocacy organization in Washington, DC, said in a 2 December 1997 statement issued by the center that there are other, cheaper methods, such as steam pasteurization, to make sure foods are safe. In the statement, he objected to the expected added expense of several cents per pound to cover the cost of irradiation equipment, ad added that "the meat and poultry industry should invest in new technologies that create clean products with less expense."
Food and Water, Inc., a Vermont-based activist group, argues that irradiation lowers the nutritional value of meats. Food specialists agree, but counter that the loss is no greater than when meat is cooked. Food and Water also questions the safety of the radiation process, arguing that it may cause illnesses and chromosome damage in those that eat it. But Pauli dismisses that possibility. He says the FDA approved irradiation under a legal standard that it would cause no harm, according to "competent scientists."
How irradiated meat will fare at the counter is uncertain. Opponents and proponents each have polls supporting their view on whether the public approves or disapproves. But consumers will know what they are getting, since a label indicating the meat has been irradiated must appear on the package. The label, however, is too small, according to Food and Water.
In the meantime, there will be efforts to make sure that conditions are optimal for meat irradiation, notes Doyle. One problem to be solved is to make sure "off" flavors don't develop. "The fattier the food, the more likely it is that off flavors will develop," he notes. He also points out that ground beef, which can be 20-30 % fat, may pose a challenge.
Genes and Ozone
New research suggests that whether or not a person reacts to toxic levels of ozone in the air depends upon their genes. If the animal studies that support this association are confirmed in humans, this newest example of the interaction between genes and the environment could have untold implications for industry, insurance, and health.
Researchers believe that knowing they are susceptible could help people protect themselves on bad ozone days, and they hope that clinical genetic therapies might also eventually be developed. Others say information on susceptibility could lead to stricter government regulation of air quality to reduce nitrogen oxides from car exhausts, which combine with oxygen and sunlight to form ozone.
Yet another view is that such information could create a subpopulation of people at risk for discrimination on the basis of their genetic makeup. "It could be a sticky issue if the gene ran in certain ethnic or racial groups, or if disclosure of the gene could risk insurance coverage," says pulmonologist Jeffrey Drazen of the Harvard Medical School in Cambridge, Massachusetts.

|
Ozone culprits. New research shows that not just exposure but also genetics may play a role in susceptibility to the effects of ozone created when sunlight and oxygen mix with auto exhaust. |
"This could become quite a societal issue," acknowledges Steven Kleeberger, one of the scientists who reported the link in the December 1997 issue of Nature Genetics. Kleeberger, a researcher at the Johns Hopkins University department of environmental health sciences, predicts that, given genetic susceptibility, the health effects of ozone will become even more of a regulatory and political issue in the future. The findings of Kleeberger's research, along with those of a second study by George Leikauf and colleagues from the University of Cincinnati that were published in the same issue, move the field of air pollution genetics solidly forward. Now, scientists don't talk about if such susceptibility genes are identified in humans, but when.
Both studies used strains of inbred mice with differing responses to ozone. Kleeberger's team selected one strain of mice that was resistant to ozone and one strain that was very responsive. They crossbred the groups, then bred the groups' offspring to select for expression of genes on chromosome 11 and chromosome 17 that control responsivity to ozone. The Cincinnati researchers also found a locus on chromosome 11 that broadly overlapped with Kleeberger's area, indicating the two teams may be honing in on the same gene.
There was significant activity in the segment on chromosome 17, and in searching the mouse genome database for this chromosome, Kleeberger and his team identified several candidate genes that may be causing the activity. One, the tumor necrosis factor alpha (TNF-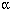 ) gene, seems a highly logical candidate, Kleeberger says. TNF is a pro-inflammatory cytokine that influences genes in the immune response cascade. To test their hypothesis, Kleeberger's team treated the susceptible strain of mice with antibodies that neutralized the TNF-
) gene, seems a highly logical candidate, Kleeberger says. TNF is a pro-inflammatory cytokine that influences genes in the immune response cascade. To test their hypothesis, Kleeberger's team treated the susceptible strain of mice with antibodies that neutralized the TNF-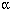 protein. The reaction of these mice to ozone was similar to that of the resistant mice. Although Kleeberger calls this evidence "intriguing," it is not proof that TNF-
protein. The reaction of these mice to ozone was similar to that of the resistant mice. Although Kleeberger calls this evidence "intriguing," it is not proof that TNF-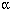 is the controlling gene in ozone susceptibility, he says. More linkage and physical map studies are needed to identify what he suspects may be a bevy of major and contributing genes associated with differing susceptibilities to ozone.
is the controlling gene in ozone susceptibility, he says. More linkage and physical map studies are needed to identify what he suspects may be a bevy of major and contributing genes associated with differing susceptibilities to ozone.
