Residential pesticide applications represent a potential source of human exposure to pesticides. Limited information is available to guide public health officials in regard to exposure, appropriate reentry intervals, and consumer education. Residential exposure monitoring after indoor pesticide applications has normally consisted of three measurements: air concentrations, house dust concentrations, and surface residues available for transfer to human skin. Recent studies have focused on broadcast and total release aerosol applications for flea control because these applications treat surfaces likely to be contacted by crawling infants or small children. Several techniques have been developed to estimate dermal pesticide exposure in residences, including standardized wipe sampling procedures (
1-3), whole-body garments, and cloth rollers (
4,5). A polyurethane foam (PUF) roller was also developed to simulate the exposure of a crawling infant or walking toddler (
6), and has been used in residences (
7). Hand press studies have been conducted to estimate surface-to-skin transfer processes (
7,8). Each of these techniques has been used under realistic postapplication conditions, but none have been validated. Thus, it is not known to what extent the measurements are representative of the transfer of pesticide residues from treated surfaces to human skin.
The objectives of this study were 2-fold: to compare pesticide transfer estimates from carpet to skin using hand press, hand drag, wipe, and PUF roller methods; and to compare pesticide transfer estimates from target (carpet) and nontarget (furniture) surfaces to skin using hand press and wipe sampling methods.
This investigation consisted of two sequential studies: study 1 involved broadcast and total aerosol release applications (foggers); study 2 used a total aerosol release application. A previous study of these application methods documented air concentrations and surface deposition for 7 days postapplication (3).
Field parameters. Study 1 was conducted in two unoccupied college dormitory rooms. The rooms had identical dimensions: 4.75  3.35
3.35  2.69 m. Total floor surface area was 15.94 m2, and total volume was 42.8 m3. All rooms were carpeted wall-to-wall with 0.64-cm thick nylon pile. Each room contained two desks and one clothing cabinet that were made of wood, and one closet. The floor of each room was divided into 113 sections (1
2.69 m. Total floor surface area was 15.94 m2, and total volume was 42.8 m3. All rooms were carpeted wall-to-wall with 0.64-cm thick nylon pile. Each room contained two desks and one clothing cabinet that were made of wood, and one closet. The floor of each room was divided into 113 sections (1  1 ft), and each section was assigned a unique location identification code. A path that consisted of fourteen 1
1 ft), and each section was assigned a unique location identification code. A path that consisted of fourteen 1  1 ft squares was reserved to provide access to all sampled areas throughout the room. Sections were then selected randomly for deposition or wipe sampling. The rest of the sections were evenly allocated for PUF roller, hand press, or hand drag sampling. Almost the entire floor space was sampled, and each section was only sampled once.
1 ft squares was reserved to provide access to all sampled areas throughout the room. Sections were then selected randomly for deposition or wipe sampling. The rest of the sections were evenly allocated for PUF roller, hand press, or hand drag sampling. Almost the entire floor space was sampled, and each section was only sampled once.
Both broadcast and total release aerosol applications were conducted according to label instructions. The formulated product used for broadcast applications, Dursban L.O. (U.S. Environmental Protection Agency registration 464-571; Dow Elanco, Midland, MI), contained 41.5% chlorpyrifos [O,O-diethyl-O-(3,5,6-trichloro-2-pyridyl) phosphorothioate]. It was applied approximately 40 cm above the carpet as a 0.5% aqueous spray (40 mL/3.785 L water) with a hand-held fan broadcast nozzle attached to a CO2 pressurized tank by a licensed pest control applicator. It took approximately 1 min to spray the entire room. This room was actively ventilated for 30 min immediately after application. Ventilation consisted of opening the two windows and one door and placing a 51  51 cm box fan on a 76-cm table in the doorway. The fan was run on medium speed, drawing air from the room and into the hallway. Windows and doors in rooms across the hallway from the study room were open to allow maximum cross-ventilation. At the end of this active ventilation period the fan was turned off, but the door and windows were left open for the rest of the study.
