
| |  | |  |
Research
|
| DDE, a Degradation Product of DDT, and
Duration of Lactation in a Highly Exposed Area of Mexico Lea A. Cupul-Uicab,1,2 Beth C.
Gladen,3,* Mauricio Hernández-Ávila,1,4
Jean-Philippe Weber,5 and Matthew P. Longnecker2 1Center
for Population Health Research, Instituto Nacional de Salud
Pública, Cuernavaca, Morelos, México; 2Epidemiology
Branch, and 3Biostatistics Branch, National Institute of
Environmental Health Sciences, National Institutes of Health,
Department of Health and Human Services, Research Triangle
Park, North Carolina, USA; 4Subsecretaría de Promoción y
Prevención de la Salud. Ministry of Health,
México City, Mexico; 5Centre de Toxicology,
Institut National de Santé Publique du Québec, Québec,
QC, Canada Abstract
Background: Higher levels of 1,1-dichloro-2,2-bis(p-chlorophenyl) ethylene (DDE) , the major degradation product of 1,1,1-trichloro-2,2-bis(4-chlorophenyl) ethane (DDT) , have been related to shorter duration of breast-feeding in previous studies. If DDE truly shortens lactation, this has public health importance regarding infant mortality and the use of DDT for malaria control. Objective: Our aim was to assess the relationship of maternal DDE concentrations with length of subsequent lactation. Methods: We conducted a relatively large study in a highly exposed area of Mexico. We followed 784 mother–son pairs to determine length of lactation. DDE and DDT were measured in maternal serum obtained within a day of delivery. We fit proportional hazard models with and without stratifying by previous breast-feeding, because an association of DDE with duration of lactation among those who breast-fed previously could be attributed to a noncausal mechanism. Results: Compared with those with DDE concentrations ≤ 3.00 µg/g, the adjusted hazard ratios of weaning according to DDE category were, for concentrations 3.01–6.00 µg/g, 1.27 [95% confidence interval (CI) , 1.04–1.55] ; for concentrations 6.01–9.00 µg/g, 1.23 (95% CI, 0.92–1.63) ; and for concentrations > 9.00 µg/g, 1.17 (95% CI, 0.92–1.49) . The corresponding ratios for women who previously breast-fed were 1.40 (95% CI, 1.06–1.87) ; 1.91 (95% CI, 1.24–2.93) ; and 1.76 (95% CI, 1.22–2.53) . Those for women who had not breast-fed previously were 1.14 (95% CI, 0.86–1.52) ; 0.90 (95% CI, 0.61–1.31) ; and 0.91 (95% CI, 0.66–1.26) . Conclusions: Data from our relatively large study in a highly exposed area of Mexico did not support the hypothesis that exposure to DDE shortens length of lactation. The association seen in women who previously breast-fed was likely attributed to a noncausal mechanism. Nonetheless, whether DDT has other important adverse effects on humans is still an open question. Key words: breast-feeding, DDE, DDT, infant, lactation. Environ Health Perspect 116:179–183 (2008) . doi:10.1289/ehp.10550 available via http://dx.doi.org/ [Online 22 November 2007]
Address correspondence to M.P. Longnecker, National Institute of Environmental Health Sciences, MD A3-05, PO Box 12233, Research Triangle Park, North Carolina 27709 USA. Telephone: (919) 541-5118. Fax: (919) 541-2511. E-mail: longnec1@niehs.nih.gov *Currently retired, Cary, NC, USA. We thank the field work team in Tapachula, Chiapas, México, for their valuable support during data collection. This study was supported in part by a contract from the National Institute of Environmental Health Sciences (NIEHS) , National Institutes of Health (NIH) (N01-ES-15467) , in part by the Intramural Research Program of the NIEHS, NIH, and in part by the Instituto Nacional de Salud Pública. The authors declare they have no competing financial interests. Received 12 June 2007 ; accepted 21 November 2007. |
|
|
 |
Mothers with higher levels of
1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene (DDE), the major
degradation product of
1,1,1-trichloro-2,2-bis(4-chlorophenyl)ethane (DDT), stopped
breast-feeding their children sooner than those with
lower levels of exposure in previous studies (Gladen and Rogan
1995; Karmaus et al. 2005; Rogan et al. 1987). Shorter duration
of breast-feeding increases infant mortality (Gartner et al.
