Bisphenol-A Dental Sealants: The Inappropriateness of Continued Reference to a Single Female Patient
Olea et al. (1 ) have described the collection, chemical anlaysis, and bioassay using MCF-7 cells in vitro of saliva taken from dental patients treated with 50 mg of a dental sealant based on bisphenol-A (BPA). Saliva was collected over the hour immediately following treatment, and the main sample analyzed was shown to contain 231 µg BPA in 27 ml of saliva. Olea et al. added the following statement to the Results section of their paper: "A subject initially selected for treatment had been treated with tooth sealant 2 years earlier; chromatograms demonstrated the presence of bisphenol-A (66.4 µg) and bisphenol-A dimethacrylate (40.2 µg) in her saliva before the second treatment." Olea et al. did not conclude that the BPA derived from the dental treatment made 2 years earlier, but that was implied.
Nagel et al. (2 ) have described how they hand-fed 2 ppb or 20 ppb of BPA in corn oil, using a micropipettor, to pregnant mice between days 11 and 17 of pregnancy. The male pups of these animals had increased prostate weights at 6 months of age. When interpreting these data in terms of the potential hazard of human exposure to BPA-based dental sealants, Nagel et al. (2 ) referred again to the female patient described by Olea et al. (1 ) as follows: "They (Olea et al.) also measured bisphenol A in the saliva of an individual who had tooth sealant applied 2 years earlier and found 66.4 µg in a 1-hr saliva collection before additional sealant treatment, suggesting that bisphenol A may be continually released after the initial dental work." Here, there is a clear implication that the BPA derived from the dental work carried out 2 years previously.
Olea et al. (1 ) noted that most of the unpolymerized sealant is leached from the polymerized sealant within 24 hr. Given that saliva is constantly swallowed and that phenols such as BPA would be expected to be rapidly excreted, the relatively high levels of BPA found in the saliva of the patient treated 2 years previously must represent only a fraction of the levels present immediately after the initial dental treatment. However, even assuming constant leaching of the BPA (66 µg/hr over 2 years), the original amount of sealant used would have been in excess of a gram. If, as is likely, the rate of leaching was not constant, the original treatment must have involved the application of many grams of the sealant. Such an unusual treatment would require careful documentation before it is used as a precendent for human hazard assessment, and in the absence of such documentation, it is suggested that this anecdote should not be referred to again in scientific papers.
The need for a balanced and scientific evaluation of the hazard posed to humans by dental treatments that use BPA-based resins is enhanced in some countries (such as the United Kingdom) where mercury-based amalgums are still routinely employed for restorative dental work. The real need in such situations is for appropriate relative risk assessments to be undertaken mindful of the effectiveness of the resin-based treatments (3 ). Faced with that need, continued and undocumented reference to the single female patient encounted by Olea et al. (1 ) is unwarranted.
John Ashby
Zeneca Central Toxicology Laboratory
Alderley Park, Cheshire, United Kingdom
References
1. Olea N, Pulgar R, Pérez P, Olea-Serrano F, Rivas A, Novillo-Fertrell A, Pedraza V, Soto AM, Sonnenschein C. Estrogenicity of resin-based composites and sealants used in dentistry. Environ Health Perspect 104:298-305 (1996).
2. Nagel SC, vom Saal FS, Thayer KA, Dhar MG, Boechler M, Welshons WV. Relative binding affinity-serum modified access (RBA-SMA) assay predicts the relative in vitro bioactivity of the xenoestrogens bisphenol A and octylphenol. Environ Health Perspect 105:70-76 (1997).
3. Ripa LW. Sealants revisited: an update of the effectiveness of pit and fissure sealants. Caries Res 27:77-82 (1993).
 2µ,
2µ,  2u, or
2u, or  2u
2u
I enjoy reading Environmental Health Perspectives; the journal is both highly informative and entertaining. Congratulations for a job well done! However, in the December 1996 issue of Volume 104, I came across an error that, in the interest of scientific accuracy, needs to be corrected.