51 cm box fan on a 76-cm table in the doorway. The fan was run on medium speed, drawing air from the room and into the hallway. Windows and doors in rooms across the hallway from the study room were open to allow maximum cross-ventilation. At the end of this active ventilation period the fan was turned off, but the door and windows were left open for the rest of the study.
The total release aerosol application was conducted with an aerosol canister [K-RID fogger (K-Mart, Troy, MI); EPA registration 9688-93) that contained 0.5% chlorpyrifos by weight. The canister was purchased commercially and weighed before and after application to estimate total mass released. The windows and the door were closed before and for 2 hr after canister activation, as specified on the label. The windows and door were then opened, and the ventilation procedures described above were followed.
In study 2, the field parameters and the total release aerosol application were identical to those described previously. The total surface area of nontarget objects (two desks and one cabinet) was divided into 48 sections and each section was randomly selected for either deposition, wipe, or hand press sampling.
Deposition sampling. Deposition samplers were double layers of 12-ply, 7.6  7.6 cm surgical gauze pads placed on top of aluminum foil. In study 1, 10 samplers were placed on the carpet of each room before application. In study 2, 14 and 6 samplers were placed on the carpet and on furniture surfaces, respectively, before application. All deposition samples were collected 30 min after application.
7.6 cm surgical gauze pads placed on top of aluminum foil. In study 1, 10 samplers were placed on the carpet of each room before application. In study 2, 14 and 6 samplers were placed on the carpet and on furniture surfaces, respectively, before application. All deposition samples were collected 30 min after application.
Wipe sampling. Wipe samples were collected with surgical gauze pads similar to those used for deposition sampling but were moistened by spraying lightly (two mists) with distilled water. Areas to be wiped were marked by metal frames with 10  10 cm cut-out interiors. The 100-cm2 surface area was wiped with three strokes. The wipe was repeated with a second pad, changing the wipe orientation by 90°. The first and second pads were combined for analysis and constituted a single sample. Wipe samples were collected in each room 0.5 and 3.5 hr after application. A disposable plastic glove was worn for each wipe sample to avoid cross-contamination.
10 cm cut-out interiors. The 100-cm2 surface area was wiped with three strokes. The wipe was repeated with a second pad, changing the wipe orientation by 90°. The first and second pads were combined for analysis and constituted a single sample. Wipe samples were collected in each room 0.5 and 3.5 hr after application. A disposable plastic glove was worn for each wipe sample to avoid cross-contamination.
PUF roller sampling. The PUF roller sampling device was rented from the Southwest Research Institute (San Antonio, TX), and all standard protocols provided with the device were followed. PUF roller samples were taken from sections marked at the beginning and end of the 100-cm long area. The PUF ring measured 8 cm in length, resulting in a sampling area of 800 cm2. Toluene-rinsed aluminum foil was placed at the beginning mark of the sampling area. The assembled PUF roller was sprayed with a light mist of distilled water 15 sec before sampling. It was then placed at the edge of the aluminum foil, and the roller was pulled over the 100-cm sampling distance at a rate of approximately 10 cm/sec. The roller assembly was lifted from the sampling area and the left arm affixed in the ring stand clamp for disassembly. The PUF ring was removed from the roller assembly with forceps and tweezers and placed in a 0.5-gal glass jar. The forceps and tweezers were then rinsed with 30 mL of toluene and collected in the same jar. PUF roller samples were collected in each room 3.5 hr after application.
Hand press and hand drag sampling. Hand contact procedures were modifications of those first described by Hsu et al. (6) and Lewis et al. (7). Twelve subjects were recruited for the studies (six per study). They were asked to wash their hands with soap and water 5 min before the study. Each subject then determined the hand pressure to be applied by pressing the entire palm (excluding fingers) on a platform scale. This procedure was repeated several times to determine the amount of pressure required to deflect the scale to approximately 5.4 kg (12 lb), a force estimated to be equivalent to that produced by a crawling infant or walking toddler (6).