2005; Leon-Cava 2002); if DDT exposure truly shortens
lactation, this has public health importance. DDT is still used
for control of malaria in some countries [Curtis 2002;
Longnecker 2005; World Health Organization (WHO) 2006],
resulting in relatively high human exposure. Characterizing
DDT's human toxicity accurately is important for making
good decisions about vector-control policy.
When the association of DDE with shortened
duration of lactation was first reported, the possibility that
a noncausal mechanism could account for the relation was
recognized (Rogan et al. 1987). A woman who breast-fed longer
in her first lactation will tend to breast-feed longer the
second time as well, whereas a woman who breast-fed shorter
once will tend to do so again. Also, because DDE is excreted
in milk (Rogan et al. 1986), the woman with longer lactation
would
have eliminated more DDE via breast milk than the one who
breast-fed less. That creates a noncausal association between
higher DDE and shorter lactation in women who previously
breast-fed. In the first study to examine the association,
higher DDE was associated with shorter lactation even among
those breast-feeding for the first time (Rogan et al. 1987),
but a second study found an association only in women who
previously breast-fed (Gladen and Rogan 1995). Subsequent
reports have given inconsistent results (Karmaus et al. 2005;
Weldon et al. 2006).
To evaluate the association
with greater statistical power, we conducted a relatively large
study in
Tapachula, Chiapas, Mexico. This population was exposed for
almost 40 years: DDT was used for agriculture until 1991 and
for malaria control until 1998. The primary aim of the present
study was to assess the relationship of maternal DDE
concentrations and length of subsequent lactation in a cohort
of 784 mother–son pairs in which maternal serum DDT and
DDE levels at delivery had been previously determined.
The participants were recruited from a
previous cross-sectional study of newly-delivered male infants
and their mothers (n = 872), conducted in 2002–2003
(Longnecker et al. 2007; Romano-Riquer et al. 2007). Because
the objective of that study was to evaluate antiandrogenic
effects of DDE, only boys were recruited. In the previous
study, women were enrolled during the postpartum period at both
of the city's hospitals, which also serve the surrounding
areas. Exclusion criteria for the mother included the following:
age > 35 years; preeclampsia or pregnancy-related diabetes
or hypertension; seizure disorders requiring daily medication;
use of corticosteroids; history of psychiatric, kidney, or
cardiac disease or repeated urinary tract infections; and
non-Spanish speaker. Infants were excluded if gestational age
at delivery, as estimated by the Capurro scale (Capurro et al.
1978) or the medical record (based on last menstrual period),
was < 36 weeks, birth weight was < 2,500 g, pregnancy was
not singleton, Apgar score at 5 min was ≤ 6,
or child required intensive care. The previous study included
questionnaires, anthropometry, and collection of maternal serum
for the measurement of DDT and DDE.
We conducted a follow-up
study of these subjects to determine length of lactation. Mother–son
pairs were eligible if they were enrolled in the
cross-sectional study, the child was not given up for adoption
(n =
1), and the mother had not used medications that inhibit or
increase milk production, including contraceptive pills with
estrogens, bromocriptine, or metoclopramide (n = 6). Eligible
mothers were invited to participate and gave informed consent
before participation in the study. The study protocol was
reviewed and approved by the institutional review boards at the
Instituto Nacional de Salud Pública in México and
the National Institute of Environmental Health Sciences in the
United States.
|

Figure 1. Kaplan-Meier
survival curves by DDE concentration categories (µg/g lipids)
in women with (n = 416) (A) and without (n = 368) (B) history of previous lactation. The graph was
truncated at 25 months.
|
Table 1.
