
| |  | |  |
Children's Health
|
| The Challenge of Preventing Environmentally Related Disease in Young Children: Community-Based Research in New York City Frederica P. Perera,1 Susan M. Illman,1 Patrick L. Kinney,1 Robin M. Whyatt,1 Elizabeth A. Kelvin,1 Peggy Shepard,1,2 David Evans,1 Mindy Fullilove,1 Jean Ford,1 Rachel L. Miller,1 Ilan H. Meyer,1 and Virginia A. Rauh1 1Columbia Center for Children's Environmental Health, Mailman School of Public Health, Columbia University, New York, New York, USA; 2West Harlem Environmental Action, Inc., New York, New York, USA Abstract
Rates of developmental and respiratory diseases are disproportionately high in underserved, minority populations such as those in New York City's Washington Heights, Harlem, and the South Bronx. Blacks and Latinos in these neighborhoods represent high risk groups for asthma, adverse birth outcomes, impaired development, and some types of cancer. The Columbia Center for Children's Environmental Health in Washington Heights uses molecular epidemiologic methods to study the health effects of urban indoor and outdoor air pollutants on children, prenatally and postnatally, in a cohort of over 500 African-American and Dominican (originally from the Dominican Republic) mothers and newborns. Extensive data are collected to determine exposures to particulate matter < 2.5 µm in aerodynamic diameter (PM2.5) , polycyclic aromatic hydrocarbons (PAHs) , diesel exhaust particulate (DEP) , nitrogen oxide, nonpersistent pesticides, home allergens (dust mite, mouse, cockroach) , environmental tobacco smoke (ETS) , and lead and other metals. Biomarkers, air sampling, and clinical assessments are used to study the effects of these exposures on children's increased risk for allergic sensitization, asthma and other respiratory disorders, impairment of neurocognitive and behavioral development, and potential cancer risk. The center conducts its research and community education in collaboration with 10 community-based health and environmental advocacy organizations. This unique academic-community partnership helps to guide the center's research so that it is most relevant to the context of the low-income, minority neighborhoods in which the cohort resides, and information is delivered back to these communities in meaningful ways. In turn, communities become better equipped to relay environmental health concerns to policy makers. In this paper we describe the center's research and its academic-community partnership and present some preliminary findings. Key words: asthma, cancer, children, development, environmental health, prenatal, prevention, susceptibility. Environ Health Perspect 110:197-204 (2002) . [Online 18 January 2002] http://ehpnet1.niehs.nih.gov/docs/2002/110p197-204perera/ abstract.html Address correspondence to F.P. Perera, Division of Environmental Health Sciences, Joseph L. Mailman School of Public Health, Columbia University, 60 Haven Avenue, B-116, New York, NY 10032 USA. Telephone: (212) 304-7280. Fax: (212) 544-1943. E-mail: fpp1@columbia.edu This work was supported by the National Institute of Environmental Health Sciences (grants P50 ES09600, RO1 ES08977, and RO1 ES06722) and the U.S. Environmental Protection Agency (grant R827027) , the Bauman Family Foundation, the Gladys and Roland Harriman Foundation, the W. Alton Jones Foundation, the New York Community Trust, and the Irving General Clinical Research Center (grant RR00645) . Received 8 January 2001 ; accepted 18 July 2001. |
|
|
 |
The Columbia Center for Children's Environmental Health (CCCEH) is studying the health effects of exposure to several common urban air pollutants in a cohort of over 500 African-American and Dominican (originally from the Dominican Republic) mothers and children residing in three low-income neighborhoods in New York City: Washington Heights, Harlem, and the South Bronx. The center's mission is to identify prenatal and postnatal exposures that increase children's risk for asthma and other respiratory disorders, neurocognitive and behavioral disorders, and cancer risk, so that the most harmful exposures can be reduced or eliminated.
Molecular epidemiologic methods are used to study the correlation between levels of exposure effects found in personal and indoor/outdoor air monitoring samples, biologic samples (i.e., maternal blood and urine, umbilical cord blood, meconium, and children's blood at 2 years of age), neurodevelopmental assessment results, and questionnaire responses. Psychosocial stressors and nutritional deficits are being measured as potential effect modifiers. This comprehensive data collection, repeated at regular time intervals from in utero through 3 years of age, affords CCCEH researchers the ability to determine prenatal and postnatal levels of exposure that may increase children's risk of the outcomes under study.
The center is measuring exposure to a range of air contaminants that are common in Washington Heights, Harlem, and the South Bronx: particulate matter < 2.5 µm in aerodynamic diameter (PM2.5), polycyclic aromatic hydrocarbons (PAHs), diesel exhaust particulate (DEP), nitrogen oxide, nonpersistent pesticides (NPPs), home allergens (dust mite, mouse, cockroach), environmental tobacco smoke (ETS), and lead and other metals such as mercury. These two Northern Manhattan neighborhoods and the South Bronx are located in a densely populated metropolitan region that has in recent years exceeded the annual PM10 (particulate matter < 10 µm in aerodynamic diameter) standard of 50 µg/m3. In addition, these lower-income neighborhoods contain substantial local sources of combustion-generated pollution. Infants and young children in urban areas also spend a large part of time indoors where irritating and allergenic substances (i.e., gaseous and particulate emissions from gas stoves, space heaters, cigarettes, pest populations, and NPPs) may increase susceptibility to allergic sensitization, respiratory symptoms, and eventually, the development of asthma.
In this paper, we describe the unique susceptibility of the fetus and infant as well as additional contributing risk factors, including environmental exposures and disease rates in the three neighborhoods from which the study cohort is derived and the CCCEH's innovative research methods and preliminary results. We also describe a community education campaign called "Healthy Home Healthy Child," which involves a partnership of 10 community-based direct service health and environmental advocacy organizations whose health educators inform neighborhood residents about how to protect themselves from unnecessary risk.
 There is mounting evidence that the fetus and infant are significantly more sensitive to a variety of environmental toxicants than adults because of differential exposure, physiologic immaturity, and a longer lifetime over which disease initiated in early life can develop. For example, experimental and human data indicate that the fetus and young child are especially vulnerable to the toxic effects of ETS, PAHs, particulate matter, nitrosamines, pesticides, polychlorinated biphenyls (PCBs), metals, and radiation (1-5) (Table 1). There is mounting evidence that the fetus and infant are significantly more sensitive to a variety of environmental toxicants than adults because of differential exposure, physiologic immaturity, and a longer lifetime over which disease initiated in early life can develop. For example, experimental and human data indicate that the fetus and young child are especially vulnerable to the toxic effects of ETS, PAHs, particulate matter, nitrosamines, pesticides, polychlorinated biphenyls (PCBs), metals, and radiation (1-5) (Table 1).
