Binding May Influence Effects
Estrogen is a term used for a variety of natural and synthetic hormones that influence the growth and function of many tissues, particularly those of the male and female reproductive systems. Estrogen plays a role in several cancers, especially breast and endometrial cancers and possibly prostate and colon cancers. The human diet contains estrogenic compounds that can bind to estrogen receptors in the body, and much research has focused on whether dietary estrogens can influence the development of cancer.
Phytoestrogens--estrogen-like compounds produced by plants--have received attention owing to epidemiologic and experimental evidence that they prevent cancer, guard against bone loss, and reduce menopausal symptoms. Mycoestrogens (mold-produced estrogens) have have deleterious effects on livestock health and reproduction.
In this month's issue, Georgi N. Nikov of Tulane University in New Orleans, Louisiana, and colleagues examine the activity of dietary estrogens in humans [EHP 108:
867-872]. They confirm that dietary estrogens have different affinities for human estrogen receptors. They also found that, once bound, each estrogen can also alter a receptor's shape and size.



Dietary factor. Soy estrogens such as daidzein (left) and genistein (middle) can displace estradiol (right) from estrogen receptors in the body.
Photo credit: R. Rajnarayanan, Tulane University
When estrogen binds to a receptor, the resulting complex interacts with a site within a target gene's regulatory region. That interaction may either promote or inhibit gene transcription. Alterations in the receptor's shape may affect how well the estrogen receptor complex can initiate or inhibit gene transcription.
Nikov and his colleagues focused on four phytoestrogens--genistein, coumestrol, daidzein, and glyceollin--and the mycoestrogen zearalenone. Genistein and daidzein are normal components of soybeans, which also produce glyceollin when subjected to certain stresses. Coumestrol is produced by clover, and zearalenone is generated by Fusarium molds, which infect grains.
The team first measured the estrogens' affinities for two estrogen receptors, ER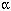 and ERß, as compared to estradiol, the form of estrogen normally found in the body. But knowing that an estrogen binds to a receptor is only half the story; what follows is perhaps even more important. Therefore, the researchers also investigated how receptor complexes interact with estrogen response element (ERE) sequences, regulatory sites that turn genes off or on.
and ERß, as compared to estradiol, the form of estrogen normally found in the body. But knowing that an estrogen binds to a receptor is only half the story; what follows is perhaps even more important. Therefore, the researchers also investigated how receptor complexes interact with estrogen response element (ERE) sequences, regulatory sites that turn genes off or on.
The researchers prepared dilutions of each estrogen and used fluorescence polarization, a relatively new approach, to study estrogen binding. This method employs fluorescent labeling of key molecules to reveal shifts in molecular size and shape; such alterations expose binding, dissociation, and conformational changes. Each estrogen's ability to displace fluorescently labeled estradiol from ER and ERß was determined. This information revealed the affinity with which the plant estrogens bind to the receptors; it also permitted calculation of how much of each is needed to saturate a known amount of receptors.
and ERß was determined. This information revealed the affinity with which the plant estrogens bind to the receptors; it also permitted calculation of how much of each is needed to saturate a known amount of receptors.
Following these affinity calculations, the team investigated the interactions of receptor complexes with ERE sequences. To this end, Nikov and his team conducted parallel experiments using two types of fluorescently labeled ERE sequences, one from the Xenopus vitellogenin (vit) A2 gene, the other from the human pS2 gene.
The researchers found that, except for glyceollin, all of the tested estrogens had a greater affinity for ERß than ER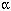 ; glyceollin had the opposite preference. However, the glyceollin-receptor complexes, unlike the other complexes, did not interact with either ERE sequence. The interactions that did occur varied according to the specific estrogen and which receptor and ERE sequence were involved; however, none were stronger than those involving estradiol.
; glyceollin had the opposite preference. However, the glyceollin-receptor complexes, unlike the other complexes, did not interact with either ERE sequence. The interactions that did occur varied according to the specific estrogen and which receptor and ERE sequence were involved; however, none were stronger than those involving estradiol.
The researchers concluded that just knowing the affinity of the estrogen for the receptor is not enough. Equally important is the way the estrogen-receptor complexes interact with ERE sequences. These interactions may in turn affect transcription of target genes and thus affect the myriad functions of estrogen in the body. -Julia R. Barrett
More Evidence for Learning Effects
For over a decade, in utero and postnatal exposure to polychlorinated biphenyls (PCBs) has been linked with reduced IQ in children, but exactly how the chemicals exert their effects on cognitive functioning remains uncertain. In this issue, Rifat J. Hussain and colleagues from the School of Public Health at the State University of New York at Albany explore PCB-induced changes in electrical activity in the hippocampus, an area of the brain involved in learning and memory [EHP 108:827-831]. Their specific goal is to assess PCBs' effects on long-term potentiation (LTP), a prolonged increase in synaptic responses that is believed to be essential for learning. The motivation for the study rests in part on previous observations that childhood lead exposure and old age, both associated with reduced cognitive functioning, are also associated with reduced LTP.
In the present study, Sprague-Dawley rat pups were exposed to PCB 153, a ubiquitous congener in human tissue and environmental media, by maternal dosing in feed during gestational day 7 through postnatal day 21. Dams were exposed at four dose levels: 0.00, 1.25, 5.00, and 20.00 milligrams per kilogram per day. One male pup from each litter was selected for study, and five animals in total (including one nonexposed control) were selected per dose group.
