
| |  | |  |
Research
|
| Bone Mineral Density Changes in Relation to Environmental PCB Exposure Susan Hodgson,1,2 Laura Thomas,2 Elena Fattore,3 P. Monica Lind,4 Tobias Alfven,4 Lennart Hellström,5,6 Helen Håkansson,4 Grazia Carubelli,3 Roberto Fanelli,3 and Lars Jarup2 1Institute of Health and Society, Newcastle University, Newcastle upon Tyne, United Kingdom; 2Department of Epidemiology and Public Health, Imperial College London, United Kingdom; 3Department of Environmental Health Sciences, "Mario Negri" Institute for Pharmacological Research, Milano, Italy; 4Institute of Environmental Medicine, Karolinska Institutet, Stockholm, Sweden; 5Department of Environmental Medicine and Public Health, County Council of Kalmar, Oskarshamn, Sweden; 6Division of Occupational and Environmental Medicine, Department of Molecular and Clinical Medicine, Faculty of Health Sciences, Linköping University, Linköping, Sweden Abstract
Background: Bone toxicity has been linked to organochlorine exposure following a few notable poisoning incidents, but epidemiologic studies in populations with environmental organochlorine exposure have yielded inconsistent results. Objectives: The aim of this study was to investigate whether organochlorine exposure was associated with bone mineral density (BMD) in a population 60–81 years of age (154 males, 167 females) living near the Baltic coast, close to a river contaminated by polychlorinated biphenyls (PCBs) . Methods: We measured forearm BMD in participants using dual energy X-ray absorptiometry ; and we assessed low BMD using age- and sex-standardized Z-scores. We analyzed blood samples for five dioxin-like PCBs, the three most abundant non-dioxin-like PCBs, and p,p´-dichlorophenyldichloroethylene (p,p´-DDE) . Results: In males, dioxin-like chlorobiphenyl (CB) -118 was negatively associated with BMD ; the odds ratio for low BMD (Z-score less than –1) was 1.06 (95% confidence interval, 1.01–1.12) per 10 pg/mL CB-118. The sum of the three most abundant non-dioxin-like PCBs was positively associated with BMD, but not with a decreased risk of low BMD. In females, CB-118 was positively associated with BMD, but this congener did not influence the risk of low BMD in women. Conclusions: Environmental organochlorine exposures experienced by this population sample since the 1930s in Sweden may have been sufficient to result in sex-specific changes in BMD. Key words: bone mineral density, p, p´-DDE, polychlorinated biphenyls, toxic equivalents. Environ Health Perspect 116:1162–1166 (2008) . doi:10.1289/ehp.11107 available via http://dx.doi.org/ [Online 16 May 2008]
Address correspondence to S. Hodgson, Institute of Health and Society, 4th Floor, William Leech Building, The Medical School, Newcastle University, Newcastle upon Tyne NE2 4HH, UK. Telephone: 44 (0) 191 222 3823. Fax: 44 (0) 191 222 8211. E-mail: susan.hodgson@ncl.ac.uk Supplemental Material is available online at http://www.ehponline.org/members/2008/11107/suppl.pdf We thank the European Commission (EU-QLK-CT-2002-02528) and the Swedish Environmental Protection Agency for financial support. Ethical approval for this study was obtained from the Karolinska Institutet ethics committee. The authors declare they have no competing financial interests. Received 29 November 2007 ; accepted 12 May 2008. |
|
|
 |
It is well known that hormones, vitamins, pharmaceuticals, and metals can have adverse effects on bone. Bone effects, mainly congenital, have also been linked to persistent organochlorine exposure following a few notable poisoning incidents. High-level, accidental dietary exposure to hexachlorobenzene resulted in severe osteoporosis (Cripps et al. 1984; Gocmen et al. 1989), and infants exposed in utero to high concentrations of polychlorinated biphenyls (PCBs) and polychlorinated dibenzofurans developed irregular calcification of their skull bones (Miller 1985).