Genetic damage/potential cancer risk. Several studies suggest that the fetus clears toxicants less efficiently than the adult and may be more vulnerable to genetic damage and the resultant risk of cancer. For example, given experimental evidence that the amount of PAH crossing the placenta and reaching the fetus is less than one-tenth of the dose to the mother (10,11), the levels of PAH-DNA adducts measured in rodent fetal tissue are higher than expected (12,13). Similarly, our research in Poland has shown that PAH-DNA adduct levels in the white blood cells of newborns actually exceeded those in paired maternal samples, despite the estimated 10-fold lower dose of the parent compound to the fetus (13,14). In addition, plasma cotinine and aromatic DNA adduct levels in the Polish newborns were higher than in paired maternal samples, suggesting reduced ability of the fetus to clear cigarette smoke constituents (13,14). Increased adducts in the fetus relative to the adult could result from lower levels of phase II (detoxification) enzymes and decreased DNA repair efficiency in the fetus (3,14-16).
Respiratory disease. With respect to respiratory disease, there are critical windows in both prenatal and postnatal development during which exposure to irritants and other toxicants can modify the formation and maturation of the lung. The complete development of the human lung occurs through the sixth to eighth years of life (17). There is recent evidence from the CCCEH study of pregnant women and children that in utero sensitization to specific allergens can occur independent of maternal sensitization, possibly putting the child at higher risk of asthma (18). Children have been identified as a sensitive population to particulate matter, especially in those with respiratory symptoms (19).
Neurologic development. The exquisitely sensitive process of development of the human central nervous system has been reviewed by Faustman (20). This process involves the production of 100 billion nerve cells and 1 trillion glial cells, which then must follow a precise stepwise choreography involving migration, synaptogenesis, selective cell loss, and myelination (20). A mistake at any one step can have permanent consequences. Experimental studies of prenatal and neonatal exposure to chlorpyrifos have reported neurochemical and behavioral effects as well as selected brain cell loss (21,22). The behavioral and morphologic effects of developmental toxicants are highly dependent on the timing as well as on the dose and duration of exposure. This is illustrated by both rodent and human studies showing that the effect of irradiation on brain malformation is heightened during a window of susceptibility of fetal development (20).
The enhanced susceptibility of the fetus and newborn is likely to be compounded by cofactors including nutritional deficits, genes, and social stressors.
Nutrition. Deficits in antioxidants have been strongly implicated in asthma. These micronutrients can moderate the effect of oxidants on lung function, reducing oxidative stress and resultant tissue damage and airway inflammation (23-25). With respect to growth and development and cancer, antioxidants remove free radicals and oxidant intermediates, thereby inhibiting chemical-DNA binding that has been associated with decreased weight, length, and head circumference at birth (9) and also with cancer (26). In addition, essential fatty acid status contributes to observed variations in cognitive and motor function and to low birth weight and reduced head circumference (27-29). Nutritional deficits are associated with poverty, although there is interindividual variation in nutritional status within socioeconomically disadvantaged populations.
Genetics. Genetic susceptibility can take the form of common polymorphisms that affect the toxicity to the individual. For example, there are two genes that can increase an individual's vulnerability to organophosphates (OPs) such as chlorpyrifos by reducing the reservoir of functioning protective enzymes (30). The first gene has a prevalence of 4% and results in a poorly functioning form of the enzyme acetycholinesterase; the second gene results in a relatively inactive form of the enzyme, paraoxonase (prevalence of 30-38%) (20,30). Other examples of gene-environment interactions involve the gene coding for the 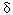 -ALA enzyme that affects lead metabolism and storage (30), and the P450 and glutathione-S-transferase genes that play a role in activation and detoxification of PAH and influence PAH-DNA damage (30). Genetic susceptibility may vary by race and ethnicity. -ALA enzyme that affects lead metabolism and storage (30), and the P450 and glutathione-S-transferase genes that play a role in activation and detoxification of PAH and influence PAH-DNA damage (30). Genetic susceptibility may vary by race and ethnicity.
Individual- and community-level psychosocial stressors. The notion that community-level conditions can produce profound effects on host susceptibility to disease is derived from the long-standing existence of strong social class gradients in health (31). Recent studies have shown that women who live in violent, crime-ridden, physically decayed neighborhoods are more likely to experience pregnancy complications and adverse birth outcomes, after adjusting for a range of individual level sociodemographic attributes and health behaviors (32,33). Other studies have suggested that the stresses of racism and community segregation are associated with lower birth weight (34,35). Several studies have shown that the effects of individual poverty on birth outcomes are exacerbated by residence in a disadvantaged neighborhood (36).
Children in the United States suffer from unacceptably high rates of developmental disorders, asthma, and cancer [Table 2 ( 30,37-42)]. Rates of asthma and behavioral disorders have increased in the past decades ( 30). Although improved detection and reporting have contributed, environmental factors are known or suspected to play a role. The rates of these diseases are disproportionately high in underserved, minority populations such as those in New York City where CCCEH is located. African Americans and Latinos in Northern Manhattan and the South Bronx represent high risk groups for asthma, adverse birth outcomes, impaired development, and some types of cancer ( 43,44) (Table 2).

Washington Heights, a low-income community in northern Manhattan, has a large Latino population. According to 1990 census data (45), the median household income in 1989 was $22,175, with 29.4% of the population living below the poverty level (46). Two-thirds (66.9%) of residents were Latino, with 65.2% being Dominican. Central Harlem is also a low-income minority community that was 91.9% African American in 1990 (45). The median household income in 1989 was $13,861, and 38.9% of the population lived below the poverty level. The South Bronx is another low-income minority community made up largely of Latinos (57.2%) and African Americans (31.4%). The median household income in 1989 was $12,088, and 45.8% of the population lived below the poverty level.
PM2.5, diesel exhaust particulate, and PAHs. Harlem, Washington Heights, and the South Bronx are at the center of a large sprawling metropolitan region that in recent years has been out of compliance with the National Ambient Air Quality Standard for particulate matter, exceeding the annual PM10 standard of 50 µg/m3 (47). The regional influx of polluted air is augmented by substantial local sources of combustion-generated pollution. Ambient PM2.5, DEP, and PAH levels result from region-wide pollution emissions upwind of New York City, as well as from local sources such as diesel bus depots, waste incinerators, industrial operations, and a network of commuter highways.