At the end of the exposure period, the pups' brains were removed and slices of hippocampus prepared. Evaluation of LTP was performed on brain slices from the pups exposed in vivo as well as a separate group of brain slices taken from 30-day-old male pups that received no prior in vivo exposure. These brain slices were perfused in vitro with a solution saturated with PCB 153.
Hussain and colleagues monitored the amplitude of the field excitatory postsynaptic potential (fEPSP), which reflects the intensity of the response of hippocampal neurons to stimulation. Once a stable baseline measure of electrical synaptic activity was obtained, LTP was induced by applying two separate stimulations of 100 hertz for 1 second, 5 seconds apart. LTP was measured as the increase in the amplitude of the fEPSP 60 minutes after this high-frequency stimulation.
Hussain and colleagues found that, while in vivo PCB exposure does not produce any obvious baseline changes in synaptic response prior to stimulation, it reduces the increase in fEPSP after stimulation in a dose-dependent fashion. The results of in vitro exposures are roughly similar: no obvious changes in the prestimulation period, but a degree of poststimulation LTP suppression similar to that observed in the in vivo portion of the study. This somewhat surprising observation that in vivo and in vitro exposures to PCB 153 produce similar effects may indicate that the mechanism of action of PCBs on LTP is more pharmacologic than developmental, but additional study of this observation is necessary.
While Hussain and colleagues acknowledge that PCB-induced changes in behavioral and cognitive function are complex and that the relationship between IQ and LTP is still not proven, their results are significant for at least two reasons. Reduction of LTP is potentially involved in neurobehavioral effects, and this observation provides a model system for further study of the mechanisms of PCB action. Furthermore, PCB 153, a nondioxin-like congener that is one of the most abundant PCBs in humans, has often been considered to be relatively nontoxic. These results show that that is not the case, at least for neurobehavioral effects. -Charles W. Schmidt
Measuring PAHs in Airplane Emissions
In the March 2000 issue of EHP, Joachim D. Pleil and colleagues reported the results of their study of Air Force personnel exposed to JP-8 jet fuel in the form of fuel vapors and exhaust fumes [EHP 108:183-192]. This month, Jeffrey W. Childers of ManTech Environmental Technology in Research Triangle Park, North Carolina, and colleagues (including Pleil) detail their investigation into human exposure to potentially carcinogenic compounds contained in aircraft exhaust and evaluate the use of various instruments for measuring PAH emissions [EHP 108:853-862].
Their study focused on the presence in aircraft exhaust of polycyclic aromatic hydrocarbons (PAHs). Several of these compounds have been listed by the National Toxicology Program as reasonably anticipated to be a human carcinogen, particularly those PAHs associated with the particulate phase, once they've bound to respirable particles. So an accurate measurement of the presence of particle-bound PAHs is critical to assessing the risk of exposure to personnel involved in aircraft support, maintenance, and usage. To be useful in realistically assessing both health risks and the potential need for workers to use respiratory protective equipment during periods of acute exposure, the measurement method must provide data over time, because exhaust levels rise and fall sharply during various activities associated with the aircraft.

|
Flight test. Studies of PAH concentrations in aircraft exhaust provide critical information about points during flight-related and ground-support activities--such as engine start-up (above)--when workers may face acute exposures to the possibly carcinogenic compounds.
Photo credit: Joachim Pleil, U.S. EPA
|
In this study, the authors evaluated methodologies for quantifying PAH concentrations in exhaust. The equipment to do so reliably and accurately exists today in the form of integrated-air samplers, which collect PAHs on a filter cartridge. The compounds are then extracted and measured using sophisticated laboratory analysis. However, this method, although scientifically well validated, is time-consuming, labor-intensive, and expensive, and does not provide vital information about real-time exposures or acute exposures during periods of high PAH emissions, such as start-up and shut-down of engines. With those practical limits in mind, the investigators tested three recently developed photoelectric aerosol sensors (PASs)--two desktop monitors and a prototype handheld model--which were designed to measure and record airborne PAH concentrations in real time.
Tests were conducted in a variety of locations during several different flight-related activities over a period of three days at an Air National Guard base in Georgia. In most of the monitoring events, two of each type of PAS were collocated with two integrated-air samplers. The PASs were evaluated by comparing their results to those of the integrated-air samplers.
The comparisons proved to be inconclusive. Due to inherent differences between the instruments, data from the PASs must be calibrated to the values from the air samplers, but a number of limitations made reliable calibration factors between the two types of monitor difficult to establish. For example, the integrated-air samplers are designed to capture both vapor-phase and particle-bound PAHs from ambient or indoor air over periods of 12-24 hours. In this study, the sampling events were much shorter (45-135 minutes), so the amounts of particle-bound PAHs collected by the air samplers were low enough to fall under the devices' method detection limits. On the other hand, the dynamic range of the PASs, which measure only particle-bound PAHs, were often exceeded during the monitoring events--the levels detected by the real-time monitors literally went off the charts, exceeding the range of the instruments. This also served to compromise the accuracy of the calibration factors. The authors concluded that until these calibration issues are ironed out, the real-time PAS monitors can only be used to provide semiquantitative screening estimates of PAH exposure.
But their use as screening tools is important for protection against high exposures. As presently configured, these instruments can provide an immediate warning when PAH concentrations get very high. While overall PAH concentrations were low in the monitored aircraft activities, the extremely high concentrations measured at certain points (such as engine start-up) indicate that for brief periods personnel are at risk of high acute exposure to PAHs, at which times the use of personal protective equipment may be advisable. -Ernie Hood
Last Updated: September 21, 2000