Recent epidemiologic studies in populations with environmental organochlorine exposure have yielded inconsistent results regarding whether organochlorine exposure has an effect on bone properties. Several single-sex studies have indicated a possible association between organochlorine exposure and bone quality. In 115 Swedish men from the general population, a significant association was seen between serum concentrations of the organochlorines chlorobiphenyl (CB)-28 and γ-hexachlorocyclohexane and broadband ultrasound attenuation [an indirect estimation of bone mineral density (BMD)] of the left os calcis, and between CB-167 and total-body BMD after adjustment for confounding variables (Glynn et al. 2000a). In 68 sedentary Australian women, a negative correlation between p,p´-dichlorodiphenyldichloroethylene (p,p´-DDE) levels and BMD was observed, suggesting that past exposure to DDT may be associated with reduced BMD in women (Beard et al. 2000). Other studies have not found organochlorine exposure to be associated with bone effects. In 103 peri- and postmenopausal women from the United States, no correlation was found between p,p´-DDE concentration and bone density or rate of bone loss at the spine or radius (Bohannon et al. 2000). In 153 peri- and postmenopausal Inuit women with high organochlorine exposure from their seafood diet, concentrations of CB-153 were inversely correlated with bone parameters in univariate analyses, but, again, this relationship was no longer evident after adjustment for potential confounding variables (Cote et al. 2006).
One study investigated bone effects in males and females from Sweden. In the initial study, the risk of hospitalization from fracture in fishermen and their wives was compared between those living on the east coast of Sweden (near the Baltic sea, where environmental organochlorine exposure is likely attributable to consumption of contaminated fatty fish) and those living on the west coast (Alveblom et al. 2003). There was a significantly increased incidence rate ratio for vertebral fractures among east coast fisherman's wives, with a similar (nonsignificant) tendency in east coast fishermen after adjustment for age and calendar year, compared with those dwelling on the west coast. It should be noted that this exposure assessment is rather crude and that other fracture types did not show a similar trend. Following from this study, the relationship between serum levels of CB-153 and p,p´-DDE and BMD in a subset of the Baltic coast fishermen (n = 196) and their wives (n = 184) was further studied (Wallin et al. 2005). Univariate analyses revealed significant negative associations between CB-153 and BMD in males and females, but after adjustment for age and body mass index (BMI), these relationships did not persist.
Although the epidemiologic evidence is inconsistent, an increasing number of experimental studies lend biological plausibility to organochlorine-induced bone effects (Andrews 1989; Badraoui et al. 2007). Animal studies also have demonstrated that the timing of exposure can be critical, with effects being observed at lower dose levels when exposure occurs during earlier life stages; furthermore, the same compound seems to have the potential to modulate bone quality differently depending on the developmental stage at exposure (Jamsa et al. 2001; Miettinen et al. 2005). The estrogen status of the exposed individual has also been shown to influence the toxicity of organochlorines on bone. Studies in ratsexposed to the dioxin-like CB-126 showed that bone strength and composition are impaired and that estrogen can modulate the induced effects depending on the estrogen status of the individual (Lind et al. 1999, 2000, 2004). CB-126 exposure did not affect bone mineral density or trabecular bone volume of tibia in sham-operated rats. In contrast, in estrogen-deprived ovariectomized rats, CB-126 exposure resulted in a decreased length and increased bone mineral density of tibia. Furthermore, estrogen supplementation modulated CB-126-induced effects on bone tissue in rats (Lind et al. 1999, 2000, 2004). Because of these various influences, the effect of exposure to a number of different organochlorines is likely to be difficult to predict; nonetheless, these animal studies suggest a possible causal relationship between organochlorine exposure and adverse bone effects. Such a relationship, if applicable to humans, could play a role in the observed increase in osteoporosis and osteoporotic fractures in the western world (Genant et al. 1999; Ismail et al. 2002).
The purpose of the present study was to investigate the relationship between organochlorine exposure and BMD in a subset of the Osteoporosis Cadmium as Risk Factor (OSCAR) cohort. The OSCAR cohort was established to investigate the effect of low-level cadmium exposure on bone and kidneys (Alfven et al. 2002, 2004). Because many of the examined individuals live close to the Baltic coast, these people might also have experienced elevated dietary PCB exposure from consuming contaminated fatty fish. In addition, there was potential for PCB exposure from a nearby contaminated river, which was polluted with PCB containing paper pulp from an upstream paper mill (Bremle et al. 1998).