Foremost among the local combustion sources are the cars, trucks, and buses using the highways and the commercial truck and bus routes that surround and interlace Harlem and Washington Heights. Two of the major north/south avenues passing through the center of these communities--Broadway and Amsterdam Avenues--are the principal truck routes for moving goods in and out of Manhattan. In addition, diesel bus depots, waste incinerators, and a multitude of small industrial operations release substantial amounts of airborne particulates and gaseous pollutants in these areas. Diesel engines emit 30-100 times more particles than are emitted by gasoline engines that have contemporary emission-control devices (48). Sources of diesel particulate located in northern Manhattan include six Metropolitan Transit Authority bus garages, each one housing from 200 to 400 diesel buses, a large marine transfer station, and a commerical bus terminal.
The South Bronx is similarly congested, with local pollution point sources such as a sewage waste treatment plant, traffic from seven bridges feeding into the area, as well as excess trucks and buses passing through regularly en route to the Harlem River Yards, bus depots, Hunts Point Terminal Food Distribution Center, and the New York Post printing plant. Commerce from the Hunts Point Market alone brings roughly 7,000 diesel trucks and tractor-trailer trucks into the neighborhood daily (49). The majority of the borough's waste facilities and transfer stations located in the South Bronx process 21% of the city's commercial waste and pelletize 70% of the city's sewage sludge (49). The South Bronx is located beneath the takeoff and landing corridors of LaGuardia Airport and a mile from two power plants, which are the second and fourth dirtiest in New York State (50). Residents are justifiably concerned about the potential health impacts of the many ambient pollution sources on children growing up in these communities.
The impact of diesel bus and truck traffic (DEP) on the spatial variability of fine elemental carbon particle concentrations in Harlem was demonstrated in a community-based pilot study conducted by CCCEH scientists along with high school interns from West Harlem Environmental Action (WE ACT), the center's lead community partner (51). Over a 5-day period, researchers wearing backpacks containing personal ambient air monitors that collected information on fine particle (PM2.5) and elemental carbon concentrations in the air they breathed counted trucks, buses, cars, and pedestrians at four intersections in Harlem between 1000 and 1800 hr. This study showed that reflectance measurement of the "blackness" of the particulate sample on the filter is highly correlated with elemental carbon concentration (r = 0.95) and may be used as a surrogate for diesel exposure (51). Results also showed that diesel traffic density varied widely at the four locations, with 8-hr PM2.5 concentrations ranging from 22 to 69 µg/m3. For comparison, the annual fine particle standard proposed by the U.S. Environmental Protection Agency (EPA) in 1998 is 15 µg/m3 (52).
Environmental tobacco smoke. Recent research indicates that ETS exposure is more prevalent among African Americans and Hispanics than whites (53-55). In addition, there is evidence that minorities are more susceptible to the chemicals in tobacco smoke. Higher levels of cotinine and a tobacco-specific carcinogenic nitrosamine have been reported in black smokers than in white smokers, after controlling for self-reported amount of smoking (55-57). In a recent study, African-American children had 2-fold higher cotinine levels than white children as a result of exposure to 1 cigarette/day (58). Similarly, after adjusting for cigarette dose, cotinine levels in pregnant women were higher in African Americans than in whites, while the rate of decrease in infant birth weight per nanogram of maternal cotinine was similar in the two groups (59). These findings point to the possibility that cigarette smoking may have a more deleterious effect on fetal development among African Americans than among whites (59).
Pest allergens. Goldstein et al. (60) reported levels of airborne cockroach allergen in Harlem apartments that were orders of magnitude higher than those seen in previous studies in New Orleans, Louisiana, and Rochester, Minnesota. Sarpong et al. (61) found that African-American race was a predictor of higher allergen exposures. As will be discussed below, 85% of the homes of pregnant women studied by the CCCEH had detectable cockroach allergen levels.
The context for our research is the high rates of neurodevelopmental disorders, asthma, and cancer in children in the United States ( 30). Although improved detection and reporting have contributed to marked increases in some of these disorders over the past decade, environmental factors are known or suspected to affect these increases. The rates of neurodevelopmental disorders, asthma, and cancer in children are disproportionately high in the underserved, minority populations of Washington Heights, Harlem, and the South Bronx ( 43,44,48).
Asthma. Pediatric asthma is a serious and growing public health problem in the United States (62). New York City is one of four metropolitan areas in the country with the highest annual increase in asthma mortality (34). Asthma rates vary markedly within New York City. Five of the seven New York City zip code areas with the highest asthma hospitalization rates are located in Harlem (42). In the South Bronx in 1994, prevalence of asthma among children < 17 years of age was 17.9% in Hispanics, 11.6% among non-Hispanic blacks, and 8.2% among whites (41). A recent study of pediatric asthma rates suggested that material and behavioral characteristics associated with poverty, such as parental smoking, air pollution, housing conditions, and allergens, may contribute to the disparities (63). Early life wheezing, especially in the presence of atopy and sensitization to environmental allergens, appears to be a good predictor of persistent wheezing and asthma (64-66).
Impairment of fetal growth and child development. Low birth weight is the second leading predictor of infant mortality in the United States as well as a major cause of delayed development (67,68) and a risk factor for childhood asthma (26). As a whole, the largely minority population in our three target communities is at elevated risk for low birth weight and subsequent cognitive delay compared to other U.S. populations, but here, too, rates vary. In 1997, the incidence of low birth weight was 13.5% in central Harlem, 10.5% in the South Bronx, and 7.7% in Washington Heights, compared to 7.1% in whites in New York City (69). Children are also at elevated risk of subsequent cognitive delay compared to other populations: 68% of elementary school children in Washington Heights and 74% in central Harlem are reading below grade level, compared with 46% city-wide (38).
Cancer and other outcomes. Nationally, African Americans continue to exceed white Americans in deaths from diseases with known or suspected environmental components, including cancer (70,71). Early life exposures may be important determinants of risk. It has been estimated that for genotoxic carcinogens, as much as one-half of total lifetime cancer risk may be accrued before the age of 6 years (72).
The CCCEH is one of eight national Children's Centers established in 1998 by National Institute of Environmental Health Sciences and the U.S. EPA. The center's mission is to prevent environmentally related disease in children, specifically developmental disorders, asthma, and early damage leading to increased risk of cancer. The center is based on a broad definition of "environmental toxicity" that includes toxic contaminants in the indoor and outdoor environment as well as genetic, nutritional, and community-level risk factors, including socioeconomic disadvantage, that can adversely affect the health of populations ( 73).
The specific goals of the CCCEH are to assess environmental risks to infant and child health and to test interventions to reduce those risks. The CCCEH is conducting related research projects on the impact on children's health of prenatal and postnatal environmental exposures to airborne particulate matter (PM2.5), PAHs, NO2, ETS, pesticides, and home allergens. The CCCEH is also examining the possibility that inadequate nutrition and socioeconomic and other stressors exacerbate the impact of these toxicants.