At the commencement of sampling, subjects kneeled on a clean rubber mat. Two cardboard frames were placed over the carpet in front of the subject, outlining the surface to be contacted for the right and left hands (Figure 1). The frame was 36 cm wide, providing three 12-cm columns for moving the hand across the surface. Subjects were then instructed to elevate the fingers slightly such that only the palm would contact the surface and the tip of the middle finger was aligned with the top of the guide frame. For the hand press, the palm was pressed against the carpet surface 10 times; 4 times on the outer column and 3 times each on the middle and inner columns. For the hand drag, the palm was then pressed against the carpet and the hand dragged toward the subject until the heel of the hand reached the end of the guide frame. This procedure was conducted over the three columns sequentially.
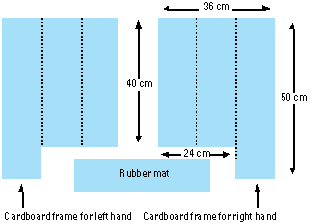
Figure 1. Cardboard guide frame for hand press and hand drag sampling.
In study 2, the assignment of right or left hand to either carpet (target) or furniture (nontarget surface) was made randomly. The hand press on the furniture surface followed the procedures described above; (i.e., 10 surface contacts within the guide frame). The hand press on the carpet surface followed the same procedures, but total surface contacted was increased 5-fold to increase skin loading (i.e., the guide frame was placed on five different carpet areas, resulting in 50 hand press contacts per subject).
An imprint of each hand (palm portion) was collected with water-soluble finger paints at the end of the sampling period to determine the actual contact surface area for each hand. Subjects covered their palms with the paints and then pressed their hands onto white paper following the study procedures. The hand prints were then mounted at a fixed distance from a camera, and digital images were recorded with a video imaging system (9). A standard target of known surface area was included as an internal standard, and the palm area and hand width for each hand print was calculated. For the hand press samples, total surface area contacted was 10 times the palm area. For the hand drag samples, total surface area contacted was calculated by multiplying the hand width by the length of carpet over which the hand drag occurred.
Handwash removal efficiency. Both hand press and hand drag samplings were conducted 3.5 hr after application. Hand washing was conducted within 2 min after sampling, following the procedures of Fenske and Lu (10). A polyethylene bag containing 250 mL of 10% isopropanol/distilled water solution was wrapped around the subject's hand, with the bag closed tightly at the wrist. The subject was asked to let the hand go limp and the hand was shaken for 30 sec (approximately 60 shakes). The handwash solution was poured immediately into a glass jar.
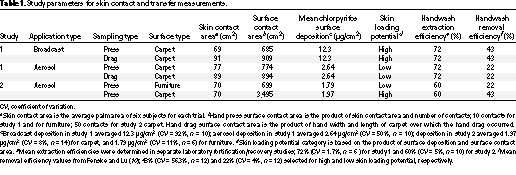
Hand press and hand drag data have been adjusted by a handwash removal efficiency factor, as well as for extraction efficiency (i.e., extraction of chlorpyrifos from the handwash solution, discussed in "Discussion"). These factors are presented in Table 1. Handwash removal efficiency is defined as the amount of chemical removed from the skin by handwashing divided by the total amount of chemical deposited on the skin,  100. The 43 and 22% removal efficiency factors used to adjust data from the present studies were developed in a controlled laboratory study (10). In that study, known amounts of chlorpyrifos were transferred to the hands, and the hands were subsequently washed. The washing procedures, including handwash solution and duration of chlorpyrifos on skin, were identical for the laboratory study and the present study. The laboratory study found that removal efficiency of chlorpyrifos was not constant, but increased with skin loading (mass per unit area). Therefore, hand drag and hand press data collected in the broadcast room and hand press data collected from the carpet surface in study 2 were adjusted by a higher removal efficiency factor (43%) because of higher initial deposition values and the larger contact surface areas, respectively. Hand press data collected in the aerosol rooms in study 1 and from the furniture surfaces in study 2 were adjusted by a lower removal efficiency factor (22%).
