
NTP Study Reports

NTP Study Reports
Home » Study Results & Research Projects » NTP Study Reports » All Long-Term Reports » Abstract for TR-383: 1-Amino-2,4-Dibromoanthraquinone
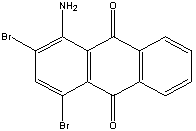
| Chemical Formula: C14H7Br2 NO2 | - | 3D Structure* |
|---|---|---|
| *To view structure, download free Chemscape Chime Plug-in | ||
1-Amino-2,4-dibromoanthraquinone
is an anthraquinone-derived vat dye, a member of a class of insoluble
dyes that are impregnated into textile fibers. Five anthraquinone-derived
dyes with representative and diverse structures, as well as the
parent chemical, anthraquinone, were selected for toxicology and
carcinogenesis evaluation. Similar to the benzidine dye initiative,
the rationale for selecting these vat dyes was to generate sufficient
toxicologic data to permit more reliable predictions of carcinogenicity
to be made on other chemicals in this class, thereby eliminating
or reducing the need to study every anthraquinone dye. 1-Amino-2,4-dibromoanthraquinone
is the last anthraquinone-derived dye in this group to be studied.
Groups of male and female F344/N
rats and B6C3F1 mice were exposed to 1-amino-2,4-dibromoanthraquinone
(87% to 97% pure) for 13 weeks or for 9, 15, or 24 months. Because
1-amino-2,4-dibromoanthraquinone was predicted to be carcinogenic,
these studies were designed to evaluate the potential for tumor
progression and regression. Absorption and excretion studies were
carried out in male F344/N rats. Genetic toxicity was determined
in vitro using Salmonella typhimurium and cultured
Chinese hamster ovary cells. Extensive chemical analyses were
performed to identify and characterize impurities of the 1-amino-2,4-dibromoanthraquinone
used in these studies.
Groups of 10 male and 10 female
rats were given 0, 2,500, 5,000, 10,000, 25,000, or 50,000 ppm
1-amino-2,4-dibromoanthraquinone in feed for 13 weeks. These levels
correspond to approximately 150 to 3,200 mg 1-amino-2,4-dibromoanthraquinone/kg
body weight per day for males and to approximately 170 to 3,200
mg/kg for females. Chemical-related mortality was limited to one
male and one female in the 50,000 ppm groups. Final mean body
weights and body weight gains of all exposed groups of rats were
significantly lower than those of the controls. Feed consumption
by all exposed groups was less than that by the controls throughout
the study and generally decreased with increasing exposure concentration.
Pink-red staining of the fur and tail was observed in all exposed
groups. Absolute and relative liver weights of all exposed groups
were generally significantly greater than those of the controls.
Chemical-related lesions were
present in the liver, kidney, and spleen of male and female rats.
Nonneoplastic lesions in the liver included foci of hepatocellular
alteration, diffuse hepatocellular hypertrophy (cytomegaly), hepatocellular
cytoplasmic vacuolation, bile duct hyperplasia, inflammation,
and pigmentation. These differences were observed primarily in
the 25,000 and 50,000 ppm groups of males and females; the spectrum
of proliferative lesions of the bile ducts (hyperplasia, fibrosis,
and necrotizing cholangitis) in the 25,000 and 50,000 ppm groups
was morphologically consistent with the lesion described as cholangiofibrosis.
Pigmentation was present in the renal tubule epithelium of all
groups of exposed rats; nuclear enlargement (karyomegaly) was
also present in the renal tubule epithelium in some of the exposed
rats. Accumulation of hyaline droplets in the cytoplasm of the
renal tubule epithelium and tubule lumina was present in 2,500,
5,000, 10,000, and 25,000 ppm males. Incidences of hematopoiesis
of the spleen in exposed groups of males and females were increased
compared to those in the controls.
Groups of 10 male and 10 female
mice were given 0, 2,500, 5,000, 10,000, 25,000, or 50,000 ppm
1-amino-2,4-dibromoanthraquinone in feed for 13 weeks. These levels
correspond to approximately 500 to 10,600 mg 1-amino-2,4-dibromoanthraquinone/kg
body weight per day for males and approximately 660 to 11,700
mg/kg per day for females. There was no chemical-related mortality.