At the heart of the center's research is a prospective cohort of over 500 nonsmoking pregnant women (of which 370 women who are fully enrolled have been monitored prenatally, delivered, and given biologic samples, and are undergoing postnatal assessments). The women are either African American or Dominican and all reside in Washington Heights, Harlem, or the South Bronx. The women and their babies, once born, are closely monitored during the years after birth. Figure 1 summarizes the research design in which biomarkers play a central role. The research team gathers information from each pregnant woman to estimate her exposure to particular allergens, indoor and outdoor pollutants, and pesticides, as well as social stressors, and to monitor her health.
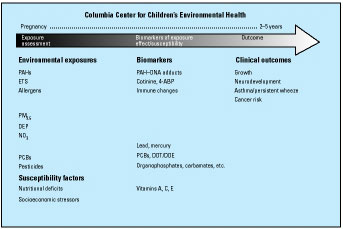
Figure 1. Summary of CCCEH research.
In the third trimester of pregnancy, each woman carries a portable monitor, which she keeps with her at all times during a 48-hr period. The monitor measures and records the levels of pollution the woman is inhaling and possibly passing on to her unborn baby. The pumps operate continuously at 4 L/min, collecting particles of 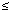 2.5 µm in diameter on a precleaned quartz microfiber filter and semivolativel vapors and aerosols on a polyurethane foam cartridge backup. Samples of dust in the home are analyzed for pollutants, pesticides, and allergens. The monitoring is repeated during successive years. In addition to a urine sample collected during pregnancy, blood is collected from each woman after giving birth, and from the infant's umbilical cord to test for biomarkers that indicate exposure to common home allergens (including cockroach, dust mite, and rodent), air pollutants (including ETS and PAHs), pesticides, and PCBs, and for nutritional deficits, indicated by low levels of antioxidants. 2.5 µm in diameter on a precleaned quartz microfiber filter and semivolativel vapors and aerosols on a polyurethane foam cartridge backup. Samples of dust in the home are analyzed for pollutants, pesticides, and allergens. The monitoring is repeated during successive years. In addition to a urine sample collected during pregnancy, blood is collected from each woman after giving birth, and from the infant's umbilical cord to test for biomarkers that indicate exposure to common home allergens (including cockroach, dust mite, and rodent), air pollutants (including ETS and PAHs), pesticides, and PCBs, and for nutritional deficits, indicated by low levels of antioxidants.
Biomarkers of immune changes and procarcinogenic DNA damage are also measured in the blood samples. The child's blood is tested again at 2 years of age. After delivery, the health of each baby is followed closely to check for respiratory problems and other early signs of asthma and developmental disorders. CCCEH staff interview the mothers every 3 months and collect information about each child's growth, feeding, allergies, and medical condition.
The descriptive results briefly summarized here are based on samples analyzed from the first 250 pregnant women enrolled in the study cohort unless otherwise noted.
PAHs. Analysis of personal air samples from the first 250 pregnant women enrolled showed universally detectable levels of one or more carcinogenic PAHs. Total PAH exposures averaged 4.0 ng/m3 and varied significantly among the women, with a range of 0.36-44.81 ng/m3 (74). These PAH concentrations can be compared to those in a previous study of nonsmoking women who worked outdoors in the Czech Republic, a country with elevated air pollution from coal burning (average of 12.5 ± 6.8 ng/m3, range 2.9-26 ng/m3, in one of the most polluted areas) (75). Thus the mean PAH concentration in the New York City population is lower, but the range is wider and the upper bound higher. To our knowledge, this is the first study in the United States to quantify personal exposures to PAHs among urban women.
Environmental tobacco smoke. There was also widespread exposure to ETS. Analysis of cotinine in the first 224 maternal and newborn blood samples collected showed that 41% of mother/newborn pairs had cotinine levels < 25 ng/mL. Of these, 49% of infants and 53% of mothers had levels of cotinine indicative of ETS exposure ( 0.03-25 ng/mL). Maternal and newborn plasma cotinine levels were significantly higher if the mother reported smoking by household members or regular visitors, compared to mothers who reported no smoking in the house (p < 0.001 Mann-Whitney test). 0.03-25 ng/mL). Maternal and newborn plasma cotinine levels were significantly higher if the mother reported smoking by household members or regular visitors, compared to mothers who reported no smoking in the house (p < 0.001 Mann-Whitney test).
Pesticides. Analysis of the first 127 personal air samples tested showed that all of the women and developing fetuses were exposed to at least three different pesticides known to be neurotoxic: the OPs chlorpyriphos and diazinon, and the carbamate propoxur (74). These pesticides are widely used to control cockroaches and other pests in urban homes. The range of exposure was similar to that seen in the Non-Occupational Exposures to Pesticides Study (NOPES) among residents in Jacksonville, Florida, a location selected to represent an area with high household pesticide use (76).
Nonpersistent pesticides. Levels of 29 NPPs widely used for residential pest control in these minority communities (including chlorpyrifos, propoxur, bendiocarb, carbaryl, and diazinon), have also been measured in a subset of urine samples collected from 60 women during pregnancy, as well as in 142 maternal and 155 cord blood samples collected at delivery. Pesticides and/or their metabolites were commonly detected in these biologic samples. In general, cord pesticide levels were similar to and, in most cases, significantly correlated with levels detected in maternal blood. Collectively, these results show substantial NPP exposure among the cohort. Consistent with experimental data, these findings suggest that pesticides are readily transferred from the mother to the developing fetus.
Validating a new biomarker. Measurements of xenobiotics in meconium may provide dosimeters that integrate exposure during the latter half of gestation. In a validation study, OP metabolites collected from 20 noncohort newborns were measured in meconium samples by laboratory technicians blinded to the babies' pesticide exposure status (77). Six metabolites were analyzed: diethylphosphate, diethylthiophosphate, dimethylphosphate, diethyldithiophosphate, dimethylthiophosphate, and dimethyldithiophosphate. Results indicate that OP metabolites in meconium may have utility as a biomarker of prenatal exposure. OP levels were detected frequently in the meconium samples, and detection limits were comparable to or lower than those in urine. Metabolite levels in meconium measured several orders of magnitude higher than those usually detected in umbilical cord blood samples.
