The Centers for Disease Control and Prevention (CDC) recommends that the calcium disodium edetate (CaNa
2EDTA) challenge test be considered for children who have blood lead levels of 1.21-2.12 µmol/L (25-44 µg/dL) to determine whether chelation is indicated (
1). This recommendation is based on a past study (
2,3). However, the challenge test is a potentially difficult procedure because of the required 8-hr urine collection, often on an active toddler. By identifying children who are likely to have a negative challenge test, a medical provider can minimize the number of children undergoing this test. The objective of this study was to identify factors likely to predict the outcome of challenge tests.
The Finger Lakes Lead Poisoning Prevention and Treatment Center at Rochester General Hospital (Rochester, NY) offers medical services to children who have elevated blood lead levels in a nine-county area surrounding Rochester, New York. Children can be referred to this center by their physicians for a challenge test with CaNa2EDTA if their blood lead levels are 1.21-2.12 µmol/L (25-44 µg/dL). Parents of all children admitted to Rochester General Hospital sign a consent form on admission; the form allows staff physicians to review the chart and abstract data for research purposes.
We obtained a venous lead level, erythrocyte protoporphyrin (EP) level, serum iron, total iron binding capacity (TIBC), and hematocrit immediately before administering CaNa2EDTA. We administered CaNa2EDTA at a dose of 500 mg/m2 either intravenously over 1 hr or intramuscularly mixed 3:1 by volume with 2% lidocaine; the result of a challenge test is unaffected by whether the CaNa2EDTA is administered intramuscularly or intravenously (4). Subsequently, an 8-hr urine sample was collected and stored in an acid-washed, lead-free plastic container. Maintenance oral fluid intake was encouraged throughout the urine collection; if oral intake fell below maintenance in any 2-hr period during the 8-hr collection, we administered intravenous fluids at a maintenance rate.
Blood and urine lead levels were measured by graphite furnace atomic absorption spectrometry (5). Blood samples were assayed twice, and the mean was reported. The laboratory participates in a proficiency testing program for the blood lead assay. There is no proficiency testing program for the determination of lead in urine; however, quality control is conducted on urine samples acquired from the New York State Department of Health (Albany, NY). During 1997 and 1998, 62 quality control samples were assayed, and the values of all were within two standard deviations of the mean (6). The EP level was assayed fluorimetrically (7). Serum iron was measured using Ferrozine (Hach Chemical Co., Ames, IA) to form a colored complex with reduced iron (8). TIBC was measured using Ferrozine (9).
The results of a challenge test were eligible for analysis if there were no losses of urine during the 8-hr urine collection. We compared the mean age and blood lead level at the time of referral among the excluded challenge tests and the eligible challenge tests to assess whether the two groups were comparable.
The proportions of positive challenge tests were determined for different strata of blood lead levels for all eligible children having challenge tests and for those whose EP level was  0.62 µmol/L (
0.62 µmol/L ( 35 µg/dL). To identify variables predictive of a positive challenge test [i.e., urine lead:CaNa2EDTA ratio
35 µg/dL). To identify variables predictive of a positive challenge test [i.e., urine lead:CaNa2EDTA ratio  0.60 (1)] that we could introduce into multivariate analysis, we calculated Pearson correlation coefficients between the urine lead:CaNa2EDTA ratio and blood lead level at the time of referral, blood lead level on the day of the challenge test, age, hematocrit, iron saturation (i.e., serum iron/TIBC), EP level, and RATE (i.e., the blood lead level on the day of the challenge test minus the blood lead level at the time of referral, divided by the number of days between the two blood lead level measurements. RATE is a measure of the rate of change of the blood lead level; a positive RATE indicates the blood lead level rose between the time of referral and the challenge test. We defined significant comparisons (i.e., Pearson correlation coefficient with a p-value
0.60 (1)] that we could introduce into multivariate analysis, we calculated Pearson correlation coefficients between the urine lead:CaNa2EDTA ratio and blood lead level at the time of referral, blood lead level on the day of the challenge test, age, hematocrit, iron saturation (i.e., serum iron/TIBC), EP level, and RATE (i.e., the blood lead level on the day of the challenge test minus the blood lead level at the time of referral, divided by the number of days between the two blood lead level measurements. RATE is a measure of the rate of change of the blood lead level; a positive RATE indicates the blood lead level rose between the time of referral and the challenge test. We defined significant comparisons (i.e., Pearson correlation coefficient with a p-value 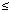 0.10) as explanatory variables, and used these in multivariate analysis. We also conducted bivariate analysis on these explanatory variables to assess whether each individually would predict the outcome of a challenge test (i.e., positive versus negative); sensitivity, specificity, positive predictive value, and negative predictive value were calculated.
