
| |  | |  | |
| Arsenic Concentrations in Well Water and Risk of Bladder and Kidney Cancer in Finland Päivi Kurttio,1 Eero Pukkala,2 Hanna Kahelin,3 Anssi Auvinen,4 and Juha Pekkanen1 1National Public Health Institute, Unit of Environmental Epidemiology, Kuopio, Finland
2Finnish Cancer Registry, Helsinki, Finland
3Geological Survey of Finland, Espoo, Finland
4Radiation and Nuclear Safety Authority, Helsinki, Finland Abstract
We assessed the levels of arsenic in drilled wells in Finland and studied the association of arsenic exposure with the risk of bladder and kidney cancers. The study persons were selected from a register-based cohort of all Finns who had lived at an address outside the municipal drinking-water system during 1967-1980 (n = 144,627) . The final study population consisted of 61 bladder cancer cases and 49 kidney cancer cases diagnosed between 1981 and 1995, as well as an age- and sex-balanced random sample of 275 subjects (reference cohort) . Water samples were obtained from the wells used by the study population at least during 1967-1980. The total arsenic concentrations in the wells of the reference cohort were low (median = 0.1 µg/L ; maximum = 64 µg/L) , and 1% exceeded 10 µg/L. Arsenic exposure was estimated as arsenic concentration in the well, daily dose, and cumulative dose of arsenic. None of the exposure indicators was statistically significantly associated with the risk of kidney cancer. Bladder cancer tended to be associated with arsenic concentration and daily dose during the third to ninth years prior to the cancer diagnosis ; the risk ratios for arsenic concentration categories 0.1-0.5 and  0.5 µg/L relative to the category with < 0.1 µg/L were 1.53 [95% confidence interval (CI) , 0.75-3.09] and 2.44 (CI, 1.11-5.37) , respectively. In spite of very low exposure levels, we found some evidence of an association between arsenic and bladder cancer risk. More studies are needed to confirm the possible association between arsenic and bladder cancer risk at such low exposure levels. Key words: arsenic, bladder cancer, drinking water, kidney cancer, rural, urinary organ cancers, well water. Environ Health Perspect 107:705-710 (1999) . [Online 27 July 1999] 0.5 µg/L relative to the category with < 0.1 µg/L were 1.53 [95% confidence interval (CI) , 0.75-3.09] and 2.44 (CI, 1.11-5.37) , respectively. In spite of very low exposure levels, we found some evidence of an association between arsenic and bladder cancer risk. More studies are needed to confirm the possible association between arsenic and bladder cancer risk at such low exposure levels. Key words: arsenic, bladder cancer, drinking water, kidney cancer, rural, urinary organ cancers, well water. Environ Health Perspect 107:705-710 (1999) . [Online 27 July 1999] http://ehpnet1.niehs.nih.gov/docs/1999/107p705-710kurttio/ abstract.html Address correspondence to P. Kurttio, National Public Health Institute, Unit of Environmental Epidemiology, PO Box 95, FIN-70701 Kuopio, Finland. Telephone: 358-17-201 362. Fax: 358-17-201 155. E-mail: paivi.kurttio@ktl.fi We thank E. Kokki, M. Vahteristo, and P. Tiittanen from the National Public Health Institute ; T. Luostarinen from the Finnish Cancer Registry ; and the local environmental health personnel and the study subjects. The Academy of Finland, Research Council of Health (grant 34213) , the Yrjö Jahnsson Foundation (grants 3772 and 4133) , and the Environment, Health and Society Programme of the University of Kuopio financially supported the study. Received 1 February 1999 ; accepted 7 May 1999. |
|
|
 |
Natural mineral deposits containing arsenic may result in elevated levels of arsenic in ground water. The World Health Organization (WHO) classifies inorganic arsenic as a human carcinogen; chronic ingestion is associated with skin cancer and inhalation is associated with lung cancer ( 1). Arsenic concentrations in noncontaminated drinking water are usually below 3 µg/L ( 1- 4). The present WHO drinking-water guideline value of 10 µg/L ( 4) is based mainly on epidemiologic studies on arsenic exposure and skin cancer in Taiwan.