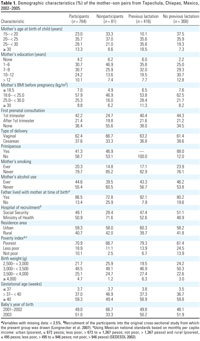
|
Table 2.

|
Table 3.

|
Of those who were eligible (n = 865), 90.6%
(n =
784) were followed. Of the remaining 81 (9.4%), for 10 subjects
the
recorded address did not exist, for 59 the address was found
but the mother was not, for 4 the mother or father refused to
participate, and for 8 the mother or child had died.
Follow-up started on 20 January 2004. At
that time, the ages of the babies were 3.3 to 25.1 months
(median, 13.2). For children already weaned at the first visit,
we recorded the duration of lactation. For those still being
breast-fed, we continued home visits approximately every 2
months until the child was weaned.
The schedule of visits varied across the
participants for logistical reasons. The median age at the
first visit was 15.7 months (25th and 75th percentiles, 11.6
and 21.6 months). The number of visits ranged from 1 to 7
(median, 3), and the time between visits ranged from 0.8 to
11.4 months (median, 2.2). The interval from weaning until the
next study visit (i.e., the recall period for duration of
lactation) had a median of 7.4 months (quartiles, 2.4 and
14.1).
Length of lactation. A questionnaire
was administered to the mothers during each home visit. Interviewers
received special training before
beginning the study and periodic retraining throughout. Length
of lactation was defined as the last time the child received
any breast milk, regardless of the introduction of liquids or
solid foods. Mothers were first asked whether they ever
breast-fed the child and, if so, whether they were currently
breast-feeding. If they had stopped, we asked the number of
months they had breast-fed and the date they completely
stopped. Twenty-six inconsistencies were corrected; if the
mother said at one visit that she was still breast-feeding, but
at the next visit said she weaned before the age at the
previous visit, we used the age at the previous visit as the
duration. For 11 mothers (1.4%) who reported that their attempt
to breast-feed failed, a duration of 5 days was assigned. If
the child was not weaned by the last study visit (n =
97, 12.4%), duration was censored at that time.
DDE and DDT. DDE
and DDT were measured in maternal serum obtained within 1 day
of delivery. DDE and DDT were quantitated after solid phase
extraction (C18 column purification), using gas
chromatography with mass spectrometry detection (Saady and
Poklis 1990; Smith 1991). The limit of detection for both
analytes was 0.2 µg/L. The between-assay coefficients of
variation were 7% for DDE (at 10 µg/L) and 6% for DDT (at
2.5 µg/L). A recovery of 97% was obtained for both
analytes. Total serum lipid content was estimated based on
levels of free and total serum cholesterol, triglycerides, and
phospholipids (Patterson 1991), measured using standard
enzymatic methods. DDE and DDT concentrations were expressed
as micrograms per gram of lipids.
Statistical analysis. To test the hypothesis under study, we used a Cox
proportional hazard model for weaning. We used Kaplan-Meier
survival curves to describe unadjusted duration. All analyses
were done using Stata Statistical Software (release 9.0;
StataCorp, College Station, TX, USA).
As described above, an artifactual
association of DDE with shorter lactation duration can occur
in women who previously breast-fed, but not in women who did
not.
A true association should be observed in both groups. Thus, we
first constructed models using all participants, but then also
stratified on previous breast-feeding.
In the primary analysis, DDE categories
were defined as ≤ 3 µg/g, 3.01–6 µg/g,
6.01–9 µg/g, and > 9 µg/g. These
categories were chosen before examining the DDE–outcome
relationships, and resulted in a large exposure contrast
between those in the high and low categories while maintaining
adequate numbers for analysis. For DDT, we used these
categories: ≤ 0.25 µg/g, 0.26–0.75 µg/g,
0.76–1.99 µg/g, and ≥ 2 µg/g.