0.10) as explanatory variables, and used these in multivariate analysis. We also conducted bivariate analysis on these explanatory variables to assess whether each individually would predict the outcome of a challenge test (i.e., positive versus negative); sensitivity, specificity, positive predictive value, and negative predictive value were calculated.
 We conducted multivariate analysis using logistic regression. We devised two models of a child diagnosed with a moderately elevated blood lead level [1.21-2.12 µmol/L (25-44 µg/dL)]. In the first model a medical provider determines the likelihood of a positive challenge test conducted within 2 weeks of the diagnosis; in the second model, a medical provider obtains a second blood lead level and EP level 2-4 weeks after the diagnosis and then determines the likelihood of a positive challenge test. In the first model we used referral blood lead level and age as independent variables among the challenge tests conducted within 2 weeks of the referral; in the second model we used blood lead level at the time of the challenge test, age, EP level, and RATE as independent variables among the challenge tests conducted 2-4 weeks after the referral. The aforementioned independent variables were all significantly correlated to the urine lead:CaNa2EDTA ratio (Table 1). Both blood lead level measures were coded into 0.24 µmol/L (5 µg/dL) strata [i.e., 1.21-1.40 µmol/L (25-29 µg/dL), 1.44-1.64 µmol/L (30-34 µg/dL), etc.]. Age was dichotomized into < 36 months and
We conducted multivariate analysis using logistic regression. We devised two models of a child diagnosed with a moderately elevated blood lead level [1.21-2.12 µmol/L (25-44 µg/dL)]. In the first model a medical provider determines the likelihood of a positive challenge test conducted within 2 weeks of the diagnosis; in the second model, a medical provider obtains a second blood lead level and EP level 2-4 weeks after the diagnosis and then determines the likelihood of a positive challenge test. In the first model we used referral blood lead level and age as independent variables among the challenge tests conducted within 2 weeks of the referral; in the second model we used blood lead level at the time of the challenge test, age, EP level, and RATE as independent variables among the challenge tests conducted 2-4 weeks after the referral. The aforementioned independent variables were all significantly correlated to the urine lead:CaNa2EDTA ratio (Table 1). Both blood lead level measures were coded into 0.24 µmol/L (5 µg/dL) strata [i.e., 1.21-1.40 µmol/L (25-29 µg/dL), 1.44-1.64 µmol/L (30-34 µg/dL), etc.]. Age was dichotomized into < 36 months and  36 months strata. EP level was dichotomized into < 0.62 µmol/L (< 35 µg/dL) and
36 months strata. EP level was dichotomized into < 0.62 µmol/L (< 35 µg/dL) and  0.62 µg/dL (
0.62 µg/dL ( 35 µg/dL) strata. RATE was dichotomized into < 0 µg/dL/day and
35 µg/dL) strata. RATE was dichotomized into < 0 µg/dL/day and  0 µg/dL/day strata. We introduced all combinations of the respective variables for each of the two models into forced-entry logistic regression using SPSS software (SPSS, Inc., version 4.0, Chicago, IL). Our objective was to find the model and set of independent variables that would correctly classify all true-positive challenge tests (i.e., 100% sensitivity) while minimizing the number of true-negative challenge tests that were falsely classified.
0 µg/dL/day strata. We introduced all combinations of the respective variables for each of the two models into forced-entry logistic regression using SPSS software (SPSS, Inc., version 4.0, Chicago, IL). Our objective was to find the model and set of independent variables that would correctly classify all true-positive challenge tests (i.e., 100% sensitivity) while minimizing the number of true-negative challenge tests that were falsely classified.
A total of 217 children were referred between January 1992 and September 1998 for 253 challenge tests (mean 1.2 challenge tests per child; range 1-5). Among these children, 51% were male; 68% were African American, 14% were Hispanic, 16% were white, and 2% were an undefined race. The mean age at the time of the challenge test was 37.7 months, and the mean blood lead level at the time of referral was 1.83 µmol/L (38 µg/dL); all blood lead levels were assayed on venous blood.