Epidemiologic studies based on high arsenic exposure in Taiwan (5-11), Argentina (12,13), Japan (14,15), and Chile (16) suggest an increased risk of cancer of the urinary organs. The lowest arsenic exposure levels associated with detrimental health effects have been reported in a case-control study in the United States in which the arsenic concentrations in drinking water ranged from 0.5 to 180 µg/L (17). In that study, no overall association was found between arsenic exposure and cancer risk, but arsenic and cigarette smoking tended to have a synergistic effect on the risk of bladder cancer.
As much as 12-14% of the Finnish population live outside areas with municipal water supply and use private wells (18). Water quality of private wells is seldom monitored. Concentrations of arsenic measured in a rural town in central Finland were up to 100 times higher than drinking-water quality guideline values (19). According to geologic surveys, there are several other areas in Finland that are prone to high arsenic concentrations (20), but no countrywide survey on arsenic concentrations in well water has been carried out. The aims of the present study were to determine whether arsenic exposure from well water is associated with increased risk of bladder and kidney cancers and to assess the concentrations of arsenic in drilled wells used for drinking water in Finland.
Selection of study population. Town quarters or villages, in which less than 10% of the population belong to the municipal drinking-water system, were identified based on the 1985 Population Census file of the Statistics Finland. The source population was defined from the Population Registry as 144,627 persons born in 1900-1930 who had lived in these areas at the same address at least from 1967 to 1980 (Figure 1). The year 1967 was chosen because it was the earliest for which information on residency had been recorded systematically.
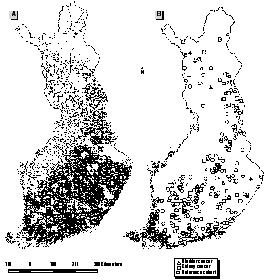
During 1981-1995, 884 bladder cancer cases and 644 kidney cancer cases were identified within the cohort through a record link with the Finnish Cancer Registry. Because there are plans to conduct similar studies based on the same reference cohort for stomach cancer and leukemia, a case-cohort design was used (21). Random selection of the reference cohort was stratified by single birth year (range 1900-1930) and sex (i.e., 62 strata). The number of persons selected to the reference cohort within each stratum was four times the highest number of site-specific cancer cases in that stratum (i.e., either kidney, bladder, leukemia, or stomach cancer). Altogether, 4,590 persons were selected in the reference cohort.
Final study series. The current residents at the addresses occupied by the case and referent persons in 1967-1980 were contacted initially either by the local health inspectors or by mailed questionnaire. The following questions were asked: Did the house have a drilled well? When was the drilled well established? Had the drilled well been used as the primary source of drinking water?
Because drilled wells have been reported to contain essentially higher arsenic concentrations than other types of wells (22), only those addresses where drinking water from drilled wells was consumed before the year 1981 were selected for further study (Table 1). The use of the drilled wells for drinking water had started before 1981 but may have continued until 1996. Local health inspectors collected well-water samples from the selected addresses of 509 subjects.

Those 380 subjects (76% of cases and 74% of reference cohort eligible) for whom arsenic exposure could be estimated (i.e., the well-water sample was available and well water had been used as drinking water) were included in the final analysis (Table 1). The excluded and included subjects did not differ by vital status (40%/52% alive), age (mean birth year 1917/1916), sex (42%/39% women), occupation (61%/64% farmers), or education (2%/5% with more than 9 years of education). Sixty-one cases with bladder cancer and 49 with kidney cancer were included (including 3 cases with cancer of the renal pelvis, 41 with unilateral kidney cancer, 1 with cancer in both kidneys, and 4 with cancer in an unidentified site). The reference cohort consisted of 275 subjects (including 3 subjects who developed bladder cancer and 1 subject who was diagnosed with kidney cancer).
Questionnaire. Information on residential history and drinking-water consumption at the selected addresses and potential confounders such as smoking, use of analgesics and diuretics in the 1970s, education, and occupation was collected using a questionnaire mailed to the study subjects (i.e., cases and referents who had lived at the selected addresses during 1967-1980) or their next of kin (Tables 2 and 3). Subjects received the questionnaires before the water sampling in 1996. In addition, information on the present occupation (previous occupation for retired persons) and education was obtained from the Population Censuses of 1970, 1975, and 1980.