Full details of the OSCAR study population can be found elsewhere (Alfven et al. 2002). In brief, subjects 16–80 years of age who had resided near a nickel-cadmium battery plant for at least 5 years between 1910 and 1992 were invited to participate in the OSCAR study; 904 (62%) agreed to participate. In addition, workers with previous or current occupational exposure were invited to participate, and 117 (48%) agreed to take part. In total, 1,021 individuals (60%) agreed to participate in the study. The OSCAR participants provided information on employment, residence, smoking, diet, medical history (especially regarding kidney diseases and diseases related to osteoporosis). Specially trained nurses collected urine and blood samples and measured height and weight. Forearm BMD was measured with an ambulant instrument (Osteometer DTX-200; Meditech A/S, Rødovre, Denmark), using dual energy X-ray absorptiometry, which is commonly used to evaluate BMD (Alfven et al. 2000; Glynn et al. 2000a; Wallin et al. 2005). The distal site in the nondominant forearm was measured with the patient in a supine position. This site includes both the radius and the ulna from the 8-mm point (the point where the radius and ulna are separated by 8 mm) and 24 mm proximally, and contains 10–20% trabecular bone (Schlenker and VonSeggen 1976). Internal variation was checked by daily calibration using a phantom, and the measurements from the ambulant instrument were validated against a stationary hospital-based instrument (Jarup et al. 1998).
In addition to investigating BMD as a continuous variable, we created a dichotomous variable representing presence or absence of low BMD for use in logistic regression analyses. Low BMD was assessed by computing an age- and sex-standardized Z-score. A common definition of low BMD is Z-score less than –1 (Kanis et al. 1997), which indicates 1 standard deviation below a sex- and age-standardized mean obtained from a reference population provided by the instrument supplier.
Blood samples from a subset of the OSCAR cohort consisting of participants ≥ 60 years of age (n = 325) were included in this study [insufficient sample remained and/or labeling was unclear in 23 out of 348 samples (6.6%), meaning that we could assess only 325 out of 348 eligible participants]. Samples were analyzed for five mono-ortho chlorine substituted congeners (CBs 105, 118, 156, 157, and 167), expressed in terms of toxic equivalency (TEQmono-ortho), and individual CB-118 levels, to assess the effect of the dioxin-like activity. The sum of three most abundant non-dioxin-like (or di-ortho chlorine substituted) CBs 138, 153, and 180 (σCAP3PCB),and individual CB-153 levels were also analyzed. We measured CB-153 because the concentration of this congener correlates well with total and dioxin-like PCB concentrations in plasma and serum (Glynn et al. 2000b). σCAP3PCB is considered to be an indicator of the content of total PCBs in human samples; this measure represented, on average, 61% of the human PCB body burden (European Food Safety Authority 2005). Finally, we also analyzed p,p´-DDE, the persistent metabolite of DDT (dichlorodiphenyltrichloroethane) because this organochlorine has been measured in other studies exploring the effects of organochlorines on BMD (Beard et al. 2000; Glynn et al. 2000a; Wallin et al. 2005). Ethical approval for this study was obtained from the Karolinska Institutet ethics committee, and all participants gave their written, informed consent before the OSCAR study.
The analytical method for the measurement of PCBs was initially developed in serum (Mariani et al. 2002; Turci et al. 2004), but has been extended to plasma and whole blood. Before the analysis of the OSCAR samples for this study, analyses of PCBs in whole blood, serum, and plasma derived from the same blood sample were performed, and results showed no major differences between these matrices in the distribution of the congeners, and the coefficients were close to 1 [see Supplemental Material, Figure 1 (online at http://www.ehponline.org/members/2008/11107/suppl.pdf)]. Analysis of whole blood, however, has been suggested to better reflect the PCB body burden (Janak et al. 1999). Briefly, after thawing, aliquots of 0.5 mL of blood samples were spiked with a mixture of the eight PCB congeners (13C12 labeled; Wellington Laboratories, Guelph, Ontario, Canada) and p,p´-DDE. Sulfuric acid was added to a final volume of 2.5 mL. After cleanup overnight on an Extrelut column (Merck, Darmstadt, Germany), samples were concentrated to a small volume and analyzed by high-resolution gas chromatography (CG)–high resolution mass spectrometry using a trace CG (Finnigan, Bremen, Germany) provided with a GC PAL autosampler (CTC-Analytics, Zwingen, Switzerland) coupled to a Thermofinnigan MAT95XP mass spectrometer. The mass spectrometer was operated in electron ionization mode, using selected ion monitoring at a mass resolution of 10,000. Quantification was done by the isotopic dilution method. We carried out blank analyses routinely for every 20 samples, and for the definition of the limit of detection (LOD), a signal-to-noise ratio of 3:1 was chosen. For the TEQmono-ortho calculation the latest toxic equivalency factors (TEFs) were used (Van den Berg et al. 2006) and when the CB concentration values were below the LOD, they were entered as 0 (lower bound method).