A total of 75 challenge tests had urine losses during the 8-hr collection period, leaving 178 that were eligible for analysis. These eligible challenge tests were made up of children with a mean age of 38.2 months, and a mean blood lead level at the time of referral of 1.83 µmol/L (38 µg/dL). The mean values of these two variables were not significantly different from the respective mean values of the sample of excluded challenge tests. There was a mean of 16 days (range 3-78 days) between the blood lead level at the time of referral and the challenge test; 89 (50%) challenge tests were conducted within 14 days of the referral blood lead level, and 73 (41%) were conducted 15-31 days after the referral blood lead level. Although nearly all children had a blood lead level between 1.21 and 2.12 µmol/L (25-44 µg/dL) at the time of referral [two children who each had a referral blood lead level of 2.27 µmol/L (47 µg/dL) were challenged at the request of a parent], by the day of the challenge test some children had a blood lead level outside this range. The mean value of blood and urine tests among the eligible challenge tests are reported in Table 2.

Among the eligible 178 challenge tests, 40 (22%) were positive and 138 were negative. Table 3 lists the proportion of challenge tests that were positive at different strata of blood lead levels among the eligible challenge tests. There were no positive challenge tests among children with a blood lead level 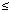 1.40 µmol/L (
1.40 µmol/L (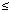 29 µg/dL), whereas 71% of the challenge tests were positive among children with a blood lead level 2.41-2.61 µmol/L (50-54 µg/dL). Restricting the sample to children with EP levels
29 µg/dL), whereas 71% of the challenge tests were positive among children with a blood lead level 2.41-2.61 µmol/L (50-54 µg/dL). Restricting the sample to children with EP levels  0.62 µmol/L (
0.62 µmol/L ( 35 µg/dL) resulted in higher proportions of positive challenge tests for nearly every stratum of blood lead level (Table 2).
35 µg/dL) resulted in higher proportions of positive challenge tests for nearly every stratum of blood lead level (Table 2).

Table 1 lists the Pearson correlation coefficients between several factors and the urine lead:CaNa2EDTA ratio. Blood lead level at the time of referral, blood lead level on the day of the challenge test, age, EP level, and RATE were significantly correlated to the urine lead:CaNa2EDTA ratio (i.e., p 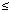 0.10), and were defined as explanatory variables. Table 4 lists the sensitivity, specificity, positive predictive value, and negative predictive value among the latter explanatory variables. Although the correlation coefficient did not define iron saturation as an explanatory variable, we nevertheless conducted bivariate analysis on it because a past study found that it predicted the outcome of a challenge test (10).
0.10), and were defined as explanatory variables. Table 4 lists the sensitivity, specificity, positive predictive value, and negative predictive value among the latter explanatory variables. Although the correlation coefficient did not define iron saturation as an explanatory variable, we nevertheless conducted bivariate analysis on it because a past study found that it predicted the outcome of a challenge test (10).

In bivariate analysis, we found age to only be a fair predictor of the outcome of a challenge test. Among challenge tests conducted at age < 36 months, 15% were positive, while among challenge tests conducted at age  36 months, 31% were positive. However, age at this cutoff lacks both sensitivity and specificity in predicting the outcome of a challenge test (Table 4).
36 months, 31% were positive. However, age at this cutoff lacks both sensitivity and specificity in predicting the outcome of a challenge test (Table 4).
Among the 178 challenge tests, 150 were accompanied by an EP level; this subsample of challenge tests is comparable to the eligible challenge tests with regard to age and all variables listed in Table 2. A low EP level is an excellent predictor of a negative challenge test. Three percent of challenge tests with an EP level < 0.62 µmol/L (< 35 µg/dL) were positive, and 31% of challenge tests with an EP level  0.62 µmol/L (
0.62 µmol/L ( 35 µg/dL) were positive. Blood lead level was comparable between these two strata of EP level. The mean blood lead level on the day of chelation among children with EP levels < 0.62 µmol/L (< 35 µg/dL) and EP level
35 µg/dL) were positive. Blood lead level was comparable between these two strata of EP level. The mean blood lead level on the day of chelation among children with EP levels < 0.62 µmol/L (< 35 µg/dL) and EP level  0.62 µmol/L (
0.62 µmol/L ( 35 µg/dL) was 1.49 µmol/L [31 µg/dL; range 0.72-2.32 µmol/L (15-48 µg/dL)] and 1.74 µmol/L [36 µg/dL; range 0.58-2.61 µmol/L (12-54 µg/dL)], respectively. The EP level is very sensitive; 97% of positive challenge tests had an EP level
35 µg/dL) was 1.49 µmol/L [31 µg/dL; range 0.72-2.32 µmol/L (15-48 µg/dL)] and 1.74 µmol/L [36 µg/dL; range 0.58-2.61 µmol/L (12-54 µg/dL)], respectively. The EP level is very sensitive; 97% of positive challenge tests had an EP level  0.62 µmol/L (
0.62 µmol/L ( 35 µg/dL). However, this test lacks specificity because only 25% of negative challenge tests had an EP level < 0.62 µmol/L (< 35 µg/dL).