Variables considered a priori as
potential confounders were hospital of recruitment, rural/urban
residence, mother's age at birth, education (none,
1–6, 7–9, 10–12, ≥ 13 years), prepregnancy
body mass index (BMI), smoking (ever, never), poverty status
(poorest, somewhat poor,
not poor) and previous breast-feeding (yes, no). We used weight
at the first follow-up visit to calculate BMI if the
prepregnancy weight was missing (n = 127, 16.2%). Smoking
referred to any time before or during the pregnancy; only 10
women smoked
during pregnancy.
We measured poverty
status using national standards, based on monthly per capita
income in Mexican pesos.
In urban areas, those with an income < 672 Mexican pesos
were in the poorest category; they would have difficulty buying
adequate food. Of the remainder, those < 1,367 pesos lacked
adequate income for other human needs, so were still considered
poor. In rural areas, the cut points were 495 and 946 pesos,
respectively [SEDESOL (Secretaría de Desarrollo Social)
2002]. If income at recruitment was missing (n =
77, 9.8%) we used income at the first follow-up visit.
Our model assumes proportionality of the
hazards. Because residence area and hospital of recruitment
were important confounders and were not proportional, we
stratified the baseline hazard by these two variables.
We evaluated additional confounders using
the change in estimate method, starting with all variables and
deleting one by one in a stepwise way (Greenland 1989). A
variable was considered a confounder if removing it caused a
change in the hazard ratio for DDE (considered both as a
continuous and categorical variable) of ≥ 10%.
Potential confounders were mother's alcohol use, whether the mother
received support for breast-feeding, timely initiation of breast-feeding,
trimester of first prenatal consultation, type of delivery,
whether the father was living with the mother at the time of
delivery, whether the mother received instructions about how to
breast-feed, baby's year of birth, and nipple anatomy
problems such as flat or inverted nipples. Support for breast-feeding
(e.g., orientation, motivation, and help with home tasks) was
defined as any noneconomic help that the mother received from
relatives or people close to her. Initiation was considered
timely if the baby began suckling within 1 hr of birth (Setty
2006). None of these variables was shown to be a confounder in
the whole population or in either stratum of previous breast-feeding.
We looked also for
interactions of DDE (considered as a continuous variable) with
poverty index,
mother's BMI before pregnancy, baby's year of
birth, nipple anatomy problems, and previous breast-feeding.
Interaction was evaluated with the likelihood ratio test. If
the p-value
was ≤ 0.10, the corresponding interaction term was considered
for further analysis. Previous breast-feeding had an important
interaction with DDE. After stratifying on previous breast-feeding,
no other interactions met the criteria.
Reasons for weaning. Lactation may
stop for a number of reasons, only some of which are relevant to
our hypothesis. We asked the mother
the reason she had not been breast-feeding or had interrupted
breast-feeding. In a secondary analysis, participants who reported
stopping for
reasons unrelated to the hypothesis were censored at the time
that they reported weaning; the assumption was that the pair
would have continued with breast-feeding had not some external
cause intervened, because there was no biological failure of
the lactation. Reasons that were considered unrelated to the
hypothesis included mother and child separation, use of
hormonal contraception, and pregnancy. Because almost all
the participants gave more than one reason, combinations that
included these external events were censored also.
Participants were young
(mean, 24 years of age), few attended college, and most lived
in the city and
its
surrounding areas (Table 1). Because Tapachula is one of
the poorest states in the country, the fact that 70% of the
participants were in the poorest category and only 10% were not
poor was expected. Children < 2,500 g of birth weight and
< 36 weeks of gestational age were excluded from the
original study, which explains the absence of these groups.
Those not participating (n =
81) were on average less exposed to DDE (median, 1.7 µg/g lipids)
than were participants (median, 2.7 µg/g lipids). Among
the participants, DDE was higher in women without previous breast-feeding
[median, 3.36; interquartile range (IQR), 5.58] than in women
with previous breast-feeding (median, 2.26; IQR, 3.29). This
was expected, because DDE is eliminated via breast milk (Rogan
et al. 1986).