We evaluated associations between organochlorine levels and BMD in SPSS 12.0 for Windows (SPSS Inc., Chicago, IL, USA) using univariate and multivariate linear regression and logistic regression analyses. Given the probable hormonal action of organochlorines on bone, and given the very large difference in osteoporosis incidence in males and females after 60 years of age, it seemed most appropriate to assess the relationships between organochlorines and BMD measures in males and females separately. Information on potentially confounding lifestyle factors and food consumption habits collected for the OSCAR cohort were included in the linear regression model if they fulfilled the stepwise criteria (probability-of-F-to-enter LESSEQ 0.10, probability-of-F-to-remove ≥ 0.15) to aid the interpretation of the results. Because the OSCAR cohort was originally set up to explore the effect of cadmium on BMD, blood cadmium was included as a potential confounding variable in the linear and logistic regression analyses presented.
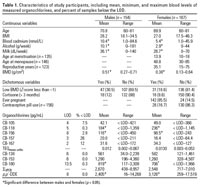
The blood concentration levels ranged from 0.002 to 0.067 pg TEQmono-ortho/mL for the dioxin-like PCBs, from 438 to 8,960 pg/mL for σCAP3PCB and from 16.4 to 17,500 pg/mL for p,p´-DDE (Table 1). Women had significantly higher levels of CB-118 and p,p´-DDE than did men.
Table 1.
|
Table 2.

|
Table 3.

|
In unadjusted analyses, significant negative correlations were seen between CB-118 and BMD (Pearson correlation coefficient = –0.130; p = 0.020) when males and females were investigated as one group. When males and females were investigated separately, none of the individual CB congeners was significantly correlated with BMD. There were significant positive correlations between each of the five organochlorine markers of interest. The dioxin-like organochlorines CBs 105, 118, 156, and 167 were all strongly correlated with TEQmono-ortho in males, females, and the whole group (Pearson correlation coefficient > 0.6); CB-157 showed far weaker, even nonsignificant correlations with the other organochlorines. CB-118 showed consistently strong correlations with the TEQmono-ortho measure (correlation coefficient > 0.9) and with the σCAP3PCB measure (correlation coefficient > 0.6). The abundant non-dioxin-like congeners were always strongly correlated with each other (correlation coefficient > 0.6), and CB-153 was always very strongly correlated with the σCAP3PCB measure (correlation coefficient > 0.9). In males, p,p´-DDE showed relatively strong correlations with TEQmono-ortho, CB-118, σCAP3PCB, and CB153 (correlation coefficient ~ 0.5), though these correlations were weaker in women (correlation ~ 0.4).
As anticipated, in bivariate analyses, age was negatively correlated with BMD, and BMI was positively correlated with BMD (data not shown). In males, alcohol was negatively correlated and milk consumption was positively correlated with BMD. In females, age at menopause and number of reproductive years (age at menopause minus age at menstruation) were both positively correlated with BMD; age at menstruation was negatively correlated with BMD. Contraceptive pill use was associated with an increased BMD, as was ever having been pregnant.
Table 2 shows the results from multivariate linear regression analysis in males and females. In males, multivariate linear regression analysis indicated that age, BMI, blood cadmium, and milk consumption explained 25% of the variability in BMD; none of the organochlorines was significantly associated with BMD when entered into the model individually. The organochlorine variables were then entered stepwise into the model, sequentially, in order of the smallest probability of F (if F LESSEQ 0.10), until no more variables were eligible for inclusion or removal (F ≥ 0.15). In this model CB-118 was negatively associated (B = –0.00024; p = 0.002) and σCAP3PCB positively associated (B = 0.00002; p = 0.003) with BMD in males, explaining an additional 6% of the variability in BMD. In females, multivariate linear regression analysis indicated that age, BMI, blood cadmium, age at menstruation, and ever having being pregnant explained 39.5% of the variability in BMD. When organochlorines were entered individually into this model, CB-118 was significantly positively associated with BMD (B = 0.00008; p = 0.045), explaining a further 2% of the variability in BMD. No additional organochlorines contributed significantly to the model after stepwise entry. Variables representing interactions between the organochlorines and cadmium did not explain any further variability in BMD.