35 µg/dL). However, this test lacks specificity because only 25% of negative challenge tests had an EP level < 0.62 µmol/L (< 35 µg/dL).
A decreasing RATE (i.e., < 0 µg/dL/day) was a fair predictor of a negative challenge test. Eighty-six percent of challenge tests with a decreasing RATE were negative. This proportion did not markedly change when RATE was stratified by the number of days between the dates of the referral blood lead level and the challenge test. Among challenge tests done within 14 days of the referral blood lead level, 81% with a decreasing RATE were negative. Among challenge tests done within 15-31 days of the referral blood lead level, 88% with a decreasing RATE were negative. However, RATE at this cutoff lacks both sensitivity and specificity in predicting the outcome of a challenge test (Table 4).
 Of the two models for which multivariate logistic regression was performed, the second model (a medical provider who obtains a repeat blood lead level 2-4 weeks after diagnosing a child with a moderately elevated blood lead level) was the preferential scenario for predicting the outcome of a challenge test when blood lead level at the time of chelation, age, and RATE were entered into the analysis. Table 5 lists the predicted probability of a challenge test according to this logistic regression model by blood lead level, age, and RATE. A predicted probability of 0.15 was the threshold above which all true-positive challenge tests were identified (100% sensitivity), but 39% of true-negative challenge tests were falsely classified as positive.
Of the two models for which multivariate logistic regression was performed, the second model (a medical provider who obtains a repeat blood lead level 2-4 weeks after diagnosing a child with a moderately elevated blood lead level) was the preferential scenario for predicting the outcome of a challenge test when blood lead level at the time of chelation, age, and RATE were entered into the analysis. Table 5 lists the predicted probability of a challenge test according to this logistic regression model by blood lead level, age, and RATE. A predicted probability of 0.15 was the threshold above which all true-positive challenge tests were identified (100% sensitivity), but 39% of true-negative challenge tests were falsely classified as positive.
The CDC recommends that the CaNa2EDTA challenge test be considered for children who have blood lead levels of 1.21-2.12 µmol/L (25-44 µg/dL) to determine whether chelation is indicated (1). However, some authors no longer recommend the challenge test because of the potential toxicity of CaNa2EDTA (11,12), and the difficulty of obtaining complete urine samples (12,13).
The assertion that CaNa2EDTA is toxic arises from a study reporting that rats receiving a single dose of CaNa2EDTA had an increase in brain lead level (14). In that study, the dose of CaNa2EDTA associated with increased brain lead levels was 150 mg/kg, but this phenomenon did not occur at a dose of 75 mg/kg. Because an even lower dose of CaNa2EDTA, approximately 25 mg/kg, is recommended for the CaNa2EDTA challenge test (1), the results of that study are not generalizable to children who are candidates for the challenge test.
The assertion that it is difficult to obtain complete urine samples is accurate. However, this argues not for the abandonment of this test, but rather that it be conducted by personnel experienced in collecting urine from young children. In 1997, our center obtained complete collections on 83% of children; the remaining children sustained inconsequential urine losses during the last 1-2 hr of the collection, a period when CaNa2EDTA-induced lead excretion is minimal.
At present, the blood lead level has become the sole factor determining whether a child undergoes chelation (1). Nevertheless, children with comparable blood lead levels can have great differences in body lead burden and thus different degrees of lead diuresis in response to a chelating agent. For example, only 3% of children with an EP level < 0.62 µmol/L (< 35 µg/dL) had a positive challenge test, in comparison to 31% of children who had an EP level  0.62 µmol/L (
0.62 µmol/L ( 35 µg/dL) despite comparable blood lead levels. This reflects the heterogeneity that exists within the population of lead-exposed children and highlights the limitations of the blood lead level in determining whom to chelate. Authors have raised this issue and argued that the challenge test is useful for identifying whom to treat when blood lead levels are moderately elevated (15,16). Our study supports this contention. Among children who have comparable blood lead levels, the challenge test differentiates children who will not have a significant CaNa2EDTA-induced lead diuresis, and thus will not benefit from chelation, from those who will have a significant CaNa2EDTA-induced lead diuresis, and thus are more likely to benefit from chelation. Nevertheless, further research is needed to define what constitutes a significant level of chelation-induced lead diuresis (13), preferably by linking it to neurocognitive outcomes.