The median duration of lactation was 10.8
months (quartiles, 5 and 17.8). About 40% of women breast-fed
their child within an hour and almost all within a day after
birth (Table 2). One-third introduced formula within the first
month. The proportion of women with inverted nipples was lower
than in a previous report from a white U.S. population (3.05%)
(Park et al. 1999). Few women reported problems initiating
breast-feeding.
Most did not work outside the home.
The median duration of lactation according
to category of DDE was 12.0 months (≤ 3.00 µg/g), 9.0
months (3.01–6.00 µg/g), 9.0 months
(6.01–9.00 µg/g), and 11.0 months (> 9.00
µg/g). Among women with no previous breast-feeding, those
most exposed had the longest median duration (≤ 3.00 µg/g,
7.4 months; 3.01–6.00 µg/g, 9.0 months; 6.01–9.00
µg/g, 11.5 months; and > 9.00 µg/g, 11.3
months), whereas among women with previous breast-feeding,
those least exposed had the longest median duration (≤ 3.00
µg/g, 12.6 months; 3.01–6.00 µg/g, 8.8
months; 6.01–9.00 µg/g, 8.0 months; and > 9.00
µg/g, 9.0 months) (Figure 1).
Unadjusted hazard ratios (HRs) showed
similar results (Table 3); a statistically significant increase
in the hazard of weaning with an increase of DDE levels was
seen only in women who previously breast-fed. Adjustment for
potential confounders yielded essentially the same results.
Adding an interaction term between previous breast-feeding and
DDE (data not shown) produced similar results; the interaction
term was significant (p = 0.007), and the magnitude of the DDE
effects was similar. We also evaluated DDT exposure, which showed
no
association with duration of lactation in any of the models
(data not shown). Results from a secondary analysis, censoring
women who weaned for reasons not considered related to DDE
exposure, were similar.
We also explored whether
DDE was associated with problems initiating breast-feeding. The
adjusted odds ratio per unit increase in DDE levels (natural
log scale) for women who had not breast-fed previously was 1.96
[95% confidence interval (CI), 1.18–3.26] and for women
who had was 0.97 (95% CI, 0.60–1.57). Results for DDT
were similar (data not shown).
Our results do not support an association
between length of lactation and DDE or DDT. A statistically
significant increase in the hazard of weaning associated with
DDE was observed only in women who previously breast-fed, which
probably reflects a noncausal mechanism. If the association
between DDT and duration of breast-feeding were causal, it
should also be present among women who had never breast-fed.
The present results are
similar to those reported in another region (Durango) of Mexico
(Gladen and
Rogan 1995). In that study, an association was observed only
in women who previously breast-fed. In a recent study among
low-income Mexican-American mothers from an agricultural region
of California, no association was seen (Weldon et al. 2006).
However, our results differ from a prospective study conducted
in North Carolina, which showed an association in those with
no
previous lactation (Rogan et al. 1987). A small study in
Michigan with no contemporaneous measure of DDE also showed an
association, but only in women who did not smoke during
pregnancy (Karmaus et al. 2005). Using approximate conversions
of milk to serum, we compared exposures in these studies.
Median exposure was higher in Durango (4.2 µg/g) than in
our study (2.7 µg/g), whereas U.S. women had lower
medians (North Carolina, 1.7; California, 1.1; and Michigan, ~ 1.0
) (Karmaus et al. 2005; Weldon et al. 2006). However, the
proportion with high levels of DDE (> 8 µg/g lipids)
was larger in our study (17%) than in the Durango study (9%),
increasing our power. There is no obvious explanation for the
differences between the current study and the very similar
study in North Carolina. Although the previous authors used
weeks predominantly breast-fed as the outcome, results using
total lactation were similar (Chen and Rogan 2003). The
earliest study on DDE and duration of lactation (Rogan et al.