A dichotomous variable representing presence or absence of low BMD (defined as Z-score less than –1) was used in logistic regression analyses; 47 of 154 (30.5%) of the males, and 31 of 167 (18.6%) of the females had low BMD. Those organochlorines that were found to significantly explain variability in BMD after stepwise entry into the multivariate linear regression models (CB-118 and σCAP3PCB in males and CB-118 in females) were included in the multivariate logistic regression as categorical variables (tertiles). Where a linear dose–response relationship seemed apparent by tertiles, these variables were reentered into the model as continuous variables to maximize power in assessing the dose–response relationship. Table 3 shows the results from logistic regression analysis in males and females. In males, the odds ratio (OR) for low BMD decreased with increasing BMI and milk consumption and increased with increasing blood cadmium levels. The risk of low BMD increased by tertile of CB-118, and when analyzed as a continuous variable, the OR was 1.06 (95% confidence interval 1.01–1.12) for every 10 pg/mL CB-118. The risk of low BMD did not show a clear trend across tertiles of σCAP3PCB. In women, the ORs for low BMD decreased with increasing BMI and increased with increasing age at menstruation; however, the risk of low BMD did not show a consistent trend with tertiles of CB-118.
The present study is one of the largest to date to assess the relationship between organochlorine levels in blood and BMD in an environmentally exposed population. The findings of this study suggest that even at relatively low levels of organochlorine exposure, BMD might be affected, at least in males, after controlling for major confounding variables. Despite the large variability of the individual values, spanning over three orders of magnitude, the blood concentration levels of these pollutants in this population are in the range of the values detected in human serum and/or blood samples for the general population (Botella et al. 2004; Turci et al. 2004; Wilhelm et al. 2003). Nonetheless, there was a significant positive dose–response relationship in men between the dioxin-like congener CB-118 and BMD (OR = 1.06 per 10 pg/mL; 95% confidence interval 1.01–1.12). In females, after finding a small but significant positive association between CB-118 and BMD in the linear regression analysis, we anticipated a decrease in risk of low BMD with increasing levels of CB-118; however, the risk of low BMD did not show a consistent trend with tertile of CB-118. This contradictory finding, in conjunction with the small change in proportion of BMD variability explained by this variable in the linear regression suggests that CB-118 is unlikely to exert an important influence on BMD in females in this population sample.
In this study, women had significantly higher levels of CB congener 118 and p,p´-DDE than did men. Consistently higher levels of CB-118 in women were also reported in Native Americans in the United States (Schaeffer et al. 2006), but higher levels of p,p´-DDE in women have not been reported elsewhere. In fact, other studies have reported no difference in p,p´-DDE levels by sex (Sandanger et al. 2006) or reported higher p,p´-DDE levels in males than in females (Bjerregaard et al. 2001; Jonsson et al. 2005). Lower levels of p,p´-DDE in younger females are likely attributable to breast-feeding (which may reduce plasma levels of persistent organic pollutants) and lower dietary exposure; as such, this sex difference is likely to be less evident in populations > 40 years of age, by which age the sex imbalance in organochlorine accumulation due to breast-feeding will be reduced (Sandanger et al. 2003).
These sex differences in organochlorine levels are unlikely to be explained by age, which was similar for the males and females in this study. The main source of exposure to organochlorines for this population is anticipated to be dietary, mainly via fish, to some extent caused by intake of salmon from the Emån river (a well-known salmon-fishing river), which has been polluted with PCBs from an upstream paper mill. More important, the local population may be exposed to PCBs by eating fish from the nearby Baltic Sea. Farmed salmon and wild herring in the Baltic Sea are known to have high concentrations of PCBs. Although individual-level data on fish consumption were not available for analysis, it has been reported by the Swedish National Food Administration that older women eat more fish than older men (Becker et al. 2007). The difference observed in levels of organochlorines in this study could therefore be attributable to a combination of factors including sex-specific differences in consumption patterns, differential metabolism (Landi et al. 1998), or elimination by lactation (Sandanger et al. 2003), although these mechanisms have not been shown to be specific to CB-118 or to p,p´-DDE.