Arsenic measurements. The well-water samples were collected between July and November 1996. The samples were collected in random order, blinded in regard to the case-referent status. Each sample was filtered through a 0.45-µm membrane filter and acidified with HNO3 at the sample site. The samples were sent to the laboratory within 2 days.
Total arsenic was determined by Perkin Elmer Sciex Elan 6000 inductively coupled plasma mass spectrometry (Perkin-Elmer, Rotkreuz, Switzerland). The detection limit for arsenic was 0.05 µg/L. The performance of the analytical method was monitored continuously including certified reference materials within the analytical batches. Sixty-four of the samples were analyzed twice. The median coefficient of variation was 2.2%.
To evaluate the validity of water sampling, two water samples were taken from 36 randomly selected wells at two different times (on average 31 days apart; range 2 hr-88 days). The arsenic concentrations in the original samples and field duplicates were not significantly different (median of the absolute difference between two samples was 0.02 µg/L; p = 0.2 in the Wilcoxon signed ranks test).
Estimates of arsenic exposure. The arsenic exposure for cases with cancer and members of reference cohort was estimated in two periods: from the third to ninth calendar years (shorter latency) and from the tenth or earlier calendar years (longer latency) prior to the cancer diagnosis (or the respective year for referent persons).
The daily dose of arsenic from drinking water was calculated from the arsenic concentration of well water and from the reported consumption of well water in the 1970s. If questionnaire data were not available, the consumption of drinking water was set as the mean from the reference cohort. Men consumed on average 1.6 L/day (range 0.1-4.5 L/day) and women consumed 1.6 L/day (0.6-3.0 L/day) of well water. At two addresses (one member of the reference cohort and one kidney cancer case), water samples from two drilled wells were available. In those cases, the higher arsenic concentration was included in the calculations of arsenic concentration and daily dose, and actual use for cumulative exposure.
Consumption of well water started on average in 1970 [standard deviation (SD) = 8 years] and stopped in 1990 (SD = 6 years). The cumulative dose was defined as an integral of duration and intensity of arsenic exposure from well water. The cumulative dose for the shorter latency was calculated from the beginning of the use of well water until 2 years before the cancer diagnosis. For the longer latency, the cumulative dose was calculated until 10 years before the cancer diagnosis. The arsenic concentration in drinking water before the beginning and after the end of the consumption of the well water was considered as null. Exposure from other sources was not taken into account.
Statistical analyses. The study was based on the case-cohort design described by Prentice (21) and Barlow (23). In the analysis of a case-cohort study, the risk set at each event time (year of cancer diagnosis) consists of the case that failed at that particular time and all members of the reference cohort who were at risk at the time. Members of the reference cohort were weighted in inverse proportion to the sampling fraction (4,590/ 144,627). The robust variance matrix was estimated using SAS/IML (Interactive Matrix Programming Language; SAS Institute, Cary, NC). The 95% confidence intervals (CIs) were calculated from the robust variance estimates and standard errors.
Risk ratios (RRs) were estimated using PHREG (Cox's Multivariable Proportional Regression Analysis) procedure of SAS, calculated both for continuous and categorical arsenic exposure (class limits set roughly at the 50th and 75th percentiles of the reference cohort). To limit the influence of the few observations with high levels of arsenic, we estimated linear models after log-transforming the arsenic exposure indicator. Cigarette smoking is an established risk factor for both bladder and kidney cancers (24,25) and high body mass index (BMI) is an established risk factor for kidney cancer (26). The multivariate models of bladder cancer were adjusted for age, sex, and smoking (never smoked, ex-smoker stopped smoking before 1970, smoker in the 1970s). The multivariate models of kidney cancer were adjusted for age, sex, smoking, and BMI [< 25,  25 weight in kilograms/(height in meters)2]. Subjects with missing data on smoking and BMI (Table 3) were included in the models as a separate category. Use of analgesics and diuretics in the 1970s and education were not associated with the risk of bladder or kidney cancers and were therefore not included in the final models. 25 weight in kilograms/(height in meters)2]. Subjects with missing data on smoking and BMI (Table 3) were included in the models as a separate category. Use of analgesics and diuretics in the 1970s and education were not associated with the risk of bladder or kidney cancers and were therefore not included in the final models.