Pest allergens. In the initial 117 prenatal dust samples analyzed, high levels of cockroach (63%, 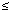 798 U/g) and mouse (46%, 798 U/g) and mouse (46%, 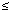 1,478 µg/g) allergens were detected in kitchens, but low levels of dust mite allergens were detected. Bla g 2 exposure has been associated with asthma morbidity (78). The high correlation (r = 0.82) between the two cockroach Blatella germanica allergens, Bla g 1 and Bla g 2, led us to discontinue further analyses of Bla g 1. Analysis of dust mite allergens has also been discontinued because the dust mite Dermatophagoides pteronyssinus is less viable in New York City apartments due to its need for high humidity. Homes of 158 1-year-old children have been revisited to collect postnatal dustborne allergens. Cockroach allergens were observed in 80% of postnatal kitchen dust samples ( 1,478 µg/g) allergens were detected in kitchens, but low levels of dust mite allergens were detected. Bla g 2 exposure has been associated with asthma morbidity (78). The high correlation (r = 0.82) between the two cockroach Blatella germanica allergens, Bla g 1 and Bla g 2, led us to discontinue further analyses of Bla g 1. Analysis of dust mite allergens has also been discontinued because the dust mite Dermatophagoides pteronyssinus is less viable in New York City apartments due to its need for high humidity. Homes of 158 1-year-old children have been revisited to collect postnatal dustborne allergens. Cockroach allergens were observed in 80% of postnatal kitchen dust samples (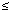 325 U/g), and mouse allergen was detected in 58% of kitchen samples ( 325 U/g), and mouse allergen was detected in 58% of kitchen samples (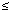 723 µg/g). These data show that exposure to these allergens is prevalent, ranging up to levels associated with health effects. 723 µg/g). These data show that exposure to these allergens is prevalent, ranging up to levels associated with health effects.
Allergic sensitization. Immune biomarkers have been analyzed in the initial subset of mothers and newborns, including total IgE levels, allergen-specific IgE levels, mitogen or allergen-induced lymphocyte proliferation, and cytokines. Among the most important findings to date are that in the first 167 women enrolled and their babies, there was a high prevalence of in utero allergic sensitization as evidenced by the lymphocyte proliferation assay (79). For example, as many as 54% of the lymphocyte samples from newborns proliferated in response to cockroach allergen. In many cases, cord blood allergen-induced proliferation occurred in the absence of maternal blood allergen-induced proliferation. Although only low levels of dust mite allergens have been detected in dust samples, 65% of maternal and 40% of newborn blood samples have proliferated in reponse to dust mite allergen (79). Prospective follow-up of the cohort enables us to determine the clinical significance of these early biomarkers and to determine which biomarkers may identify a child at risk for subsequent asthma.
Respiratory symptoms. Preliminary analyses of the first 158 infants enrolled showed that 46% experienced wheezing and other respiratory symptoms between birth and 3 months of age, and had slightly greater symptom occurrence at 6 (54%) and 9 months of age (50%). These symptom levels corresponded with a high rate of doctor and emergency department visits for respiratory problems (80). It is unknown at this time whether these respiratory symptoms at such a young age indicate a greater risk for developing asthma between ages 4 and 5 years, but continued follow-up of this cohort will reveal if these early symptoms are predictive. In a cross-sectional study that surveyed the asthma prevalence of 1,285 children < 17 years of age living in the Bronx, 14.3% of the children had asthma at one time and 8.6% suffered from asthma in the last year (81). This asthma prevalence rate among Bronx children is twice the U.S. rate (4.3%).
Lead and mercury. In the initial 137 samples analyzed, umbilical cord lead levels ranged from nondetectable to 7.6 µg/dL, while mercury levels ranged from nondetectable to 9.4 µg/L. These levels are below the levels of immediate health concern for both contaminants ( 10 µg/dL for lead, 10 µg/dL for lead,  30 µg/L for mercury), but thresholds have not been established for their neurotoxic effects. 30 µg/L for mercury), but thresholds have not been established for their neurotoxic effects.
Organochlorines. Analyses of PCBs, DDT/DDE, and other organochlorines have been completed on 105 plasma samples. Levels were low and generally nondetectable, thus further analyses have been discontinued.
This is the first study to comprehensively measure pollutants and dustborne allergens in homes of newborns and young children in communities of color in northern Manhattan and the South Bronx. The preliminary results show that exposures are variable (over several orders of magnitude), with cockroach allergen levels ranging above concentrations associated with asthma morbidity (78,82,83) and pesticide levels that exceed current health-based standards. Although their full significance for children's health remains to be determined, these findings indicate substantial in utero exposure and even early signs of potential risk from toxicants known or suspected to be capable of contributing to the childhood illnesses and disorders of concern to center investigators.
The CCCEH is modeled on the premise that the challenge of prevention requires an interdisciplinary, community-based research strategy to identify preventable risk factors and act upon that information. It incorporates both molecular epidemiology and multilevel analyses ( 84,85). Molecular epidemiology using biomarkers is a relatively new and useful tool in defining environment-susceptibility relationships, when used in conjunction with reliable monitoring and epidemiologic methodologies to provide individual estimates of exposure, dose, biologic response and susceptibility to pollutants ( 86-93). The center's research also follows the paradigm of the Chinese box described by Susser and Susser ( 85) in that it aims to integrate mutiple levels of organization (individual/molecular and community/ecologic) in design, analysis, and interpretation, nesting the individual level analysis within the overarching environment of the community.
Within the center's study area of northern Manhattan and the South Bronx, there is considerable variation in exposure, susceptibility factors, and risk belying the stereotype of a uniformly disadvantaged population. This allows us to study dose-response relationships as well as community impacts on individual outcomes. We anticipate that this multilevel approach will provide a more complete understanding of the complexity of the disease process, permitting the development of a variety of independent and complementary approaches to intervention at the individual and macro levels. These interventions might include education regarding lifestyle changes, regulations to control involuntary exposure to toxic pollutants, and even broader social policy changes.
The center has an active group of advisors on its Community Advisory Board (CAB). WE ACT is the center's lead community partner and has advocated for improved environmental conditions in Harlem for over a decade. Directors of nine additional health service and environmental advocacy organizations that are well-established in Washington Heights, Harlem, and the South Bronx also serve on the CAB: Alianza Dominicana, Best Beginnings, Community Association of Progressive Dominicans, E.C.H.O. for Sustainable Development, Harlem Dowling West Side, Heart of Harlem, Northern Manhattan Perinatal Partnership, St. Mary's Episcopal Church, and The South Bronx Clean Air Coalition. For several years, these organizations have provided direct health services to community residents, advocated for improved care as well as better access to care, and worked steadily toward improving environmental conditions in these communities.
In collaboration with its CAB, the center developed a community education campaign called "Healthy Home Healthy Child." The campaign works to increase local residents' awareness of environmental health threats and preventive techniques to reduce pollution exposure to themselves, their families, and, in particular, their children. Focus groups were established with residents of Washington Heights, Harlem, and the South Bronx to help identify environmental issues that were of special concern to these communities. Additionally, 555 young mothers were surveyed in public places to gauge awareness of environmental hazards. The center's scientists combined their knowledge of current research findings with community concerns to target seven environmental hazards--air pollution, cigarette smoke, nutrition, pesticides, lead poisoning, drugs and alcohol, and garbage management--in the "Healthy Home Healthy Child" campaign.