No other epidemiologic studies have reported similar sex differences with respect to BMD to those found in this study, although most studies on this topic have focussed on single-sex populations. Similarly the literature on organochlorine exposure and sex-specific effects on BMD in experimental animals is very limited (van der Ven et al. 2006), although sex differences with respect to other outcomes have been observed, possibly indicating a greater sensitivity of males to reproductive system and neurobehavorial effects (Gray et al. 1997a, 1997b). Whether the likely hormonal action of organochlorines could explain or contribute to the different effects on BMD seen in males and females in this study is not clear.
The findings from this study suggest an effect of organochlorine exposure on BMD at levels of exposure experienced in a population in southern Sweden, but there are several limitations of the study that need to be considered. Although this is one of the largest studies to date, when males and females were split into separate groups for analysis, the power of the study to detect statistically significant relationships between organochlorine exposure and BMD was limited. In this study we had data on blood levels of organochlorines in adults, but we do not know about early life exposures, which may be more important than adult exposures in determining the effects of organochlorines on bone. Although it was possible to control for several potentially confounding variables, the linear regression analyses indicate that more than half the variability in BMD is likely to be explained by other, unknown variables that could not be taken into account in this study. Further work should be directed toward establishing whether the sex-specific effects observed in this study are also evident in other environmentally exposed populations. More detailed lifestyle data could be used to elucidate whether any sex-specific effects are caused by a hormonal action of the organochlorines (or the hormonal status of the individuals at the time of exposure), or whether these observations are due to other sex-specific differences related to lifestyle, diet, or occupation.
The purpose of this study was to investigate the relationship between environmental organochlorine exposure and BMD in a population potentially exposed to PCBs from the environment to add to the sparse and inconsistent literature on this important topic. The findings of this study indicate that exposure to some CB congeners, even at relatively low levels, may influence BMD and that this effect may be sex specific.
|
|
 |
| [References Listed in PubMed]
References Alfven T, Elinder CG, Carlsson MD, Grubb A, Hellstrom L, Persson B, et al. 2000. Low-level cadmium exposure and osteoporosis. J Bone Miner Res 15(8): 1579–1586.
Alfven T, Elinder CG, Hellstrom L, Lagarde F, Jarup L. 2004. Cadmium exposure and distal forearm fractures. J Bone Miner Res 19(6):900–905.
Alfven T, Jarup L, Elinder CG. 2002. Cadmium and lead in blood in relation to low bone mineral density and tubular proteinuria. Environ Health Perspect 110: 699–702.
Alveblom AK, Rylander L, Johnell O, Hagmar L. 2003. Incidence of hospitalized osteoporotic fractures in cohorts with high dietary intake of persistent organochlorine compounds. Int Arch Occup Environ Health 76(3):246–248.
Andrews JE. 1989. Polychlorinated biphenyl (Aroclor 1254) induced changes in femur morphometry calcium metabolism and nephrotoxicity. Toxicology 57(1):83–96.
Badraoui R, Abdelmoula NB, Sahnoun Z, Fakhfakh Z, Rebai T. 2007. Effect of subchronic exposure to tetradifon on bone remodelling and metabolism in female rat. Comptes Rend Biol 330(12):897–904.
Beard J, Marshall S, Jong K, Newton R, Triplett-McBride T, Humphries B, et al. 2000. 1,1,1-Trichloro-2,2-bis(p-chlorophenyl)-ethane (DDT) and reduced bone mineral density. Arch Environ Health 55(3):177–180.
Becker W, Darnerud PO, Petersson-Grawé K. 2007. Fiskkonsumtion—risk och nytta [in Swedish]. Uppsala, Sweden:National Food Administration.
Bjerregaard P, Dewailly E, Ayotte P, Pars T, Ferron L, Mulvad G. 2001. Exposure of Inuit in Greenland to organochlorines through the marine diet. J Toxicol Environ Health A 62(2):69–81.
Bohannon AD, Cooper GS, Wolff MS, Meier DE. 2000. Exposure to 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene (DDT) in relation to bone mineral density and rate of bone loss in menopausal women. Arch Environ Health 55(6):386–391.
Botella B, Crespo J, Rivas A, Cerrillo I, Olea-Serrano MF, Olea N. 2004. Exposure of women to organochlorine pesticides in Southern Spain. Environ Res 96(1): 34–40.
