Fast Fetal Absorption of a Common Compound
Most maternally administered chemicals have the potential to cross the placental barrier, and the question is not whether a chemical crosses the placenta, but the rate at which it does so. In this issue, Osamu Takahashi and Shinshi Oishi of the Tokyo Metropolitan Research Laboratory of Public Health present their findings that the placenta fails to act as a barrier to a potentially toxic compound, 2,2-bis(4-hydroxyphenyl)propane, also known as bisphenol A or BPA [EHP 108:931-935]. The researchers found that BPA can be transferred across the placental barriers of female Fischer 344 rats to their fetuses. They also discovered that BPA is absorbed and distributed in maternal internal organs and fetuses extremely rapidly.
|
|
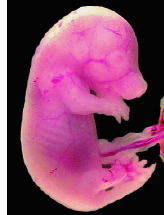
Rapid dam-fetus transfer. It took only 20 minutes for maternally ingested BPA to reach maximum concentration in fetal Fischer 344 rats.
Photo credit: Greenwell/EHP |
BPA has been widely used in plastics manufacture and as a fungicide, antioxidant, flame retardant, rubber chemical, and polyvinyl chloride stabilizer. The estimated production of BPA in Japan in 1995 was about 260,000 tons. Release and migration of BPA from various resins and plastic products into the environment have been recognized; small amounts of the compound are detectable in river and tap water.
BPA has not been found to be carcinogenic or teratogenic. At doses of 5.0-6.3 grams per kilogram per day (g/kg/day), it is nephrotoxic in mice. Doses of 400-1,000 milligrams (mg)/kg/day of BPA have produced estrogenic activity in immature rats or female mice whose ovaries have been surgically removed, and doses at 437-1,750 mg/kg/day have adversely affected the reproductive system and sperm production in male mice. However, although lower doses (2-400 micrograms [µg]/kg/day) of BPA administered into the uterus through the mother's circulation have been reported to affect reproductive organs in male offspring of mice and rats, scientists have to date been unable to reproduce these results, so such toxicity remains controversial.
What impressed Takahashi and Oishi was the speed with which BPA was transferred from the pregnant rat dam to her fetuses. On day 18 of gestation, they administered 1 g/kg BPA dissolved in propylene glycol at 25% weight per volume. This dose is about one-fourth the median lethal dose for rats. It took only 20 minutes for the BPA to attain its maximum concentrations in maternal blood, liver, and kidneys (these concentrations were 14.7, 171.0, and 36.2 µg/g, respectively). Likewise, in 20 minutes, BPA concentration in the fetuses reached its maximum at 9.22 µg/g. Six hours after administration of BPA, its concentrations in the maternal blood and organs and in the fetuses were reduced to 2-5% of maximum. In the view of the authors, the attainment of maximum concentrations of BPA in the mother rat and her fetuses suggests that BPA's absorption and distribution are extremely rapid and that the placenta does not act as a barrier to BPA.
One factor that determines whether a compound is absorbed by the fetus are its lipophilic or hydrophilic properties--that is, whether it dissolves better in a lipid or in water. Compounds with a high octanol/water partition coefficient (Pow)--compounds that are more lipophilic--cross slowly; examples are pentachlorophenol and DDT, whose log Pow is 5.0-6.0 (such compounds, nevertheless, can be accumulated). By contrast, chemicals with a lower log Pow, such as diethylstilbestrol and salicylic acid, are more hydrophilic and they more easily cross the placenta; their log Pow may vary between -0.9 and 5.0. (The log Pow of BPA, which crosses the placenta so rapidly, is 3.3.) Takahashi and Oishi suggest that the speed and degree of transplacental absorption may be mediated by the lipophilic or hydrophilic properties of a chemical.