100. The 43 and 22% removal efficiency factors used to adjust data from the present studies were developed in a controlled laboratory study (10). In that study, known amounts of chlorpyrifos were transferred to the hands, and the hands were subsequently washed. The washing procedures, including handwash solution and duration of chlorpyrifos on skin, were identical for the laboratory study and the present study. The laboratory study found that removal efficiency of chlorpyrifos was not constant, but increased with skin loading (mass per unit area). Therefore, hand drag and hand press data collected in the broadcast room and hand press data collected from the carpet surface in study 2 were adjusted by a higher removal efficiency factor (43%) because of higher initial deposition values and the larger contact surface areas, respectively. Hand press data collected in the aerosol rooms in study 1 and from the furniture surfaces in study 2 were adjusted by a lower removal efficiency factor (22%).
Analysis. Samples were stored in glass jars at -20°C until analysis. Extraction of gauze pads involved the addition of 30 mL of toluene to the sample jar and agitation on a mechanical shaker table at high speed for 30 min. PUF roller samples were extracted with 210 mL of toluene in a 0.5-gal glass jar. The jar was placed on a rotary tumbler with two parallel rotary arms for 4 hr. A 10-mL aliquot of the handwash solution was placed in a 2-oz sample jar and 250 µL of 1 N HCl was added to the sample jar and shaken by hand for 30 sec. Then 10 mL of toluene was added to this sample jar, which was placed on a shaker table on high speed for 30 min. After 5 min separation time, the solvent layer was drawn off for analysis. Extraction efficiency means and standard deviations were as follows: deposition and wipe samples, 101 ± 2.5%; PUF roller samples, 110 ± 3.7%; and handwash samples, 71.7 ± 1.2% for study 1 and 60.2 ± 3.0% for study 2. Data have not been adjusted for gauze pad or PUF roller samples because recoveries were essentially 100%. Handwash sample data were adjusted by the mean extraction efficiency values.
Samples were analyzed on a Varian 3700 gas chromatograph (Varian Associates, Walnut Creek, CA) equipped with an electron capture detector and a 6-ft  2 mm i.d. column packed with 1.95% SP 2401 on 80/100 Supelcoport (Supelco, Inc., Bellefonte, PA). The limit of detection for 1-µL injections was 3 pg/µL. The limit of detection for all sampling media (handwash solution, PUF, and gauze pad) was 0.8 ng/cm2 carpet surface area.
2 mm i.d. column packed with 1.95% SP 2401 on 80/100 Supelcoport (Supelco, Inc., Bellefonte, PA). The limit of detection for 1-µL injections was 3 pg/µL. The limit of detection for all sampling media (handwash solution, PUF, and gauze pad) was 0.8 ng/cm2 carpet surface area.
Laboratory blank and solvent blank samples were run with each set of samples and had no detectable chlorpyrifos. Field deposition and wipe samples were collected in triplicate in each room before application to determine if chlorpyrifos residues were present at the site. No background chlorpyrifos levels were found in these samples. Fortification/recovery studies were conducted for all sampling media. All the quality control samples were treated in a manner identical to field samples in regard to handling, storage, extraction, and analysis.
Most measurements from handwash samples in study 1 were near the limit of analytic detection; therefore, carpet contact area was increased 5-fold for study 2. Small sample sizes and the skewed distribution of some of the data sets led to the use of nonparametric statistical tests, including Mann-Whitney U and Kruskal-Wallis one-way analysis of variance (ANOVA) tests, for data analysis.
Residue levels deposited and removed from carpet in study 1 are presented in Table 2, and include data from both broadcast and total aerosol release applications. A comparison of the various sampling technique results is provided in Figure 2.