Written materials on each topic were developed in collaboration with WE ACT to ensure that the literature incorporated local cultural values and were at an appropriate reading level for the community. The materials aim to educate community parents about sources of environmental hazards, their health consequences, and how to take steps to prevent or at least diminish everyone's exposure to toxic pollutants. The literature is distributed at health fairs, various community events, and through the 10 CAB organizations' regular activities. A WE ACT health educator delivers presentations to parents at day care centers, distributing campaign materials throughout Washington Heights and West Harlem. The CCCEH is currently collaborating with the full CAB to train health educators on staff at each organization to deliver presentations on the seven environmental health topics. The effort will expand the reach of the campaign into more neighborhoods, including those in the South Bronx, and improve its sustainability.
The center and CAB have also hosted community events including an environmental health fair attended by over 300 study cohort mothers and children as well as local residents, and a large national conference called "The Health of Our Children in the Urban Environment: A Dialogue among Scientists, Community Leaders and Policymakers" held 27 March 2000. At this conference, David Satcher, U.S. Surgeon General, and Kenneth Olden (National Institute of Environmental Health Sciences, Research Triangle Park, NC) addressed an audience of 500 local residents and professionals in environmental health-related fields about the need for exactly the kind of research combined with community outreach that the center is conducting. CAB members participated in the panel discussions addressing the intersection of scientific research with community health and public policy.
The CAB is invaluable for its counsel and is a necessary stepping stone toward building a larger communication network within local neighborhoods and changing perceptions that have previously inhibited a better and more substantial working relationship between community members and scientific researchers. The center's partnership with its CAB helps to dissolve some of the barriers to better health care and at-home prevention practices by opening lines of communication with community leaders who may have had no former relationship with the university. With that contact, multiple opportunities arise for education of local residents who have established relationships with these community-based organizations on which they depend for health services and advocacy. Ultimately, a larger segment of the community is reached in the dissemination of important health findings and preventive methods for reducing risk.
The health of a society can be judged by the health of its children. As a society we can and must do better in preventing harm to this vulnerable group. This will entail the early identification of preventable risks and the prompt translation of this knowledge into protective policies and interventions.
Preventive steps such as banning the clearly neurotoxic pesticide chlorpyrifos for home use are now being taken to protect fetuses and young children. However, we need to better understand the interactions between multiple exposures and susceptibility factors that may disproportionately affect children, particularly those in certain social and ethnic groups, putting them at greater risk from toxic pollutants.
The CCCEH is working to understand the health risks from early life exposures to environmental contaminants in combination with susceptibility factors and is attempting to address these threats through education, policy-relevant research, and the timely dissemination of research results. We hope that this work and that of the other national centers for children's environmental health will help improve the environments in which children grow so they may reach their full potential in life.
|
|
 |
[References Listed in PubMed]
References and Notes
1. Perera FP. Molecular epidemiology: insights into cancer susceptibility, risk, assessment, and prevention. J Natl Cancer Inst 88:496-509 (1996).
2. Whyatt RM, Perera FP. Application of biologic markers to studies of environmental risks in children and the developing fetus. Environ Health Perspect 103(suppl 6):105-110 (1995).
3. Calabrese EJ. Age and Susceptibility to Toxic Substances. New York:John Wiley and Sons, 1986.
4. WHO. Principles for Evaluating Health Risks from Chemicals during Infancy and Early Childhood: The Need for a Special Approach. Environmental Health Criteria 59. Geneva:World Health Organization, 1986.
5. Mott L, Vance F, Curtis J. Handle with Care: Children and Environmental Carcinogens. New York:Natural Resources Defense Council, 1994.
6. Needleman HL. Lead levels and children's psychologic performance [Letter]. N Engl J Med 301:163 (1979).
7. Shimizu Y, Kato H, Schull WJ. Risk of cancer among atomic bomb survivors. J Radiat Res 32 (Suppl 2):54-63 (1991).
8. Jacobson JL, Jacobson SW. Intellectual impairment in children exposed to polychlorinated biphenyls in utero. N Engl J Med 335:783-789 (1996).
9. Perera FP, Whyatt RM, Jedrychowski W, Rauh V, Manchester D, Santella RM, Ottman R. Recent developments in molecular epidemiology: a study of the effects of environmental polycyclic aromatic hydrocarbons on birth outcomes in Poland. Am J Epidemiol 147:309-314 (1998).
10. Srivastava VK, Chauhan SS, Srivastava PK, Kumar V, Misra UK. Fetal translocation and metabolism of PAH obtained from coal fly ash given intratracheally to pregnant rats. J Toxicol Environ Health 18:459-469 (1986).
11. Neubert D, Tapken S. Transfer of benzo(a)pyrene into mouse embryos and fetuses. Arch Toxicol 35:2943-2953 (1988).
12. Lu LJW, Disher RM, Reddy MV, Randerath K. 32P-Postlabeling assay in mice of transplacental DNA damage induced by the environmental carcinogens safrole, 4-aminobiphenyl and benzo(a)pyrene. Cancer Res 46:3046-3054 (1986).
13. Lu LJW, Wang MY. Modulation of benzo(a)pyrene-induced covalent DNA modification in adult and fetal mouse tissues by gestation stage. Carcinogenesis 11:1367-1372 (1990).
14. Whyatt RM, Jedrychowski W, Hemminki K, Santella RM, Tsai WY, Yang K, Perera FP. Biomarkers of polycyclic aromatic hydrocarbon-DNA damage and cigarette smoke exposures in paired maternal and newborn blood samples as a measure of differential susceptibility. Cancer Epidemiol Biomarkers Prev 10:581-588 (2001).
15. National Research Council. Pesticides in the Diets of Infants and Children. Washington, DC:National Academy Press, 1993.
16. Laib RJ, Klein KP, Bolt HM. The rat liver foci bioassay: age dependence of induction by vinyl chloride of ATP-deficient foci. Carcinogenesis 6:65-68 (1985).
17. Plopper CG, Fanucci MV. Do urban environmental pollutants exacerbate childhood lung diseases? Environ Health Perspect 108:A252-A253 (2000).
18. Miller RL, Biedermann SA, Bell CA, Halasz G, Jerzynska J, Whyatt RM, Perera FP, Ford JG. Cord blood proliferation and cytokine production in response to indoor allergens in an inner-city cohort. Am J Respir Crit Care Med (in press).
