
| |  | |  |
Research Highlight
|
| EnvirBreast Milk Monitoring Programs (BMMPs): World-Wide Early Warning System for Polyhalogenated POPs and for Targeting Studies in Children's Environmental Health Kim Hooper |
|
|
 |
Dniprodzerzhinsk, Kyiv, and Qzyl-Orda, names unfamiliar to most of us, are cities located literally on the other side of the globe, 12 time zones away, in the former Soviet Union countries of the Caucasus or Central Asia. Two exposure assessments of polychlorinated persistent organic pollutants (POPs), including the organochlorine pesticides (OCPs), polychlorinated biphenyls (PCBs), and polychlorinated dibenzodioxins and furans (PCDDs/PCDFs), are part of longer term studies of children's health in these regions, and are described in this and recent issues of EHP ( 1- 3). These studies use breast milk as a convenient, noninvasive means of estimating body burdens of these POPs in the mother, fetus, infant, and child. They illustrate the global interest in children's environmental health, particularly from exposures to POPs, whose stability makes them intrinsically hazardous, and hint at the formidable challenges, both scientific and nonscientific, that await researchers undertaking such studies.
Breast milk is an ideal medium for assessing exposures to POPs. POPs enter humans largely as contaminants of dietary animal products, where they sequester in adipose tissue, serum, and breast milk and equilibrate at similar levels on a fat weight basis. With long (5-10 year) half-lives, POPs persist in humans and in breast milk as they do in the environment. Breast milk mimics sediments of rivers or lakes as a storage reservoir for POPs, serves as an indicator of past human exposures or environmental conditions, and complements environmental monitoring data in air, water, soil, and food.
POPs can be especially hazardous because, once released into the environment, they invariably end up in breast milk and in the infant. 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) levels, for example, in the infant at birth are 25% of the maternal levels (4). Breast-fed infants typically ingest TCDD at 50-100-fold higher levels than adults, on a body-weight basis (5,6). Infants absorb 90% of the ingested TCDD (7) and may exceed the adult acceptable daily intake.
Breast milk monitoring programs (BMMP) are valuable as early warning systems for POPs. BMMPs, using standardized collection and analytical protocols established by the World Health Organization (8), operate in several European countries including Sweden, Germany, and the Netherlands. Time-trend data from BMMPs indicate the effectiveness of regulatory actions and provide early warning of POP chemicals of emerging concern. Time-trend data, for example, from BMMPs indicate that POP levels are declining in many Western European countries (Figure 1) (9,10), indicating effective regulation. Recent published results from a Swedish BMMP show a dramatic 5-fold increase over the past decade in contaminant levels of a polybrominated diphenyl ether (PBDE), a flame retardant, in breast milk (Figure 1) (9), indicating a new chemical of concern. There are no BMMPs to determine past or present PBDE levels or time trends in the United States. As seen from the studies described below, more is known about the breast milk contamination and POP body burdens of the mother, infant, and child living in Ukraine or Kazakhstan than, for example, about similar groups living in California (Figure 2) (11).
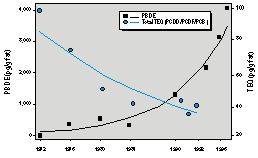
Figure 1. Persistent organochlorine pollutant levels in breast milk in Stockholm, Sweden. Abbreviations: PBDE, polybrominated diphenyl ether; PCDD/PCDF/PCB, polychlorinated dibenzo-p-dioxin and dibenzofuran and polychlorinated biphenyl; TEQ, toxic equivalent. Adapted from Noren and Merironyte (9) with permission from Organohalogen Compounds (Ecoinforma Press).
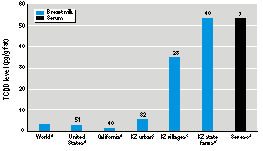
Figure 2. TCDD levels in breast milk and serum from residential populations. KZ, Kazakhstan. Numbers above bars indicate numbers of donors.
aData from the International Agency for Research on Cancer (10). bStockton, California; data from Hooper et al. (11). cData from Hooper et al. (2). dData from Hooper et al. (3) [n = 17 in zone A (53 pg/g) and n = 23 in zone B (21 pg/g)]. eSerum geometric mean from zone A residents sampled in 1992-1993; data from Landi et al. (14).
What do the studies in Ukraine and Kazakhstan show? First, they are the first robust assessments of POP body burdens in former Soviet Union countries, albeit with different objectives and study designs. Second, they demonstrate the versatile and valuable role of breast milk analysis in exposure assessment. Third, the contamination measured was not as severe as anticipated, although body burdens of some POPs were 5-30-fold higher than in European countries.