Bremle G, Larson P, Hammar T, Helgee A, Troedsson B. 1998. PCB in a river system during sediment remendation. Water Air Soil Pollut 107:237–250.
Cote S, Ayotte P, Dodin S, Blanchet C, Mulvad G, Petersen HS, et al. 2006. Plasma organochlorine concentrations and bone ultrasound measurements: a cross-sectional study in peri-and postmenopausal Inuit women from Greenland. Environmental Health: A Global Access Science Source 5:33. Available: www.ehjournal.net/content/pdf/1476-069X-5-33.pdf [accessed 28 July 2008].
Cripps DJ, Peters HA, Gocmen A, Dogramici I. 1984. Porphyria turcica due to hexachlorobenzene: a 20 to 30 year follow-up study on 204 patients. Br J Dermatol 111(4): 413–422.
European Food Safety Authority. 2005. Opinion of the scientific panel on contamination in the food chain on a request from the commission related to the presence of non dioxin-like polychlorinated biphenyls (PCB) in feed and food. Eur Food Safety Auth J 284:1–237.
Genant HK, Cooper C, Poor G, Reid I, Ehrlich G, Kanis J, et al. 1999. Interim report and recommendations of the World Health Organization Task-Force for Osteoporosis. Osteoporos Int 10(4):259–264.
Glynn AW, Michaelsson K, Lind PM, Wolk A, Aune M, Atuma S, et al. 2000a. Organochlorines and bone mineral density in Swedish men from the general population. Osteoporos Int 11(12):1036–1042.
Glynn AW, Wolk A, Aune M, Atuma S, Zettermark S, Maehle-Schmid M, et al. 2000b. Serum concentrations of organochlorines in men: a search for markers of exposure. Sci Total Environ 263(1–3):197–208.
Gocmen A, Peters HA, Cripps DJ, Bryan GT, Morris CR. 1989. Hexachlorobenzene episode in Turkey. Biomed Environ Sci 2(1):36–43.
Gray LE, Ostby JS, Kelce WR. 1997a. A dose-response analysis of the reproductive effects of a single gestational dose of 2,3,7,8-tetrachlorodibenzo-p-dioxin in male Long Evans hooded rat offspring. Toxicol Appl Pharmacol 146(1): 11–20.
Gray LE, Wolf C, Mann P, Ostby JS. 1997b. In utero exposure to low doses of 2,3,7,8-tetrachlorodibenzo-p-dioxin alters reproductive development of female Long Evans hooded rat offspring. Toxicol Appl Pharmacol 146(2):237–244.
Ismail AA, Pye SR, Cockerill WC, Lunt M, Silman AJ, Reeve J, et al. 2002. Incidence of limb fracture across Europe: results from the European Prospective Osteoporosis Study (EPOS). Osteoporos Int 13(7):565–571.
Jamsa T, Viluksela M, Tuomisto JT, Tuomisto J, Tuukkanen J. 2001. Effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin on bone in two rat strains with different aryl hydrocarbon receptor structures. J Bone Miner Res 16(10):1812–1820.
Janak K, Jensen E, Becher G. 1999. Determination of polychlorinated biphenyls in human blood by solid-phase extraction including on-column lipid decomposition. J Chromatogr B Biomed Sci Appl 734(2):219–227.
Jarup L, Alfven T, Persson B, Toss G, Elinder CG. 1998. Cadmium may be a risk factor for osteoporosis. Occup Environ Med 55(7):435–439.
Jonsson BAG, Rylander L, Lindh C, Rignell-Hydbom A, Giwercman A, Toft G, et al. 2005. Inter-population variations in concentrations, determinants of and correlations between 2,2,´4,4,´5,5´-hexachlorobiphenyl (CB-153) and 1,1-dichloro-2,2-bis (p-chlorophenyl)-ethylene (p,p´-DDE): a cross-sectional study of 3161 men and women from Inuit and European populations. Environmental Health: A Global Access Science Source 4:27. Available: www.ehjournal.net/content/pdf/1476-069X-4-27.pdf [accessed 28 July 2008].
Kanis JA, Delmas P, Burckhardt P, Cooper C, Torgerson D. 1997. Guidelines for diagnosis and management of osteoporosis. The European Foundation for Osteoporosis and Bone Disease. Osteoporos Int 7(4):390–406.