Arsenic concentrations in reference wells. The arsenic concentrations in the wells of the reference cohort ranged from < 0.05 to 64 µg/L (median 0.14 µg/L; CI, < 0.05-4.5; Table 4). Five percent of the reference cohort had arsenic concentrations > 5 µg/L, and 1% (4/275) had consumed well water exceeding the WHO drinking-water quality guideline value of 10 µg/L. The median daily dose of arsenic from well water was 0.2 µg (5th and 95th percentiles were 0.04 and 7 µg, respectively). The cumulative dose before 1980 was 0.8 mg (5th and 95th percentiles were 0.08 and 26 mg, respectively).

Ninety-four percent of the drilled wells were still in use in 1996. The depth of the well (median 42 m, range 5-172 m) did not correlate (Pearson correlation coefficient 0.06) with the (log-transformed) arsenic concentrations. Water treatment was rare: it was reported for only 11 wells, most frequently for iron removal.
Characteristics of cases versus controls. The study population was a demographically rather homogeneous group of people with a mainly agricultural background (Table 2). The residence locations of cancer cases and referents did not differ systematically (Figure 1). Residential mobility was remarkably low; 71% of those alive in 1996 still lived in the same place where they had lived in 1967. Cigerette smoking in the 1970s was more common among the cancer cases than among the reference cohort; the association between smoking and bladder cancer was statistically significant (Tables 3 and 5).

Arsenic and bladder cancer. We observed an increasing trend for arsenic concentrations in drinking water and the daily dose of arsenic with shorter latency, but not with longer latency (Table 6). The point estimate for arsenic concentration > 0.5 µg/L was statistically significantly elevated. The association between arsenic exposure and bladder cancer tended to be stronger among those who smoked in the 1970s (Table 7).


Arsenic and kidney cancer. We found no evidence for an association between arsenic concentration, daily dose, or cumulative dose and the risk of kidney cancer (Table 8).

High concentrations of arsenic in drilled wells were previously detected in southwestern Finland (19,22), and geochemical surveys (20) suggest high arsenic concentrations in other areas as well. However, our findings indicate that substantial arsenic exposure through drilled well water in Finland is uncommon. Assuming that the entire arsenic dose from drinking water was in inorganic form, the median dose of inorganic arsenic from well water in the reference cohort was approximately 0.2 µg/day. This is similar to the estimated dose from drinking water in the Czech Republic (2), but lower than that estimated in the United States (4 µg/day) (27).
We have previously shown that the steady-state current arsenic exposure can be adequately estimated by asking subjects about daily drinking-water consumption at home (19). In the present study, however, we were interested in the water consumption in the 1970s, which is more prone to misclassification and affected by recall bias. The earlier arsenic concentrations were considered to be the same as they were at the time of the sampling, which may introduce errors. No published data on annual variations of arsenic concentrations in well water in Finland were available, but the concentrations may fluctuate depending on the water catchment basin of the well (i.e., the level of groundwater). Some studies have, however, reported that arsenic concentrations in wells in arsenic-rich areas remain relatively constant for decades (11,12).
The calculation of cumulative dose is more sensitive to errors than daily dose or concentration. The year of beginning of well-water use may not have been recalled adequately, and an error in the duration of well-water use has a significant influence on the amount of cumulative dose. The arsenic concentrations in drinking water other than those measured from drilled wells were considered zero. This may introduce misclassification in the cumulative dose, especially when arsenic concentrations in drilled wells were as low as in other sources of drinking water. This misclassification, however, is likely to be nondifferential.
The arsenic doses found in previous studies (5-17) on arsenic exposure and cancer in urinary organs were much higher than those found in this study. In the present study, we used individual arsenic exposure variables instead of area-based measures used in most previous studies. Studies in Taiwan have suggested that cancers of the bladder, kidney, lung, liver, skin, and possibly some other sites are associated with drinking-water arsenic concentrations ranging from 10 to 1,800 µg/L (5-8,28) and from 350 to 1,140 µg/L (9). Furthermore, ingested arsenic exposure has been associated with the increased risk of cancers in urinary organs in Argentina, Japan, and Chile. The lowest cut points of the categorical arsenic concentrations were 40 µg/L in Argentina (12,13) and 50 µg/L in Japan (14,15). In Chile, the regional average arsenic concentrations in drinking water ranged from 43 to 570 µg/L (16). The only earlier nonecological study on bladder cancer risk and lower arsenic concentrations (from 0.5 to 180 µg/L) in drinking water in the United States did not show a clear association (17). A follow-up study among English patients showed that orally administered arsenic medication with substantially higher doses was associated with an increase in bladder cancer mortality (29).