Figure 2. Comparison of PUF roller, wipe, hand press, and hand drag sampling techniques for measuring dermal transfer of chlorpyrifos following broadcast and aerosol applications (mean ± standard deviation).
PUF, polyurethane foam.
Broadcast application. Average chlorpyrifos deposition on the broadcast room carpet was 12.3 µg/cm2, close to the theoretical deposition value of 12.9 µg/cm2 calculated on the basis of label recommendations. However, deposition variability was high [coefficient of variation (CV) 92%].
Wipe sampling 30 min after application removed an average of 343 ng/cm2, or 2.8% of the initial deposit. Wipe sampling 3 hr later removed approximately one-third as much chlorpyrifos (115 ng/cm2), suggesting a continued drying of the carpet. PUF roller samples collected 3.5 hr after application averaged 182 ng/cm2, or 1.5% of the initial deposit. No significant difference was observed between the 3.5-hr wipe and PUF roller samples. Results from both techniques were highly variable, but not substantially different from the variability seen in deposition.
Hand press and hand drag results were similar for the broadcast application (5.9 and 4.5 ng/cm2, respectively) and significantly lower than either the wipe or PUF roller measurements (Kruskal-Wallis ANOVA, p = 0.0007). The combined hand measurements indicated that 5.2 ng/cm2 was removed from the carpet by skin. Transfer to skin was approximately 23 times lower than the wipe sampling estimate and approximately 36 times lower than the PUF roller estimate.
Aerosol application. Study 1. Average carpet deposition after the total release aerosol application was 2.64 µg/cm2, significantly lower than the broadcast application (Mann-Whitney U test, p = 0.0009), but consistent with deposition values produced in a previous study with similar aerosol canisters (3). Deposition variability was lower for this type of application (CV 50%) than for the broadcast application.
Average wipe sampling values 30 min and 3.5 hr after application were not significantly different from each other, nor from the average PUF roller value (70, 57, and 80 ng/cm2, respectively). Wipe sampling and the PUF roller removed 2-3% of the initial deposit. Variability of these techniques was similar and substantially higher than the deposition variability.
Hand press and hand drag results were similar (3.4 and 1.4 ng/cm2, respectively), and were highly variable. The combined hand measurements were significantly lower than either the wipe or PUF roller measurements (Kruskal-Wallis ANOVA, p = 0.0006), and indicated that approximately 2.4 ng/cm2 was removed from the carpet by skin. Transfer to skin was approximately 24 times lower than the wipe sampling estimate, and approximately 33 times lower than the PUF roller estimate.
Study 2. Study 2 data are presented in Table 3 and illustrated in Figure 3. Average chlorpyrifos deposition was similar on carpet and furniture surfaces (1.97 and 1.79 µg/cm2, respectively). These values were not significantly different from each other or from aerosol deposition in study 1. Deposition was uniform for both surfaces (CVs 8-10%). The average hand press value for carpet was 5.1 ng/cm2, approximately 11 times lower than the average wipe sample value from study 1 (Mann-Whitney U test, p = 0.0004; no carpet wipes were collected in study 2). Wipe sampling removed an average of 56 ng/cm2 from furniture surfaces, whereas hand press sampling recovered an average of 12.4 ng/cm2. The 4.5-fold difference between wipe and hand press samples was statistically significant (Mann-Whitney U test, p = 0.002). The hand press values for carpet and furniture were significantly different (Mann-Whitney U test, p = 0.009), even though the initial depositions of chlorpyrifos on these two different surfaces were similar. These findings indicate that chlorpyrifos was 2.4 times more available for transfer to skin from furniture surfaces such as desktops than from carpets.

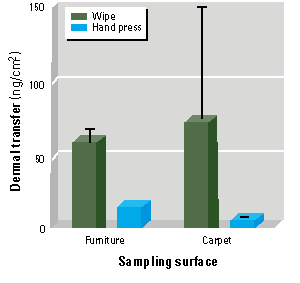
Figure 3. Comparison of wipe and hand press sampling techniques of dermal transfer of chlorpyrifos from two different sampling surfaces in study 2. Wipe sample data from carpet surface were obtained from study 1 (mean ± standard deviation).