In the case of BPA, because the placenta essentially presents no barrier, the compound was rapidly passed through and could show toxicologic effects; yet in this study it largely cleared after several hours. However, the BPA dose of 1 g/kg was quite high. When a dose of 0.1 g/kg BPA was administered, its concentrations in the mother and fetuses could be determined accurately only near peak levels. -Julian Josephson
Vulnerability to Lead
Absorption Varies with Genotype
Genetic factors may modify the toxicokinetics of lead in humans--that is, how lead is absorbed, distributed, metabolized, and excreted. Attention has centered on the enzyme 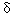 -aminolevulinic dehydratase (ALAD), which is coded by a single gene with two alleles, ALAD1 and ALAD2. In this month's issue, Brian S. Schwartz of the Johns Hopkins School of Hygiene and Public Health and colleagues present their study on the influence of polymorphisms in the ALAD and vitamin D receptor (VDR) genes on concentrations of lead in blood and the tibia, and the amount of lead chelatable with dimercaptosuccinic acid (DMSA) and excreted in the urine [EHP 108:949-954]. Their results confirm past observations that the ALAD gene modifies lead toxicokinetics and furnish new evidence that the VDR gene does so as well.
-aminolevulinic dehydratase (ALAD), which is coded by a single gene with two alleles, ALAD1 and ALAD2. In this month's issue, Brian S. Schwartz of the Johns Hopkins School of Hygiene and Public Health and colleagues present their study on the influence of polymorphisms in the ALAD and vitamin D receptor (VDR) genes on concentrations of lead in blood and the tibia, and the amount of lead chelatable with dimercaptosuccinic acid (DMSA) and excreted in the urine [EHP 108:949-954]. Their results confirm past observations that the ALAD gene modifies lead toxicokinetics and furnish new evidence that the VDR gene does so as well.
Earlier research established that ALAD is a principal lead-binding protein in red blood cells, and recent data suggest that polymorphisms in the VDR gene influence tibia lead concentrations. In particular, the polymorphism denoted as BsmI has three genotypes, designated as bb, Bb, and BB, that appear to be linked with lead concentrations in bone.
In this study, 798 Korean lead workers and 135 controls without occupational lead exposure provided blood samples, and blood lead was assessed with an atomic absorption spectrometer. Tibia lead was evaluated using 109Cd-induced X-ray fluorescence, and chelatable lead was measured as a 4-hour urinary lead excretion after the study subjects were orally dosed with DMSA. The scientists also completed ALAD and VDR genotyping on the subjects. Whether the workers used tobacco or alcohol was taken into account during statistical analysis.
Adjusted analyses showed that subjects with the VDR B allele had significantly higher blood and tibia lead concentrations and greater excretion of lead after DMSA dosing than did those with the VDR bb genotype. The effect of the VDR B allele on blood lead concentrations turned out to be greater than that of the ALAD2 allele. However, the mechanisms by which these genes influence blood lead concentrations probably differ, because ALAD2 and VDR B were both associated with higher blood lead concentrations, but only VDR B was associated with higher tibia lead concentrations and greater lead excretion after DMSA dosing.
After binding to the VDR, vitamin D is known to increase intestinal absorption of lead. It is possible that the influence of vitamin D on lead absorption from the gut differs by genotype, and that people with the VDR B allele have greater absorption of lead via the intestines and greater uptake and subsequent release of lead from bone. The scientists note, however, that tibia lead measurements by X-ray fluorescence were complicated by the fact that such analyses are standardized to bone mineral content. Thus, higher tibia lead readings could be attributed to higher lead content, lower calcium content, or both.
Subjects with the ALAD2 allele showed higher blood lead concentrations but no differences in tibia or chelatable lead concentrations compared with subjects lacking this allele. The results reinforce observations that the ALAD2 allele increases red blood cell binding of lead, and so probably decreases the relative deposition of lead in critical target organs, possibly protecting against the toxicity of lead by increasing the amount of lead excreted in urine.