In the Ukraine study, Gladen et al. measured levels of several POPs in a large number (200) of participants in a reproduction/child health study of urban populations in which samples of colostrum (milk  5 days after birth) were collected and archived from the two major cities of Dniprodzerzhinsk and Kyiv. Mothers were selected without regard to parity, with half under 30 years of age (81% primiparae) and half older (33% primiparae). The small (5 ml) sample volume and use of gas chromatography with electron-capture detector (GC/ECD) limited chemical analysis to major POPs that were present at high (nanograms per gram of fat) levels, including 9 common chlorinated pesticides and 18 major PCB congeners. Using GC/ECD, large numbers of samples can be analyzed relatively cheaply.
5 days after birth) were collected and archived from the two major cities of Dniprodzerzhinsk and Kyiv. Mothers were selected without regard to parity, with half under 30 years of age (81% primiparae) and half older (33% primiparae). The small (5 ml) sample volume and use of gas chromatography with electron-capture detector (GC/ECD) limited chemical analysis to major POPs that were present at high (nanograms per gram of fat) levels, including 9 common chlorinated pesticides and 18 major PCB congeners. Using GC/ECD, large numbers of samples can be analyzed relatively cheaply.
Viable comparisons with European countries could be made because POP levels in colostrum and milk had been shown to be similar in earlier studies: median levels of some PCBs and dichlorodiphenyldichloroethylene (DDE) in milk 6 weeks after birth were 80-90% of levels measured in colostrum (12). Most chlorinated pesticides and PCB congeners in the Ukraine were comparable to levels of Western Europe, but DDE was 2-5-fold higher, and ß-hexachlorocyclohexane (ß-HCH) levels were 3-10-fold higher. POP levels were higher in the industrial city of Dniprodzerzhinsk than in the capital city of Kyiv. More importantly, Gladen et al. show the utility and convenience of colostrum as a monitoring medium, with the advantage of collection at hospitals.
The Kazakhstan study (1-3) was the first comprehensive, congener-specific evaluation of polychlorinated contaminants in a former Soviet Union country, analyzing the 17 PCDDs/PCDFs and 40 dioxinlike co-planar PCBs in breast milk by gas chromatography-high resolution mass spectrometry (HRGC/HRMS), and 19 OCPs by GC/ECD (1-3). Congener-specific analyses of PCDDs/PCDFs and PCBs by HRGC/ HRMS, in contrast to the Ukraine study, required large individual volumes (100 ml) of breast milk and cost 5-fold more than GC/ECD. Thus, selection of participants was critical. POP levels in breast milk change with age and parity of the mother; TCDD, for example, increases with age of the mother and decreases with increasing parity (roughly 20-25% with each breast-fed child) (13). Selection of the study populations (n = 145) controlled for parity (primiparae) and lactation period (2-8 weeks) and avoided economic and cultural selection biases.
POP levels were similar to those of Western Europe, except that ß-HCH levels were 2-40-fold higher and levels of TCDD were remarkably high in samples from rural villages in a southern cotton-growing region (1,2). The TCDD levels, 20-fold higher than U.S. levels, approached present levels found in trichlorophenol or 2,4,5-trichlorophenoxyacetic acid (2,4,5-T) production workers with past occupational exposures (10) or in residents contaminated by an industrial explosion in zone A of Seveso, Italy (14) (Figure 2).
Congener-specific analysis was powerful in identifying the center of the TCDD contamination in Kazakhstan and the likely source of exposure. The distinctive PCDD/PCDF congener profile found in the milk samples, with TCDD as the dominant congener, resembled the "signature" profile for the defoliants
2,4,5-T and Agent Orange (50% 2,4,5-T). TCDD-contaminated batches of 2,4,5-T were produced in Russia in the 1960s. The rural region lies in the fourth largest cotton-growing region in the world, and defoliants were reportedly aerially applied on cotton for 20 years, from 1965 to 1985 (3).
Congener-specific profiles and ratios pinpointed TCDD contamination to a cluster of cotton-growing state farms adjoining an agricultural catchment basin (3). The food supply was widely contaminated. The exposure, apparently chronic and long term (> 20 years), still persists. Future efforts focus on identifying the major sources of TCDD exposures (e.g., fish in the adjacent reservoir) and on adverse health outcomes in women, infants, and children.
As seen in these studies, BMMPs perform valuable functions: they identify areas of POP contamination; they assess maternal and perinatal body burdens; and they identify at-risk populations of mothers, infants, and children in need of follow-up health outcome studies. They also evaluate the effectiveness of regulatory strategies for POPs, including pollution prevention and hazardous waste management, and provide data for successful regulatory decision making. BMMPs are needed in the United States for data gaps, especially for PCDDs/PCDFs, PCBs, PBDEs, and other emerging POPs of interest. A network of health care providers could function as a national BMMP, with maternity health maintenance organizations collecting colostrum samples to assess POP levels in the general population. Lower income groups may have higher risks for POP exposures. The Supplemental Nutrition Program for Women, Infants, and Children (WIC) Centers could collect milk samples to assess levels in these underserved populations.