The percentages of chlorpyrifos transferred to human skin relative to initial deposition, wipe sampling, and PUF roller sampling are summarized in Table 4. Skin removed between 0.04 and 0.69% of the chlorpyrifos deposited on surfaces by either broadcast or aerosol applications. Hand contact removed 3-9% of the amount measured by wipe sampling from carpet and 22% from furniture. Hand contact removed 3-6% of the amount measured by PUF roller sampling.

The central finding of these studies is that current methods for measuring transferable residues on carpets and furniture surfaces after commercial pesticide applications substantially overestimated the amount of residue removed by skin contact. This finding held true across application types and for different skin loadings. The magnitude of the observed differences was striking: more than 20-fold for wipe sampling and more than 30-fold for PUF roller sampling in the first study, and more than 10-fold in the second study. This effect was less evident for furniture surfaces, but still significant. Although this paper focuses on quantifying residue transfer from surface to skin using different surface sampling techniques, no attempt is made to quantify the amount of chlorpyrifos residues that is then subsequently absorbed.
A major difference between this study and previous studies investigating dermal transfer was the use of quantitative removal efficiency factors to adjust for incomplete handwashing. These removal efficiency factors increased hand exposure estimates 2-4 times. Had the adjustment factors not been used, the discrepancy between surface sampling methods and skin contact values would have been even greater.
The only other studies that have compared surface transfer measurement techniques to actual skin transfer have involved the PUF roller (6,7). The first of these studies (6) was a preliminary report of a pilot laboratory trial involving three hand contacts and three PUF roller contacts with aluminum foil that had been treated with known amounts of several pesticides. The authors reported no difference between the two sampling methods, although inspection of the original data indicates that PUF roller values were always greater than hand contact values. In later studies of carpeted rooms that had not been treated recently (7), hand press sampling was conducted in parallel with PUF roller sampling. Transferable residues were approximately twice as high for the PUF roller when compared to the hand press. Neither of these earlier studies measured pesticide contact and transfer after residential pest control applications; therefore, it is difficult to compare the findings directly with those reported here.
The results of the present study are consistent with those of a recent study of similar applications (3). In that study, deposition after broadcast application was approximately 7 times greater than total aerosol release application. The present study found a 4- to 7-fold difference. Wipe sampling in the previous study 1 hr after application demonstrated a relatively higher transferable residue for aerosol applications as compared to broadcast applications (0.8 vs 4% of initial deposit); the values for the present study at 3.5 hr after application were 0.9 and 2.2% for broadcast and aerosol, respectively.
Another recent study of chlorpyrifos broadcast applications included wipe sampling on untreated surfaces but did not include skin contact measurements (11). The study extrapolated environmental concentrations to child doses through a series of worst-case assumptions. Among these were that skin contact with furniture removed 75% of surface residues, and that skin contact with toys removed 100% of total residues in or on the toys. The results of our study present a contrasting picture, as indicated in Table 4. Skin contact was able to remove < 1% of the chlorpyrifos deposited on carpets and furniture and from 4 to 22% of the amount removed from these surfaces by wipe sampling.
The removal of pesticide residues from treated surfaces by human skin contact is a complex phenomenon, requiring understanding of the chemical nature of the compound under study, as well as application and formulation characteristics. A thorough analysis must also incorporate the physiology and condition of the skin and human behavior patterns that produce variable pressure and motion applied to surfaces. Thus, substantial uncertainties remain in current estimates of dermal exposure. Although many efforts have been devoted to developing and comparing new techniques to quantify dermal transfer from contaminated surfaces, most of the techniques have not been validated with human studies. Further studies of pesticide transfer to skin in residential settings are merited to reduce uncertainties in this important component of aggregate pesticide exposure.