Feed consumption and final mean body weights of exposed groups
were similar to those of the controls. Red staining of the fur
was observed in all exposed groups. Absolute and relative liver
weights of the exposed groups were greater than those of the controls
except for the absolute liver weight of 2,500 ppm males. Absolute
and relative kidney weights of 25,000 and 50,000 ppm males were
lower than those of the controls.
Chemical-related lesions were
limited to the livers of males and consisted of pigmentation of
hepatocytes at all exposure concentrations and centrilobular hepatocellular
hypertrophy at 10,000, 25,000, and 50,000 ppm. Minimal pigment
was present in the liver of one female in the 25,000 ppm group
and in one female in the 50,000 ppm group.
Groups of 70 male and 70 female
rats were given 0, 5,000, or 10,000 ppm 1-amino-2,4-dibromoanthraquinone
in feed for 103 weeks. In addition, groups of 50 male and 50 female
rats were given 2,000 ppm 1-amino-2,4-dibromoanthraquinone in
feed for 104 weeks. These exposure concentrations were approximately
equal to 90, 240, or 490 mg 1-amino-2,4-dibromoanthraquinone/kg
body weight for males and 110, 285, or 600 mg/kg for females.
Ten animals from each group were evaluated for histopathology
at 9 months. Additional groups of 10 animals from the 0 and 10,000
ppm groups were evaluated for histopathology at 15 months.
Survival, Body Weights,
Feed Consumption, and Clinical Findings
In the 2-year study, survival
of the 10,000 ppm males and females was significantly lower than
that of the controls. Survival of the 2,000 and 5,000 ppm groups
was similar to that of the controls. During the last year of the
study, the mean body weights of exposed males were 80% to 91%
those of controls, and the mean body weights of exposed females
were 67% to 84% those of controls. Feed consumption among exposed
groups was generally similar, but was less than that by controls.
The fur and urine of all exposed male and female groups were discolored.
Pathology Findings
In the 2-year study, 1-amino-2,4-dibromoanthraquinone
was associated with significant chemical-related increases in
the incidences of benign and malignant neoplasms in the liver,
large intestine, kidney, and urinary bladder of males and females.
Chemical-related nonneoplastic proliferative and degenerative
lesions occurred in the liver, kidney, urinary bladder, and forestomach
of males and females.
The incidences of foci of hepatocellular
alteration and pigmentation in the liver of males and females
were increased at the 9-month interim evaluation, and a hepatocellular
adenoma was present in one 5,000 ppm male. At the 15-month interim
evaluation, hepatocellular adenoma or carcinoma (combined) occurred
in all males and nine females in the 10,000 ppm groups. By the
end of the 2-year study, hepatocellular adenoma, carcinoma, cholangioma,
or cholangiocarcinoma were observed in males and females in the
5,000 and 10,000 ppm groups. In the 2,000 ppm groups, similar
liver neoplasms were present in 63% of the males and in 83% of
the females. Of the hepatocellular carcinomas in the 5,000 and
10,000 ppm groups of males and females, 31% to 49% were metastatic
to the lungs or other sites. Increases in the incidences of foci
of hepatocellular alteration (basophilic, eosinophilic, and clear
cell) and pigmentation of the liver were also observed in exposed
groups of males and females.
Adenomatous polyps (adenoma) of
the large intestine were present in six 10,000 ppm males at the
15-month interim evaluation. Incidences of adenomatous polyp (adenoma)
and carcinoma of the large intestine were significantly increased
in exposed groups of males and females after 2 years; multiple
benign and malignant intestinal neoplasms were observed in many
of these rats.
In the kidney, incidences of renal
tubule adenoma and carcinoma were significantly increased in exposed
groups of males and females after 2 years. Renal tubule adenomas
were present in two 10,000 ppm males at 15 months. There were
also chemical-related increases in the incidences and severities
of renal tubule epithelial hyperplasia, pigmentation, and transitional
cell hyperplasia in the kidney of males and females. Hyaline droplet
accumulation was present in all exposed male rats at 9 months.
Incidences of transitional cell
papilloma and carcinoma of the urinary bladder were increased
at 2 years in males and females in the 10,000 ppm groups. Transitional
cell hyperplasia was observed in exposed males and females at
the 15-month interim evaluation. Other nonneoplastic lesions observed
in the urinary bladder at 2 years included metaplasia of the transitional
epithelium and submucosal stromal tissue.