We found some suggestion of an increased bladder cancer risk associated with low arsenic exposure levels in the present study. The risk estimates for bladder cancer and arsenic concentration, as well as daily dose from 2 to 9 years before the cancer diagnosis, were above unity, and there was some indication of a dose response. A statistically significantly elevated risk of bladder cancer was observed for arsenic concentrations > 0.5 µg/L. Exposures earlier than 10 years before cancer diagnoses did not show an association with bladder cancer risk. Hence, relatively recent arsenic exposure appears to be more relevant for bladder cancer risk. This is in concordance with the hypothesis that arsenic compounds act as promoters and/or co-carcinogens in the late stage of carcinogenesis (30-32).
Cigarette smoking in the 1970s was more common among bladder cancer cases than among referents; this is in agreement with previous studies which reported that smoking is associated with bladder cancer (25). In addition, there was some suggestion of a synergistic effect of arsenic and smoking (Table 7). This finding is consistent with earlier studies in which elevated arsenic exposure tended to increase the bladder cancer risk (9,17) among smokers. Experimental studies also suggest that arsenic compounds promote the carcinogenicity and genotoxicity of the known carcinogens and genotoxic compounds (31-33).
In this study, no association was observed between kidney cancer and arsenic exposure from drinking water. In earlier epidemiologic studies, an association between arsenic exposure and kidney cancer was somewhat weaker than the association to bladder cancer (6-8,10,11,13,16).
It is difficult to compare the cancer risks between various studies because arsenic exposure has been expressed in different ways and because the extension and duration of arsenic exposure in these studies are different. The differences in the effect estimates in the studies may also be due to the differences in the possible misclassification of arsenic exposure, other sources of arsenic or other carcinogens, genetic background, health status, or nutritional status. Most of the previous studies have had a cancer mortality as an end point. The participants in this study were older than persons in previous epidemiologic studies. Applying the results for cancer mortality obtained by Smith et al. (3) from Taiwanese data, the highest exposure categories in the present study population (median arsenic concentration 1.99 µg/L) would have a relative risk for bladder cancer of 1.01 in men and 1.03 in women. Hence, the present results, relative risk > 2, are higher than expected and raise a concern of the possible role of bias or chance.
Nutritional factors may modify the metabolism and possibly carcinogenicity of arsenic. Selenium is an important anticarcinogen (34), and it has been suggested to reduce the toxicity of arsenic (35) and to inhibit the methylation of inorganic arsenic in vitro (36). Therefore, deficiency of selenium may increase the cancer risk of arsenic. In Finland, the daily intake of selenium (average 30 µg) (37) was below the recommended 50-200 µg (38) before selenium supplementation in fertilizers started in 1985.
The crude risk ratios of arsenic exposure and bladder cancer increased only slightly after adjustment for smoking. This suggests that, although the proportion of missing data on smoking was large, the results are probably not confounded by smoking. We believe that serious differential misclassification is unlikely in the present study. There were no differences between cases and noncases in the sampling or analyzing procedure. Furthermore, there were no differences in the demographic parameters of the eligible and the final study populations. It has previously been shown that relatives are able to provide rather reliable information on smoking and other major demographic and lifestyle factors (39-41).
Our results show that high arsenic levels in Finnish drilled wells used in the 1970s were uncommon and exposure through drinking water was low. We found no statistically significant association between arsenic and risk of kidney cancer. However, consistent with earlier studies, there was some evidence for an increased risk of bladder cancer associated with arsenic exposure 2-9 years before the diagnosis and some suggestion of a synergistic effect between arsenic and smoking. Due to low exposure levels in the present study, the positive association between arsenic exposure and risk of bladder cancer was not expected, and the role of bias and chance needs to be carefully considered.
|
|
 |
| [References Listed in PubMed]
References and Notes
1. IARC. Arsenic and arsenic compounds. IARC Monogr Eval Carcinog Risk Chem Hum 23:39-141 (1980).
2. Kliment V. Model of multiple exposure to contaminants in monitoring the environmental impact on population health. Cent Eur J Public Health 4:246-249 (1996).