1987) was done when levels of other organochlorines would have
been higher than in the present study. Any association with DDE
could have been attributed to confounding by such compounds.
However, Rogan et al. (1987) measured polychlorinated
biphenyls, which were not related to duration of lactation.
Another potential reason for a difference between the studies
was that because the study population has been exposed to DDT
for generations, they may comprise a group that is no longer
sensitive to DDT's effects: Susceptible subjects may not
have been able to reproduce (Longnecker et al. 2005; Venners
et
al. 2005). Yet another alternative explanation for the null
results is that we studied very poor women, who must breast-feed
because they cannot buy other food. Richer women have more
options, and may be more inclined to stop breast-feeding at the
first sign of problems.
If DDE affects lactation,
it may affect only establishment of lactation. Milk supply is
under endocrine
control for the first 2–3 days; once lactation is
established, it is more under autocrine (milk removal) than
hormonal control (Lawrence and Lawrence 2005; Neville et al.
2001). If DDE decreased milk production in the first days, but
women were nevertheless able to establish lactation, DDE may
not affect duration of lactation. Estrogens act this way; they
are effective for preventing milk production initially, but
less effective for stopping an established lactation (Anonymous
1977). Therefore, we examined whether DDE was associated with
difficulties initiating breast-feeding. We found that DDE
increased the risk of difficulty initiating breast-feeding, but
only in women who had not breast-fed previously. Such women may
be more susceptible to endocrine disruption. Even if DDE
impairs initiation, nearly all women in this study were able to
breast-feed (> 98%). Among the few (n =
11) not able to breast-feed, the median DDE level was increased
(4.8 µg/g; IQR, 5.1).
Although this is consistent with the endocrine disruption
hypothesis, the data are too sparse to draw any conclusions.
Future studies involving women from areas where DDT is
currrently used (WHO 2006) may be necessary to corroborate this
preliminary result.
The results could be biased if the
children who died (n = 8) were highly exposed, unable to breast-feed,
and consequently became terminally ill. However, a serious bias
is unlikely. Four of these children were in the lowest category
of DDE exposure; of the remaining four, two were known to have
breast-fed
at least 9 months. Thus, this potential bias is unlikely to
explain the observed results.
DDE levels were determined from serum
drawn at delivery, using standardized methods with a low
between-assay coefficient of variation. The study was double
blind: Neither interviewers nor participants knew the DDT or
DDE levels. Therefore, there is little reason to expect
important information bias. Any errors should be
nondifferential, causing bias toward the null. Length of
lactation was recorded within 7 months after weaning in half
of participants, and within 24 months for 96% of women. Errors
should again be nondifferential and biased toward the null.
Moreover, reliability of long-term recall of breast-feeding
duration is high (r = 0.86) (Tomeo et al. 1999). A limitation
of the current study was that we included only male infants,
because
this study was derived from a previous one limited to males.
However, there is no reason to suppose that any direct effect
of DDE on lactation would be different in boys and girls.
Perhaps more important was the exclusion of preterm and
low-birth-weight infants. If initiation of breast-feeding is
more difficult among such infants, this exclusion may have
diminished our ability to detect any effect.
In conclusion, our relatively large study
in a highly exposed area of Mexico did not support the
hypothesis that exposure to DDE shortened length of lactation.
The association seen in women who previously breast-fed was
likely attributed to a noncausal mechanism. Nonetheless,
whether DDT use has other important adverse effects on humans
is still an open question. |
|
 |
| [References Listed in PubMed]
References
[Anonymous]. 1977. Suppressing lactation.
Br Med J 1(6055):189.
Capurro H, Konichezky S,
Fonseca D, Caldeyro-Barcia R. 1978. A simplified method for diagnosis
of
gestational age in the newborn infant. J Pediatr 93(1):
120–122.
Chen A, Rogan WJ. 2003.