In the forestomach, the incidences
and severities of inflammation, ulceration, hyperkeratosis, and
hyperplasia of the squamous mucosa were increased in all exposed
groups of males and females at 2 years, but not at the 9- or 15-month
interim evaluations.
In exposed males and females,
the incidences of mononuclear cell leukemia were significantly
decreased. The incidences of atrophy of the seminal vesicle were
increased in exposed male rats in the 2-year study.
Stop-Exposure Evaluation
in Rats
Groups of 40 male and 40 female
rats were given 20,000 ppm 1-amino-2,4-dibromoanthraquinone in
feed for 9 or 15 months. At 9 months, 10 males and 10 females
were evaluated for histopathology (9-month interim evaluation
groups). After 9 months of exposure, an additional 10 males and
10 females were fed control diet until the end of the 15-month
evaluation (9-month stop-exposure groups), and 20 males and 20
females continued to receive 20,000 ppm 1-amino-2,4-dibromoanthraquinone
until the end of the evaluation (15-month exposure groups). The
approximate daily consumption of 1-amino-2,4-dibromoanthraquinone
was 1,335 mg/kg for males and 1,790 mg/kg for females in the 9-month
stop-exposure groups and 1,115 mg/kg for males and 1,435 mg/kg
for females in the 15-month exposure groups.
Survival was similar among groups
except for the females in the 15-month exposure group; the survival
of this group was lower than that of the controls. Lower mean
body weights were related to increased exposure duration. The
mean body weights of exposed males were 76% to 82% that of controls,
and the mean body weights of exposed females were 73% to 84% that
of controls.
For the stop-exposure evaluation,
similar chemical-related neoplasms and nonneoplastic lesions were
observed in the same sites as in the 2-year study: liver, large
intestine, kidney, urinary bladder, and forestomach.
After 9 months of dietary exposure
to a concentration of 20,000 ppm 1-amino-2,4-dibromoanthraquinone,
hepatocellular adenoma and carcinoma occurred in males and females.
Nonneoplastic chemical-related lesions in the liver of exposed
rats included pigmentation, focal hepatocellular alteration, and
bile duct hyperplasia. Neoplasms at other sites in males included
one adenomatous polyp (adenoma) in the large intestine and one
transitional cell papilloma in the urinary bladder. Hyaline droplet
accumulation was present in the kidney of exposed males at 9 months.
In the stop-exposure groups examined at 15 months, hepatocellular adenoma and carcinoma were present in most males and females. Adenomatous polyp (adenoma) of the colon, renal tubule cell adenoma, and urinary bladder transitional cell papilloma and carcinoma also occurred in males and females. Nonneoplastic chemical-related lesions included foci of hepatocellular alteration in the liver and hyperplasia of the renal tubule epithelium and urinary bladder transitional epithelium. Hyperplasia, hyperkeratosis, inflammation, and ulceration were observed in the forestomach of some male and female rats continuously exposed for 15 months.
Groups of 60 male and 60 female
mice were given 0, 10,000, or 20,000 ppm 1-amino-2,4-dibromoanthraquinone
in feed for 104 weeks. The daily compound consumption was approximately
1,690 or 3,470 mg 1-amino-2,4-dibromoanthraquinone/kg body weight
for males and 1,950 or 4,350 mg/kg for females. Ten animals from
each group were evaluated for histopathology at 15 months.
Survival, Body Weights,
Feed Consumption, and Clinical Findings
In the 2-year study, survival
of exposed males was significantly lower than that of the controls.
Survival of exposed females was similar to that of the controls.
The final mean body weights of exposed males were 83% to 85% that
of controls, and the final mean body weights of exposed females
were 81% to 86% that of controls. Feed consumption by exposed
groups was generally similar to that by controls. Discoloration
of the fur, urine, and feces was observed in all exposed groups.
Pathology Findings
In the 2-year study, 1-amino-2,4-dibromoanthraquinone
was associated with significant chemical-related increases in
the incidences of benign and malignant neoplasms in the liver,
forestomach, and lung of males and females.
Incidences of hepatocellular adenoma
and carcinoma were increased in exposed groups at the 15-month
interim evaluation and at 2 years. At 2 years, there were significant
increases in the incidences of multiple hepatocellular adenoma
and carcinoma in males and females and in the incidences of hepatoblastoma
in males. Centrilobular hypertrophy of hepatocytes in males and
foci of hepatocellular alteration and pigmentation in the liver
of males and females were also chemical-related changes.