Kim Hooper
Hazardous Materials Laboratory
California Environmental Protection Agency
Berkeley, California
|
|
 |
| [References Listed in PubMed]
References and Notes
1. Hooper K, Petreas MX, She J, Visita P, Winkler J, McKinney M, Mok M, Sy F, Garcha J, Gill M, et al. Analysis of breast milk to assess exposure to chlorinated contaminants in Kazakstan: PCBs and organochlorine pesticides in southern Kazakstan. Environ Health Perspect 105:1250-1254 (1997).
2. Hooper K, Petreas MX, Chuvakova T, Kazbekova G, Druz N, Seminova G, Sharmanov T, Hayward D, She J, Visita P, et al. Analysis of breast milk to assess exposure to chlorinated contaminants in Kazakstan: high levels of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) in agricultural villages of southern Kazakstan. Environ Health Perspect 106:797-806 (1998).
3. Hooper K, Chuvakova T, Kazbekova G, Hayward D, Tulenova A, Petreas MX, Wade TJ, Benedict K, Cheng YY, Grassman J. Analysis of breast milk to assess exposure to chlorinated contaminants in Kazakhstan: sources of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) exposures in an agricultural region of southern Kazakhstan. Environ Health Perspect 107:447-457 (1999).
4. Koppe JG, Olie K, van Wijnen J. Placental transport of dioxins from mother to fetus. Dev Pharmacol Ther 18:9-13 (1992).
5. Lindstrom G, Hooper K, Petreas M, Stephens R, Gilman A. Workshop on perinatal exposure to dioxin-like compounds. I. Summary. Environ Health Perspect 103(suppl 2):135-142 (1995).
6. Patandin S, Dagnelie PC, Mulder PGH, Op de Coul E, van der Veen JE, Weisglas-Kuperus N, Sauer PJJ. Dietary exposure to polychlorinated biphenyls and dioxins from infancy until adulthood: a comparison between breast-feeding, toddler, and long-term exposure. Environ Health Perspect 107:45-51 (1999).
7. Abraham K, Knoll A, Ende M, Papke O, Helge H. Intake, fecal excretion, and body burden of polychlorinated dibenzo-p-dioxins and dibenzofurans in breast-fed and formula-fed infants. Pediatr Res 40:671-679 (1996).
8. WHO. Levels of PCBs, PCDDs and PCDFs in Breast Milk: Results of WHO-coordinated Interlaboratory Quality Control Studies and Analytical Field Studies (Yrjanheikki EJ, ed). Environmental Health Series Rpt 34. Copenhagen:World Health Organization Regional Office for Europe, 1989.
9. Noren K, Meironyte D. Contaminants in Swedish human milk. Decreasing levels of organochlorine and increasing levels of organobromine compounds. Organohalogen Compounds 38:1-4 (1998).
10. IARC. Polychlorinated dibenzo-para-dioxins. In: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol 69: Polychlorinated Dibenzo-dioxins and Dibenzofurans. Lyon: International Agency for Research on Cancer, 1997;33-636.
11. Hooper K, Petreas MX, She J, Visita P, Winkler J, McKinney M, Cheng YY, Reisberg B, Ruhstaller K. Analysis of Breast Milk to Assess Exposure to Chlorinated Contaminants in Ethnically Diverse Populations of Stocktom, California. HML Report. Berkeley, CA:Hazardous Materials Laboratory, 1999.
12. Rogan WR, Gladen BC, McKinney JD, Carreras RN, Hardy P, Thullen J, Tingelstad J, Tully M. Polychlorinated biphenyls (PCBs) and dichlorodiphenyl dichloroethane (DDE) in human milk: effects of maternal factors and previous lactation. Am J Public Health 76:172-177 (1986).
13. Beck H, Dross A, Mathar W. PCDD and PCDF exposure and levels in humans in Germany. Environ Health Perspect 102(suppl ):173-185 (1994).
14. Landi MT, Consonni D, Patterson DG Jr, Needham LL, Lucier G, Brambilla P, Cazzaniga MA, Mocarelli P, Pesatori AC, Bertazzi PA, et al. 2,3,7,8-Tetrachlorodibenzo-p-dioxin plasma levels in Seveso 20 years after the accident. Environ Health Perspect 106:273-277 (1998).
Last Updated: April 22, 1999 |
|
 |
|
| |