19. U.S. EPA. Environmental Health Threats to Children. Washington, DC:U.S. Environmental Protection Agency, 1996.
20. Faustman EM. Mechanisms underlying children's susceptibility to environmental toxicants. Environ Health Perspect 108(suppl 1):13-21 (2000).
21. Chanda SM, Pope CN. Neurochemical and neurobehavioral effects of repeated gestational exposure to chlorpyrifos in maternal and developing rats. Pharmacol Biochem Behav 53:771-776 (1996).
22. Campbell CG, Seidler FJ, Slotkin TA. Chlorpyrifos interferes with cell development in rat brain regions. Brain Res Bull 43(2):179-189 (1997).
23. Hatch GE. Asthma, inhaled oxidants, and dietary antioxidants. Am J Clin Nutr 61(suppl):625S-630S (1995).
24. Greene LS. Asthma and oxidant stress: nutritional, environmental, and genetic risk factors. J Am Coll Nutr 14:317-324 (1995).
25. Peat JK, Briton WJ, Salome CM, Woolcock AJ. Bronchial hyperresponsiveness in two populations of Australian school children. III. Effect of exposure to environmental allergens. Clin Allergy 17:297-300 (1987).
26. Wang X, Ding H, Ryan L, Xu X. Association between air pollution and low birth weight: a community-based study. Environ Health Perspect 105:514-520 (1997).
27. Crawford MA, Doyle W, Drury P, Lennon A, Costeloe K, Leighfield M. n-6 and n-3 fatty acids during early human development. J Int Med Suppl 225(731):159-169 (1989).
28. Voigt RG, Jensen CL, Rozelle JC, Heird WC, Brown FR. Effect of alpha-linoleic acid intake in early infancy on neurodevelopmental outcome of term infants at 12 and 15 months of age [Abstract]. Pediatr Res 39:1915 (1996).
29. Makrides M, Neumann M, Simmer K, Paler J, Gibson R. Are long-chain polyunsaturated fatty acids essential nutrients in infancy? Lancet 345:1463-1468 (1995).
30. Greater Boston Physicians for Social Responsibility. In Harm's Way: Toxic Threats to Child Development. Cambridge, MA:Greater Boston Physicians for Social Responsibility, 2000.
31. Cassel J. The contribution of the social environment to host resistance: the Fourth Wade Hampton Frost Lecture. Am J Epidemiol 104:107-123 (1976).
32. Zapata BC, Rebolledo A, Atalah E, Newman B, King MC. The influence of social and political violence on the risk of pregnancy complications. Am J Public Health 82:685-690 (1992).
33. Kliegman R. Perpetual poverty: child health and the underclass. Pediatrics 89:710-713 (1992).
34. David RJ, Collins JW. Differing birth weight among infants of US-born blacks, African-born blacks, and US-born whites. N Engl J Med 337:1209-1214 (1997).
35. Wise PH. Confronting racial disparities in infant mortality: reconciling science and politics. Am J Prev Med 31:7-16 (1993).
36. Rauh V, Andrews HA, Garfinkel RS. The contribution of maternal age to racial disparities in birthweight: a multi-level perspective. Am J Public Health 91(11):1815-1824 (2001).
37. Krasner M, Heisler T, Brooks P, Billings J, Newman L, Jacobson R, Schulz A, Andrews H, Pittman J. New York City Community Health Atlas. New York:United Hospital Fund, 1994.
38. Verbeck D, Gibson C. Keeping Track of New York City's Children. A Citizens' Committee for Children Status Report. New York:Citizens' Committee for Children, Inc., 1997.
39. Centers for Disease Control. Surveillance for Asthma, United States: 1960-1995. MMWR, 47(SS1):1-28 (1998).
40. Peques M. Personal communication.
41. Crain EF, Weiss KB, Bijur PE, Hersh M, Westbrook L, Stein RE. An estimate of the prevalence of asthma and wheezing among inner-city children. Pediatrics 94(3):356-362 (1994).
42. Claudio L, Tulton L, Doucette J, Landrigan PJ. Socioeconomic factors and asthma hospitalization rates in New York City. J Asthma 36(4):343-350 (1999).
43. Weiss B, Landrigan PJ. The developing brain and the environment: an introduction. Environ Health Persepct 108:373-374 (2000).
44. May M. Disturbing behavior: neurotoxic effects in children. Environ Health Perspect 108:262-267 (2000).
45. INFOSHARE. New York:Community Studies of New York, Inc.
46. U.S. Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation. Summary Historical Figures and Federal Register References for the HHS Poverty Guidelines since 1982. Available: http://aspe.hhs.gov/poverty/ figures-fed-reg.htm [cited 9 January 2002].
47. U.S. Environmental Protection Agency Office of Air Quality Planning and Standards. National Ambient Air Quality Standards. Available: http://www.epa.gov/airs/ criteria.html [cited 9 January 2002].
48. McClellan RO. Health effects of exposures to diesel exhaust particles. Annu Rev Pharmacol Toxicol 27:279-300 (1987).
49. South Bronx Clean Air Coalition. Personal communication.
50. NYPIRG. Dirty Power on the Rise in New York: Report by the American Lung Association of New York, Environmental Advocates, and the New York Public Interest Research Group. Albany, NY:NY Public Interest Research Group, 1999.
51. Kinney PL, Aggarwal M, Northridge ME, Jannsen NAH, Shepard P. Airborne concentrations of PM2.5 and diesel exhaust particles on Harlem sidewalks: a community-based pilot study. Environ Health Perspect 108:213-218 (2000).
52. Texas Natural Resource Conservation Commision. The National Ambient Air Quality Standards. Available: www.tnrcc.state.tx.us/air/monops/naaqs.html [cited 9 January 2002].
53. Pirkle JL, Flegal KM, Bernert JT, Brody DJ, Etzel RA, Maurer KR. Exposure of the US population to environmental tobacco smoke: the Third National Health and Nutrition Examination Survey, 1988 to 1991. JAMA 275:1233-1240 (1996).
54. Chen LH, Petitti DB. Case-control study of passive smoking and the risk of small-for-gestational-age at term. Am J Epidemiol 142(2):158-165 (1995).
55. Wagenknecht LE, Manolio TA, Sidney S, Burke GL, Haley NJ. Environmental tobacco smoke exposure as determined by cotinine in black and white young adults: the CARDIA study. Environ Res 63:39-46 (1993).
56. Wagenknecht LE, Burke GL, Perkins LL, Haley NJ, Friedman GD. Misclassification of smoking status in the CARDIA study: a comparison of self-report with serum cotinine levels. Am J Public Health 82:33-36 (1992).