On pages 1264-1267 [EHP 104(12)], there is a lively discussion between John Ashby and James Huff on an issue that is of interest to many of us: nephropathy, white ravens (not rats!), and mechanisms. Prominent in both title and text and, above all, consistently, the text refers to a protein called 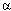 2µ (as in micro or macro) globulin. I think the term
2µ (as in micro or macro) globulin. I think the term 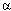 2µ globulin is a misnomer. The protein in question is called
2µ globulin is a misnomer. The protein in question is called 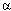 2u globulin (u as in urine)--or at least was called this way in the past. Two reviews that are readily available in any library (1,2) not only refer consistently to
2u globulin (u as in urine)--or at least was called this way in the past. Two reviews that are readily available in any library (1,2) not only refer consistently to 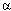 2u globulin by its correct name but also provide the necessary references to trace the origin of this name. In reading some of the earliest papers dating back to 1966, it becomes indeed obvious that the u stands for urinary.
2u globulin by its correct name but also provide the necessary references to trace the origin of this name. In reading some of the earliest papers dating back to 1966, it becomes indeed obvious that the u stands for urinary.
It is easy to see how the mistake in terminology can happen--most of us are too lazy to consistently go through a few functions in our word processing program, if it can be avoided, to print a µ (in my program it takes seven strokes) and thus usually type a u instead--like in um, which reads micrometer, or umole for micromole. Also, we are often too lazy to subscript letters. Therefore, in typesetting an 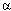 2u easily becomes an
2u easily becomes an 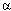 2µ. But it definitely is incorrect.
2µ. But it definitely is incorrect.
If it is of any consolation to you, you are in good company. IARC Scientific Publications No. 116, in chapters written by Huff (3 ) and by Swenberg et al. (4 ), consistently commits the same error in both text and references. And when, on a hunch, I checked one of the usually superbly edited National Research Council documents, I discovered the same error; in "Science and Judgement in Risk Assessment" (5 ), it consistently spells alpha-2µ-globulin, although in the reference list [see EPA (6 )], the spelling is correct. This is embarrassing since I was a member of the committee.
Environmental Health Perspectives is thus not the only one having made the error, but others did so as well. It is an error that should not be further propagated.
Hanspeter Witschi
Institute of Toxicology and
Enviromental Health
University of California, Davis
Davis, California
References
1. Borghoff SJ, Short BG, Swenberg JA. Biochemical mechanisms and pathobiology of alpha 2u-globulin nephropathy. Ann Rev Pharmacol Toxicol 30:349-367 (1990).
2. Swenberg JA, Short B, Borghoff S, Strasser J, Charbonneau M. The comparative pathobiology of alpha 2u-globulin nephropathy. Toxicol Appl Pharmacol 97:35-46 (1989).
3. Huff JE. Chemical toxicity & chemical carcinogenesis. Is there a causal connection? A comparative morphological evaluation of 1500 experiments. In: Mechanisms of carcinogenesis in risk identification (Vainio H, Magee PN, McGregor DB, McMichael AJ, eds), IARC scientific publication no. 116. Lyon:International Agency for Research on Cancer, 1992;437-475.
4. Swenberg JA, Dietrich DR, McClain RM, Cohen SM. Species-specific mechanisms of carcinogenesis. In: Mechanisms of carcinogenesis in risk identification (Vainio H, Magee PN, McGregor DB, McMichael AJ, eds), IARC scientific publication no. 116. Lyon: International Agency for Research on Cancer, 1992;477-500.
5. Committee on Risk Assessment of Hazardous Air Pollutants, Board on Environmental Studies and Toxicology, Commission on Life Sciences, National Research Council. Science and judgement in risk assessment. Washington, DC:National Academy Press, 1994;92-94.
6. EPA. Alpha-2u-globulin: association with chemically-induced renal toxicity and neoplasia in the male rat. EPA-625/3-91/019F. Washington, DC:U.S. Environmental Protection Agency, 1991.
Note: John Ashby agrees that the use of 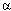 2µ/
2µ/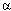 2u/
2u/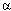 2u is a problem. His use of
2u is a problem. His use of 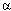 2µ is based on IARC Scientific Publications No. 116 ("Mechanisms of Carcinogenesis in Risk Identification").
2µ is based on IARC Scientific Publications No. 116 ("Mechanisms of Carcinogenesis in Risk Identification").
Last Update: April 4, 1997





