Phase II Begins: May 2004
In May 2004, a partnership of four federal agencies -- the Agency for Healthcare Research and Quality (AHRQ), the Centers for Disease Control and Prevention (CDC), the Centers for Medicare & Medicaid Services (CMS), and
NCI -- completed contract negotiations with the National Quality Forum (NQF) to begin Phase II of
the Cancer Quality of Care Measurement Project.
During this phase, the project conducted evidence-based reviews of available measures
of cancer care quality to identify measures that are ready for use. The project also
charted strategies for developing and testing new measures (though the development of new
measures was beyond the scope of Phase II).
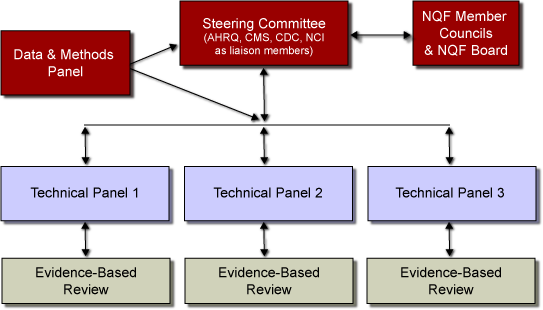
The Steering Committee for Phase II was convened (by conference call) in July 2004. At
that time, the Committee selected three topics areas, from the seven priority areas identified in Phase I, as the focus of
the Phase II analyses. These topic areas were symptom management and end-of-life care,
breast cancer diagnosis and treatment, and colorectal cancer diagnosis and treatment.
For each topic area selected, NQF created a Technical Panel to evaluate candidate
quality measures. These Technical Panels were informed by a thorough assessment of
existing quality measures carried out by an AHRQ-funded evidence-based review. In
addition, NQF staff issued a public call for measures for each topic area. The Panels
evaluated the evidence from all sources and made recommendations to the Steering Committee
on the suitability of candidate measures of cancer care quality and on future research
priorities. A Data and Methods Panel also provided overarching guidance to the Technical
Panels and the Steering Committee on the criteria for evaluating evidence and recommending
candidate measures.
Cancer Quality of Care Measures Project
Phase II: Framework for 2004-2007
 [D]
[D]
The Steering Committee delivered its recommendations to the NQF for consideration
through its Consensus Development Process. NQF's four Member Councils and Board of
Directors reviewed and voted on the Steering Committee's recommendations. NQF will use the
results of the voting to publish core measures of cancer care quality as voluntary
consensus standards.
The following six breast cancer quality of care performance measures were endorsed:
- post-breast conserving surgery irradiation;
- adjuvant chemotherapy;
- adjuvant hormonal therapy;
- college of American Pathologists Breast Cancer Protocol;
- needle biopsy diagnosis; and
- patients with early stage breast cancer who have evaluation of the axilla.
Four colorectal cancer performance measures also were endorsed:
- adjuvant chemotherapy for stage III disease;
- completeness of pathology reporting;
- college of American Pathologists Colon and Rectum Protocol; and
- surgical resection including at least 12 nodes.
Finally, nine symptom management, end of life measures were endorsed:
- family evaluation of health care survey indicators;
- comfortable dying;
- chemotherapy in the last 14 days of life;
- more than one emergency room visit in the last 30 days of life;
- more than one hospitalization in the last 30 days of life;
- intensive Care Unit admission in the last 30 days of life;
- not admitted to hospice;
- admitted to hospice for less than three days; and
- death in an acute care setting.
Through this open and rigorous assessment of the evidence, and a deliberative process
that brings a wide range of interested parties to the table, NCI believes this project
will foster consensus about what constitutes quality cancer care in several important
domains. This sets the stage for further advances in quality assessment and improvement,
all aimed at reducing the suffering and death due to cancer.
|