57. Hecht SS, Carmella SG, Foiles PG, Murphy SE. Biomarkers for human uptake and metabolic activation of tobacco-specific nitrosamines. Cancer Res 54:1912s-1917s (1994).
58. Knight JM, Eliopoulos C, Klein J, Greenwald M, Koren G. Passive smoking in children. Racial differences in systemic exposure to cotinine by hair and urine analysis. Chest 109:446-450 (1996).
59. English PB, Eskenazi B, Christianson RE. Black-white differences in serum cotinine levels among pregnant women and subsequent effects on infant birthweight. Am J Public Health 84:1439-1443 (1994).
60. Goldstein IF, Reed CE, Swanson MC, Jacobson JS. Aeroallergens in New York inner-city apartments of asthmatics. Experientia Suppl 51:133-138 (1987).
61. Sarpong SB, Hamilton RG, Eggleston PA, Adkinson FN. Socioeconomic status and race as risk factors for cockroach allergen exposure and sensitization in children with asthma. J Allergy Clin Immunol 97:1394-1401 (1996).
62. Beasley R, Crane J, Lai CK, Pearce N. Prevalence and etiology of asthma. J Allergy Clin Immunol 105(2 Pt 2):S466-472 (2000).
63. Duran-Tauleria E, Rona RJ. Geographical and socioeconomic variation in the prevalence of asthma symptoms in English and Scottish children. Thorax 54(6):476-481 (1999).
64. Sporik R, Holgate ST, Platts-Mills TA, Cogswell JJ. Exposure to house-dust mite allergen (Der p I) and the development of asthma in childhood. A prospective study. N Engl J Med 323(8):502-507 (1990).
65. Sporik R, Holgate ST, Cogswell JJ. Natural history of asthma in childhood--a birth cohort study. Arch Dis Child 66(9):1050-1053 (1991).
66. Martinez FD, Wright AL, Taussig LM, Holberg CJ, Halonen M, Morgan WJ. Asthma and wheezing in the first six years of life. The Group Health Medical Associates. N Engl J Med 332(3):133-138 (1995).
67. DHHS. The Health Benefits of Smoking Cessation: A Report of the Surgeon General. Rockville, MD:U.S. Department of Health and Human Services, 1990.
68. Bui QQ, Tran MB, West WL. A comparative study of the reproductive effects of methadone and benzo(a)pyrene in the pregnant and pseudopregnant rat. Toxicology 42:195-204 (1986).
69. New York City Health Department Bulletin. New York:New York City Health Department, 1998.
70 American Cancer Society. Cancer Facts and Figures 1996. Atlanta, GA:American Cancer Society, 1996.
71. DHHS. Report of the Secretary's Task Force on Black and Minority Health. Washington, DC:U.S. Department of Health and Human Services, 1986.
72. Day NE, Brown CC. Multistage models and primary prevention of cancer. J Natl Cancer Inst 64:977-989 (1980).
73. Fullilove MT. Response: environments as toxins. Bull NY Acad Med 67(6):571-573 (1991).
74. Whyatt RM, Camann DE, Kinney PL, Reyes A, Ramirez J, Dietrich J, Diaz D, Holmes D, Perera FP. Residential pesticide use during pregnancy among a cohort of urban minority women. Environ Health Perspect (in press).
75. Binkova B, Lewtas J, Miskova I, Lenicek J, Sram R. DNA adducts and personal air monitoring of carcinogenic polycyclic aromatic hydrocarbons in an environmentally exposed population. Carcinogenesis 16(5):1037-1046 (1995).
76. Whitemore RW, Immerman FW, Camann DE, Bond AE, Lewis RG, Schaum JL. Non-occupational exposures to pesticides for residents of two U.S. cities. Arch Environ Contam Toxicol 26:47-59 (1994).
77. Whyatt RM, Barr DB. Measurement of organophosphate metabolites in postpartum meconium as a potential biomarker of prenatal exposure: a validation study. Environ Health Perspect 109:417-420 (2001).
78. Platts-Mills TAE. How environment affects patients with allergic disease: indoor allergens and asthma. Ann Allergy 72:381-384 (1994).
79. Miller RL, Chew G, Bell CA, Biedermann SA, Aggarwal M, Kinney PL, Tsai WY, Whyatt RM, Perera FP, Ford JG. Prenatal exposure, maternal sensitization, and sensitization in utero to indoor allergens in an inner-city cohort. Am J Respir Crit Care Med 164:995-1001 (2001).
80. Meyer I. Unpublished data.
81. Crain EF, Weiss KB, Bijur PE, Hersh M, Westbrook L, Stein RE. An estimate of the prevalence of asthma and wheezing among inner-city children. Pediatrics 94(3):356-362 (1994).
82. Gelber LE, Seltzer LH, Bouzoukis JK, Pollart SM, Chapman MD, Platts-Mills TA. Sensitization and exposure to indoor allergens as risk factors for asthma among patients presenting to hospital. Am Rev Respir Dis 147(3):573-578 (1993).
83. Platts-Mills TAE, Thomas W, Aalberse RC, Vervloet D, Champmon MD. Dust mite allergens and asthma: report of a second international workshop. J Allergy Clin Immunol 89(5):1046-1060 (1992).
84. Perera FP. Molecular epidemiology: on the path to prevention? J Natl Cancer Inst 92:602-612 (2000).
85. Susser M, Susser E. Choosing a future for epidemiology: II. From black box to Chinese boxes and eco-epidemiology. Am J Public Health 86:674-677 (1996).
86. Perera FP, Weinstein IB. Molecular epidemiology and carcinogen-DNA adduct detection: new approaches to studies of human cancer causation. J Chronic Dis 35:58l-600 (1982).
87. Schulte PA, Perera FP. Molecular Epidemiology: Principles and Practices. New York:Academic Press, 1993.
88. Perera FP. Molecular cancer epidemiology: a new tool in cancer prevention. J Natl Cancer Inst 78:887-898 (1987).
89. Hulka BS. Epidemiological studies using biological markers: issues for epidemiologists. Cancer Epidemiol Biomark Prev 1:13-19 (1991).
90. Hulka BS. Using biomarkers: views from an epidemiologist. Health Environ Digest 5:1-7 (1991).
91. Omenn GS. Future research directions in cancer ecogenetics. Mutat Res 247:283-291 (1991).
92. Groopman JD, Wogan GN, Roebuck BD, Kensler TW. Molecular biomarkers for aflatoxins and their application to human cancer prevention. Cancer Res 54:1907s-1911s (1994).
93. Harris C. Chemical and physical carcinogenesis: advances and perspectives for the 1990s. Cancer Res (suppl) 51:5023S-5044S (1991).
Last Updated: January 17, 2002 |
|
 |
|
| |