35 µg/dL) despite comparable blood lead levels. This reflects the heterogeneity that exists within the population of lead-exposed children and highlights the limitations of the blood lead level in determining whom to chelate. Authors have raised this issue and argued that the challenge test is useful for identifying whom to treat when blood lead levels are moderately elevated (15,16). Our study supports this contention. Among children who have comparable blood lead levels, the challenge test differentiates children who will not have a significant CaNa2EDTA-induced lead diuresis, and thus will not benefit from chelation, from those who will have a significant CaNa2EDTA-induced lead diuresis, and thus are more likely to benefit from chelation. Nevertheless, further research is needed to define what constitutes a significant level of chelation-induced lead diuresis (13), preferably by linking it to neurocognitive outcomes.
Our study found that readily obtainable tests can identify children likely to have a negative challenge test. A low EP level is excellent at identifying such children; only 3% of challenge tests with an EP level < 0.62 µmol/L (< 35 µg/dL) were positive. This finding is comparable to the 4.8% reported by Markowitz et al. (15). A decreasing blood lead level is a fair predictor of a negative challenge test; 86% of children whose blood lead level was decreasing (i.e., RATE < 0 µg/dL/day) had a negative challenge test. Additionally, certain blood lead levels were also predictive of a negative challenge test; no challenge tests were positive at blood lead levels 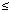 1.40 µmol/L (
1.40 µmol/L (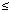 29 µg/dL). Previous studies report that iron deficiency is associated with a lowered likelihood of a positive challenge test (10,15). Our study found that iron saturation did not reliably distinguish the outcome of a challenge test; this is not surprising because iron status was previously noted to be weakly associated with the outcome of the challenge test (10). However, regarding bivariate analysis, all the aforementioned variables misclassify a large proportion of the true-positive or true-negative challenge tests.
29 µg/dL). Previous studies report that iron deficiency is associated with a lowered likelihood of a positive challenge test (10,15). Our study found that iron saturation did not reliably distinguish the outcome of a challenge test; this is not surprising because iron status was previously noted to be weakly associated with the outcome of the challenge test (10). However, regarding bivariate analysis, all the aforementioned variables misclassify a large proportion of the true-positive or true-negative challenge tests.
In logistic regression, we found that the second model--a medical provider who obtains a repeat blood lead level 2-4 weeks after diagnosing a child with a moderately elevated blood lead level [1.21-2.12 µmol/L (25-44 µg/dL)]--resulted in the best prediction when using blood lead level at the time of chelation, age, and RATE. This finding replicates, in part, the finding of a previous study that reported that blood lead level, age, and EP level are predictive of the outcome of a challenge test (15). We recommend that a provider obtain a second blood lead level 2-4 weeks after diagnosing a child with a moderately elevated blood lead level. The provider can subsequently use Table 5 to determine the predicted probability of a positive challenge test for their patient and determine whether to order such a test. According to our model, predicted probabilities  15% would correctly classify 100% of true-positive challenge tests while misclassifying only 39% of true-negative challenge tests. Alternatively, providers who do not have access to the challenge test may use Table 5 in deciding whether to chelate empirically.
15% would correctly classify 100% of true-positive challenge tests while misclassifying only 39% of true-negative challenge tests. Alternatively, providers who do not have access to the challenge test may use Table 5 in deciding whether to chelate empirically.
Providers should consider whether the aforementioned findings apply to their patients. The model may not be applicable in instances where a repeat blood lead level is obtained beyond 4 weeks of the diagnosis. Additionally, because proficiency testing programs for the assay of lead in urine do not currently exist, providers should conduct the challenge test as we described in "Methods" and be assured that their population has comparable rates of positive challenge tests by strata of blood lead level. Other studies have reported rates of positive challenge tests comparable to those in our study (15,17,18).
In summary, we recommend the challenge test for children  36 months of age who have a blood lead level between 1.45 and 1.64 µmol/L (30-34 µg/dL) and for children who have a blood lead level 1.69-2.12 µmol/L (35-44 µg/dL) regardless of age.
36 months of age who have a blood lead level between 1.45 and 1.64 µmol/L (30-34 µg/dL) and for children who have a blood lead level 1.69-2.12 µmol/L (35-44 µg/dL) regardless of age.
Not all children with a blood lead level of 2.17-2.61 µmol/L (45-54 µg/dL) had a positive challenge test. This suggests that the challenge test, rather than chelation, be considered for children whose blood lead levels are in that range. Likewise, the data also suggest that the blood lead level threshold above which chelation be conducted without a challenge test be defined higher than 2.17 µmol/L (45 µg/dL), as currently defined by the CDC (1). However, we do not currently advocate changing the threshold at which to chelate. Rather, the assay of lead in urine should be standardized by proficiency testing programs; subsequently, if our findings are replicated at other centers with a sufficiently large sample size, the change in these thresholds should be considered.