Landi MT, Consonni D, Patterson DG Jr, Needham LL, Lucier G, Brambilla P, et al. 1998. 2,3,7,8-Tetrachlorodibenzo-p-dioxin plasma levels in Seveso 20 years after the accident. Environ Health Perspect 106:273–277.
Lind PM, Eriksen EF, Lind L, Orberg J, Sahlin L. 2004. Estrogen supplementation modulates effects of the endocrine disrupting pollutant PCB126 in rat bone and uterus: diverging effects in ovariectomized and intact animals. Toxicology 199(2–3):129–136.
Lind PM, Eriksen EF, Sahlin L, Edlund M, Orberg J. 1999. Effects of the antiestrogenic environmental pollutant 3,3´,4,4´,5-pentachlorobiphenyl (PCB #126) in rat bone and uterus: diverging effects in ovariectomized and intact animals. Toxicol Appl Pharmacol 154(3):236–244.
Lind PM, Larsson S, Oxlund H, Hakansson H, Nyberg K, Eklund T, et al. 2000. Change of bone tissue composition and impaired bone strength in rats exposed to 3,3´,4,4´,5-pentachlorobiphenyl (PCB126). Toxicology 150:41–51.
Mariani G, Carasi S, Fattore E, Nichetti S, Guzzi A, Benfenati E, et al. 2002. Fast and simple method for PCB analysis in human serum samples by GC-NICI-MS. Organohalogen Compounds 55:111–114.
Miettinen HM, Pulkkinen P, Jamsa T, Koistinen J, Simanainen U, Tuomisto J, et al. 2005. Effects of in utero and lactational TCDD exposure on bone development in differentially sensitive rat lines. Toxicol Sci 85(2):1003–1012.
Miller RW. 1985. Congenital PCB poisoning: a reevaluation. Environ Health Perspect 60:211–214.
Sandanger TM, Brustad M, Odland J, Doudarev AA, Miretsky GI, Chaschin V, et al. 2003. Human plasma levels of POPs, and diet among native people from Uelen, Chukotka. J Environ Monit 5(4):689–696.
Sandanger TM, Brustad M, Sandau CD, Lund E. 2006. Levels of persistent organic pollutants (POPs) in a coastal northern Norwegian population with high fish-liver intake. J Environ Monit 8(5):552–557.
Schaeffer DJ, Dellinger JA, Needham LL, Hansen LG. 2006. Serum PCB profiles in Native Americans from Wisconsin based on region, diet, age, and gender: Implications for epidemiology studies. Sci Total Environ 357(1–3): 74–87.
Schlenker RA, VonSeggen WW. 1976. The distribution of cortical and trabecular bone mass along the lengths of the radius and ulna and the implications for in vivo bone mass measurements. Calcified Tissue Res 20(1):41–52.
Turci R, Mariani G, Marinaccio A, Balducci C, Bettinelli M, Fanelli R, et al. 2004. Critical evaluation of a high-throughput analytical method for polychlorinated biphenyls in human serum: which detector for the establishment of the reference values? Rapid Commun Mass Spectrom 18(4): 421–434.
Van den Berg M, Birnbaum LS, Denison M, De Vito M, Farland W, Feeley M, et al. 2006. The 2005 World Health Organization reevaluation of human and Mammalian toxic equivalency factors for dioxins and dioxin-like compounds. Toxicol Sci 93(2):223–241.
van der Ven LT, Verhoef A, van de Kuil T, Slob W, Leonards PE, Visser TJ, et al. 2006. A 28-day oral dose toxicity study enhanced to detect endocrine effects of hexabromocyclododecane in Wistar rats. Toxicol Sci 94(2): 281–292.
Wallin E, Rylander L, Jonssson BA, Lundh T, Isaksson A, Hagmar L. 2005. Exposure to CB-153 and p,p´-DDE and bone mineral density and bone metabolism markers in middle-aged and elderly men and women. Osteoporos Int 16(12): 2085–2094.
Wilhelm M, Ewers U, Schulz C. 2003. Revised and new reference values for some persistent organic pollutants (POPs) in blood for human biomonitoring in environmental medicine. Int J Hyg Environ Health 206(3): 223–229.
Last Updated: August 15, 2008
|
|
 |
|
| |