Sqamous cell papilloma of the
forestomach mucosa occurred in 10,000 ppm females and 20,000 ppm
males and females at the 15-month interim evaluation, and the
incidences of squamous cell papilloma and carcinoma were significantly
increased in exposed groups of males and females at 2 years. Chemical-related
hyperplasia of forestomach epithelium was also present at 15 months
and at 2 years.
Alveolar/bronchiolar adenomas
were present only in the exposed groups of males and females at
15 months, and the incidences of alveolar/bronchiolar adenoma
were significantly increased in exposed males and females at 2
years. The incidences of multiple alveolar/bronchiolar adenomas
were also increased in exposed males.
In the kidney, pigmentation was present in the renal tubules of most mice after 2 years of exposure.
Adult male F344/N rats were given
[14C]-labeled 1-amino-2,4-dibromoanthraquinone as a
single intravenous dose of 0.4 mg/kg body weight or as a single
oral dose of 2, 23, 118, 814, or 1,473 mg/kg. A 6-hour bile cannulation
study was also performed. From day 0 through day 3 after intravenous
administration, about 50% of the 14C was excreted in
the feces, 15% in the urine, and 6% in expired air. Unmetabolized
1-amino-2,4-dibromoanthraquinone accounted for less than 3% of
the excreted 14C after intravenous administration.
For oral doses administered, the amount of the dose that was absorbed
fit the equation: absorbed dose = 6.6 x log(dose). After
intravenous administration, the metabolites of 1-amino-2,4-dibromoanthraquinone
in blood were primarily in the plasma fraction (blood:plasma ratio
of approximately 0.5:1). The highest concentrations of 14C
in tissues 15 minutes after intravenous dosing were in excretory
organs, lung, kidney, small intestine, liver, adipose tissue,
and adrenal gland.
1-Amino-2,4-dibromoanthraquinone
was mutagenic in Salmonella typhimurium strains TA98 and
TA1537 in the absence of S9; with S9, an equivocal response was
observed in TA1537. 1-Amino-2,4-dibromoanthraquinone resulted
in an equivocal response in TA100 with and without S9, and no
mutagenic activity was detected with strain TA1535. In cultured
Chinese hamster ovary cells, 1-amino-2,4-dibromoanthraquinone
induced sister chromatid exchanges with and without S9; chromosomal
aberrations were induced in the absence of S9.
Under the conditions of these
2-year feed studies, there was clear evidence of carcinogenic
activity of 1-amino-2,4-dibromoanthraquinone in male and female
F344/N rats based on increased incidences of neoplasms in the
liver, large intestine, kidney, and urinary bladder. There was
clear evidence of carcinogenic activity of 1-amino-2,4-dibromoanthraquinone
in male and female B6C3F1 mice based on increased incidences of
neoplasms in the liver, forestomach, and lung.
Exposure of male and female rats
to 1-amino-2,4-dibromoanthraquinone for 2 years was associated
with basophilic focus (males only), clear cell focus, eosinophilic
focus, and pigmentation in the liver; renal tubule hyperplasia,
renal tubule pigmentation, and transitional cell hyperplasia in
the kidney; transitional cell hyperplasia, squamous metaplasia,
and stromal metaplasia (females only) in the urinary bladder;
squamous hyperplasia, hyperkeratosis, ulceration, and inflammation
of the forestomach mucosa; and seminal vesicle atrophy. Exposure
of male and female mice to 1-amino-2,4-dibromoanthraquinone for
2 years was associated with centrilobular hepatocellular hypertrophy
(males only), basophilic focus, clear cell focus (females only),
eosinophilic focus, and pigmentation in the liver; pigmentation
in the kidney; and hyperplasia, basal cell hyperplasia, hyperkeratosis,
and inflammation of the forestomach mucosa.
Synonym: ADBAQ
Report Date: August 1996
Pathology Tables, Survival and Growth Curves from NTP 2-year Studies
Target Organs & Incidences from 2-year Studies
You may link to the full technical report in pdf format ( Note: A print ready copy of the document is presented in Portable Document Format (pdf) which requires the Acrobat Reader plug-in -- download a free copy of the reader.)
Web page last updated on October 14, 2004
The National Institute of Environmental Health Sciences is one of the National Institutes of Health within the U.S. Department of Health and Human Services. The National Toxicology Program is headquartered on the NIEHS campus in Research Triangle Park, NC.