3. Smith AH, Hopenhayn-Rich C, Bates MN, Goeden HM, Hertz-Picciotto I, Duggan HM, Wood R, Kosnett MJ, Smith MT. Cancer risks from arsenic in drinking water. Environ Health Perspect 97:259-267 (1992).
4. WHO. Guidelines for Drinking-Water Quality. 2nd ed. Geneva:World Health Organization International Programme on Chemical Safety, 1996;156-167.
5. Tseng W-P. Effects and dose-response relationships of skin cancer and Blackfoot disease with arsenic. Environ Health Perspect 19:109-119 (1977).
6. Chen C-J, Kuo TL, Wu M. Arsenic and cancers. Lancet 1:414-415 (1988).
7. Chen C-J, Chen CW, Wu M-M, Kuo T-L. Cancer potential in liver, lung, bladder and kidney due to ingested inorganic arsenic in drinking water. Br J Cancer 66:888-892 (1992).
8. Wu MM, Kuo TL, Hwang Y, Chen C. Dose-response relation between arsenic concentration in well water and mortality from cancers and vascular diseases. Am J Epidemiol 130:1123-1132 (1989).
9. Chiou HY, Hsueh YM, Liaw KF, Horng SF, Chiang MH, Pa YS, Lin JS, Huang CH, Chen CJ. Incidence of internal cancers and ingested inorganic arsenic: a seven-year follow-up study in Taiwan. Cancer Res 55:1296-1300 (1995).
10. Chen C-J, Wang CJ. Ecological correlation between arsenic level in well water and age-adjusted mortality from malignant neoplasms. Cancer Res 50:5470-5474 (1990).
11. Guo H-R, Chiang H-S, Hu H, Lipsitz SR, Monson RR. Arsenic in drinking water and incidence of urinary cancers. Epidemiology 8:545-550 (1997).
12. Hopenhayn-Rich C, Biggs ML, Fuchs A, Bergoglio R, Tello EE, Nicolli H, Smith AH. Bladder cancer mortality associated with arsenic in drinking water in Argentina. Epidemiology 7:117-124 (1996).
13. Hopenhayn-Rich C, Biggs ML, Smith AH. Lung and kidney cancer mortality associated with arsenic in drinking water in Cordoba, Argentina. Int J Epidemiol 27:561-569 (1998).
14. Tsuda T, Nagira T, Yamamoto M, Kume Y. An epidemiological study on cancer in certified arsenic poisoning patients in Toruku. Ind Health 28:53-62 (1990).
15. Tsuda T, Babazono A, Yamamoto E, Kurumatani N, Mino Y, Ogawa T, Kishi Y, Aoyama H. Ingested arsenic and internal cancer: a historical cohort study followed for 33 years. Am J Epidemiol 141:198-209 (1995).
16. Smith AH, Goycolea M, Haque R, Biggs ML. Marked increase in bladder and lung cancer mortality in a region of northern Chile due to arsenic in drinking water. Am J Epidemiol 147:660-669 (1998).
17. Bates MN, Smith AH, Cantor KP. Case-control study of bladder cancer and arsenic in drinking water. Am J Epidemiol 111:523-530 (1995).
18. Kujala-Räty K, Hiisvirta L, Kaukonen M, Liponkoski M, Sipilä A. Quality of Household Water in Finland in 1996 [in Finnish]. Finnish Environment 181. Helsinki:Edita Ltd, 1998.
19. Kurttio P, Komulainen H, Hakala E, Kahelin H, Pekkanen J. Urinary excretion of arsenic species after exposure to arsenic present in drinking water. Arch Environ Contam Toxicol 34:297-305 (1998).
20. Lahermo P, Väänänen P, Tarvainen T, Salminen R. Geochemical Atlas of Finland, Part 3: Environmental Geochemistry--stream waters and sediments [in Finnish]. Espoo, Finland:Geological Survey of Finland, 1996.