Indeed, Aravinda Chakravarti, a professor of genetics and medicine at Case Western Reserve University in Cleveland, Ohio, questions the use of mice as surrogates for how ozone affects human biology. The inbred mice used in the studies surely have less genetic variation in their reaction to ozone than that found in humans, he says, and moreover, humans metabolize oxygen differently.
But Kleeberger points out that the "mouse represents a unique model to study genes in their most simple representation." Michael Blaese, chief of the clinical gene therapy branch at the National Human Genome Research Institute in Bethesda, Maryland, says it makes sense that TNF-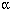 may be in some way linked to ozone susceptibility because it is involved in so many important biological activities.
may be in some way linked to ozone susceptibility because it is involved in so many important biological activities.
Even so, Blaese believes that any effort to clinically correct or repair susceptibility genes will be a long time in coming because of the inherent difficulties of gene therapy. More likely, he says, researchers will search for clinical ways to interfere with the protein encoded by the gene.
This work offers hope to millions of asthmatics, says Sharon Hipkins, director of programs and policy at the Asthma and Allergy Foundation of America in Washington, DC. It highlights "clinical recognition that there probably is a familial trait that mediates reaction to high ozone levels and that, likely, many asthmatics are affected," she says. But Kleeberger says that asthma is a very complex disorder with a number of different phenotypes and that the association between ozone and asthma susceptibility is not completely understood.
"What is most interesting about these studies is that we have always viewed air pollution as having adverse effects on society as a whole," says Drazen. "Like reactions to medications, we are learning we might be able to identify selected individuals who are affected."

Mad Cow Media
"Mad cow disease," the common name for bovine spongiform encephalopathy, or BSE, is alone enough to generate concern in anyone who consumes the daily news along with his hamburger. When the disease broke out among humans in 1996, there was panic among the public, who feared that eating beef would result in death from the "brain-rotting" disease. To help combat such public hysteria, the private charitable Sperling Foundation established the Official Mad Cow Disease Home Page, located at http://www.mad-cow.org/. The page is a repository for over 3,650 articles on BSE and related pathologies, and is updated twice a week (hourly during breaking events). News clips from a vast array of sources are warehoused and cataloged according to topic.
 The Official Mad Cow Disease Home Page is managed by Tom Pringle, scientific director of the Sperling Foundation's Creutzfeld-Jakob disease (CJD) program (CJD is commonly known as the human equivalent of BSE). Says Pringle of the site, "Basically, my ambitions for the site were to create a new paradigm for human disease research, based on unique aspects of the Web such as unlimited available space and essentially no distribution costs. This allows much more depth in terms of articles archived, much more frequent updating than a fixed journal, room for supplemental commentary and clarification by appended correspondence with scientific authors, and opportunity for value-added background links."
The Official Mad Cow Disease Home Page is managed by Tom Pringle, scientific director of the Sperling Foundation's Creutzfeld-Jakob disease (CJD) program (CJD is commonly known as the human equivalent of BSE). Says Pringle of the site, "Basically, my ambitions for the site were to create a new paradigm for human disease research, based on unique aspects of the Web such as unlimited available space and essentially no distribution costs. This allows much more depth in terms of articles archived, much more frequent updating than a fixed journal, room for supplemental commentary and clarification by appended correspondence with scientific authors, and opportunity for value-added background links."
The home page features an extensive list of links to articles divided by topic into 10 categories: General News; Prion Molecular Biology; Victims; Prion Research; BSE in Blood, Milk, Meat; How Beef is Made; Alzheimer's and CJD; Other Cow-to-Human Diseases; Epidemic; and Mad-Cow Scientists. These articles, culled from the lay press, present a chronicle of the development of the mad cow disease scare and research that is being done to identify the source of this alarming malady. The home page also lists links to several different information resources, along with a site search engine and technical e-mail correspondence and sharing of news with Pringle.
Over 200 links to different online journals, search engines, databases, and other resources are listed from the home page under The Best Links. Some of these links are for general Internet reference areas, while others are specifically relevant to the topic of BSE, such as the Food Safety and Inspection Service's listing of food recalls from 1990 through 1998. This page also contains several links to online molecular biology tools, such as the University of Illinois's NCSA Biology Workbench.
The Real Science link on the home page leads to a list of links to scientific papers on the study of BSE. The papers are grouped on the page by topic, with each link leading to an abstract and citation for the original paper. This link also includes reviews by Pringle of current research and tables--updated regularly--of known mutant strains of BSE. Pringle posts his own research as well, saying, "I strongly believe that this is the future of biomedical research and an ethical mandate in the human disease context."
The 3D Interactive Prions link allows visitors to view two- and three-dimensional renderings of the prion protein molecule. Finally, the home page includes a link to the Fundraising and Support for CJD Victims page, a list of support groups for CJD sufferers and their families.
Last Update: March 2, 1998