All studies to date suggest that the ALAD1 allele is more likely to confer health risks from lead exposure. The researchers also found that lead workers homozygous for the ALAD1 allele were much less likely to have the VDR bb genotype; the two genes are apparently linked despite the fact that they are located on different chromosomes. Although the VDR gene may play a role in susceptibility to the health effects of lead, there are not enough data to indicate whether its polymorphisms will modify health risks, and if so, which allele brings about such risks. Compared with controls, lead workers seem to have a higher prevalence of ALAD2 and VDR B. It may be that the ALAD2 and VDR B alleles are protective, and there might be selection by genotype among lead workers--perhaps because workers who become symptomatic upon exposure to lead would choose to leave the occupation--but this speculation requires further study. -Julian Josephson
A New View of ELF-EMFs
Are They Linked with Cancer Promotion?
The debate over a possible link between cancer and extremely low frequency electromagnetic fields (ELF-EMFs) began with a 1979 study that found excess cancer in people who lived near large electrical wires. It has continued through subsequent in vitro, in vivo, and epidemiological studies that often produced conflicting results. In this issue, Gang Chen of the Department of Pediatrics and Human Development at Michigan State University and colleagues used an experimental model developed to test cancer-promoting chemicals to examine whether ELF-EMFs might play a role in cancer promotion [EHP 108:967-972].
The development of cancer is a multistage process. During normal development, immature cells undergo a process called differentiation in which they become highly specialized (developing, for instance, into red blood cells) and are less able to continue proliferating. In the first stage of cancer, initiation, a cell's DNA is damaged through mutation, causing a differentiated cell to resemble an immature one, in effect reversing the process of differentiation. In the second stage, promotion, normal cellular controls go awry, and the mutated cell multiplies. ELF-EMFs are too weak to kill cells or (most scientists agree) to cause mutations and thus initiate cancer. However, they could play a role during the promotion stage of cancer, which involves so-called epigenetic mechanisms (those that affect gene expression rather than gene structure) and induce cancer in cells that have already mutated.
In the laboratory, differentiation--which can be stimulated by chemical treatment--can transform initiated cells into mature cells, converting cells that had started to become cancerous into normal-seeming adult cells. In this case, differentiation seems to be a healing process that nullifies the mutation. What the group was testing was whether ELF-EMFs could prevent differentiation in cells that had started down the road to cancer.

A different role for electromagnetic fields? A 1979 study found that children who lived near power lines (and consequently had higher ELF-EMF exposures) had a higher incidence of cancer. Although most scientists believe ELF-EMFs are too weak to initiate cancer, new research suggests they could play a role as cancer promoters.
The research used mouse leukemia cells that, when treated with dimethyl sulfoxide (DMSO), differentiate into red blood cells. The researchers group compared control cells, cells treated with DMSO, and cells treated with DMSO and maintained inside a culture chamber exposed to a 60-hertz ELF-EMF at varying strengths. In a system used to investigate the epigenetics of cancer promotion, the scientists measured three end points. Proliferation, or cell growth, was determined by measuring DNA concentration. Differentiation was measured by detecting hemoglobin, a sign that the cell had developed into a red blood cell. Youthfulness was gauged by measuring telomerase, the enzyme that builds telomeres, which keep chromosomes "young" and able to divide.
Starting at a threshold dose of about 20 milligauss (a measure of the strength of the electrical field), the 60-hertz ELF-EMF caused a dose-dependent reduction of differentiation, as well as an increase in telomerase and proliferation. These effects resemble those of chemical cancer promoters. (Under a power line, fields measure roughly 300 milligauss, and near home appliances they can exceed 1 gauss.)
While the study showed that ELF-EMFs could conceivably play a biological role in carcinogenesis, cancer-promoting chemicals require a long exposure to promote cancer, and human exposures to ELF-EMFs are hard to gauge. Because electric fields change so radically from point to point, it's too early to say if typical exposures actually promote cancer. But by stressing the importance of promotion, the study could focus future research on the environmental health effects of ELF-EMFs. -David J. Tenenbaum
Last Updated: October 18, 2000