21. Prentice RL. A case-cohort design for epidemiologic cohort studies and disease prevention trials. Biometrika 73:1-11 (1986).
22. Lahermo P, Alfthan G, Wang D. Selenium and arsenic in the environment in Finland. J Environ Pathol Toxicol Oncol 17:205-216 (1998).
23. Barlow WE. Robust variance estimation of case-cohort design. Biometrics 50:1064-1072 (1994).
24. Dreyer L, Winther JF, Pukkala E, Andersen A. Tobacco smoking. APMIS Suppl.76:9-47 (1997).
25. McLaughlin JK, Blot WJ, Devesa SS, Fraumeni JF Jr. Renal cancer. In: Cancer Epidemiology and Prevention (Schottenfeld D, Fraumeni JF Jr, eds). New York:Oxford University Press, 1996;1142-1155.
26. Muscat JE, Hoffmann D, Wynder EL. The epidemiology of renal cell carcinoma: a second look. Cancer 75:2552-2557 (1995).
27. Valberg PA, Beck BD, Bowers TS, Keating JL, Bergstrom PD, Boardman PD. Issues in setting health-based cleanup levels for arsenic in soil. Reg Toxicol Pharmacol 26:219-229 (1997).
28. Tseng WP, Chu HM, How S, Fong J, Lin C, Yeh S. Prevalence of skin cancer in an endemic area of chronic arsenicism in Taiwan. J Natl Cancer Inst 40:453-463 (1968).
29. Cuzick J, Sasieni P, Evans S. Ingested arsenic, keratoses and bladder cancer. Am J Epidemiol 136:417-421 (1992).
30. Brown JL, Kitchin KT. Arsenite, but not cadmium, induces ornithine decarboxylase and heme oxygenase activity in rat liver: relevance to arsenic carcinogenesis. Cancer Lett 98:227-231 (1996).
31. Mass MJ, Wang L. Arsenic alters cytosine methylation patterns of the promoter of the tumor suppressor gene p53 in human lung cells: a model for a mechanism of carcinogenesis. Mutat Res 386:263-277 (1997).
32. Li JH, Rossman TG. Mechanism of comutagenesis of sodium arsenite with n-methyl-n-nitrosourea. Biol Trace Elem Res 21:373-381 (1989).
33. Wang TC, Huang JS, Yang VC, Lan HJ, Lin CJ, Jan KY. Delay of the excision of UV light-induced DNA adducts is involved in the coclastogenicity of UV light plus arsenite. Int J Radiat Biol 66:367-372 (1994).
34. Schrauzer GN. Selenium. Mechanistic aspects of anticarcinogenic action. Biol Trace Element Res 33:51-62 (1992).
35. Berry JP, Galle P. Selenium-arsenic interaction in renal cells: role of lysosomes. Electron microprobe study. J Submicrosc Cytol Pathol 26:203-210 (1994).
36. Styblo M, Delnomdedieu M, Thomas DJ. Mono- and dimethylation of arsenic in rat liver cytosol in vitro. Chem-Biol Interact 99:147-164 (1996).
37. Varo P, Koivistoinen P. XII. Mineral element composition of Finnish foods. Acta Agric Scand Suppl 22:165-171 (1980).
38. National Public Health Institute. Expert Group on Nutritional Follow-up, Report on Nutrition 1995 [in Finnish]. National Public Health Institute B1/1996. Helsinki:National Public Health Institute, 1996.
39. Boyle CA, Brann EA. Proxy respondents and the validity of occupational and other exposure data. The Selected Cancers Cooperative Study Group. Am J Epidemiol 136:712-721 (1992).
40. Lyon JL, Egger MJ, Robison LM, French TK, Gao R. Misclassification of exposure in a case-control study: the effects of different types of exposure and different proxy respondents in a study of pancreatic cancer. Epidemiology 3:223-231 (1992).
41. Nelson LM, Longstreth WT Jr, Koepsell TD, Checkoway H, van Belle G. Completeness and accuracy of interview data from proxy respondents: demographic, medical, and life-style factors. Epidemiology 5:204-217 (1994).
Last Updated: July 27, 1999
|
|
 |
|
| |