Nonmalarial infant deaths and DDT use for malaria control. Emerg
Infect Dis 9(8):
960–964.
Curtis CF. 2002. Should
the use of DDT be revived for malaria vector control? Biomedica
22(4):
455–461.
Gartner LM, Morton J, Lawrence
RA, Naylor AJ, O'Hare D, Schanler RJ, et al. 2005. Breastfeeding and
the use of human milk. Pediatrics 115(2):496–506.
Gladen BC, Rogan WJ. 1995.
DDE and shortened duration of lactation in a northern Mexican
town. Am
J Public Health 85(4):504–508.
Greenland S. 1989. Modeling
and variable selection in epidemiologic analysis. Am J Public
Health 79(3):
340–349.
Karmaus W, Davis S, Fussman
C, Brooks K. 2005. Maternal concentration of dichlorodiphenyl
dichloroethylene (DDE) and initiation and duration of breast
feeding. Paediatr Perinat Epidemiol 19(5):388–398.
Lawrence RA, Lawrence RM. 2005.
Breastfeeding: A Guide for the Medical Profession. 6th ed. St.
Louis, MO:Elsevier Mosby.
Leon-Cava N. 2002. Cuantificacion
de los Beneficios de la Lactancia Materna: Reseña de la
Evidencia [in Spanish]. Washington, DC:Organizacion
Panamericana de la Salud.
Longnecker MP. 2005. Invited
commentary: why DDT matters now. Am J Epidemiol 162(8):726–728.
Longnecker MP, Gladen BC, Cupul-Uicab LA,
Romano-Riquer SP, Weber JP, Chapin RE, et al. 2007. In utero exposure to
the antiandrogen 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene
(DDE) in relation to anogenital distance in male newborns from
Chiapas, Mexico. Am J Epidemiol 165(9):1015–1022.
Longnecker MP, Klebanoff
MA, Dunson DB, Guo X, Chen Z, Zhou H, et al. 2005. Maternal serum
level of
the
DDT metabolite DDE in relation to fetal loss in previous
pregnancies. Environ Res 97(2):127–133.
Neville MC, Morton J, Umemura
S. 2001. Lactogenesis. The transition from pregnancy to lactation.
Pediatr Clin North Am 48(1):35–52.
Park HS, Yoon CH, Kim HJ.
1999. The prevalence of congenital inverted nipple. Aesthetic
Plast Surg
23(2):144–146.
Patterson DG. 1991. Method 6:
determination of specific polychlorinated dibenzo-p-dioxins
and dibenzofurans in blood and adipose tissue by isotope
dilution-high-resolution mass spectrometry. IARC Sci Publ 108:
299–342.
Rogan WJ, Gladen BC, McKinney
JD, Carreras N, Hardy P, Thullen J, et al. 1986. Polychlorinated
biphenyls
(PCBs) and dichlorodiphenyl dichloroethene (DDE) in human milk:
effects of maternal factors and previous lactation. Am J Public
Health 76(2):172–177.
Rogan WJ, Gladen BC, McKinney
JD, Carreras N, Hardy P, Thullen J, et al. 1987. Polychlorinated
biphenyls
(PCBs) and dichlorodiphenyl dichloroethene (DDE) in human milk:
effects on growth, morbidity, and duration of lactation. Am J
Public Health 77(10):1294–1297.
Romano-Riquer SP, Hernandez-Avila
M, Gladen BC, Cupul-Uicab LA, Longnecker MP. 2007. Reliability
and
determinants of anogenital distance and penis dimensions in
male newborns from Chiapas, Mexico. Paediatr Perinat Epidemiol
21(3):219–228.
Saady JJ, Poklis A. 1990.
Determination of chlorinated hydrocarbon pesticides by solid-phase
extraction
and capillary GC with electron capture detection. J Anal
Toxicol 14(5):301–304.
SEDESOL
(Secretaría de Desarrollo
Social). 2002. Technical note on the measurement of poverty
based on the results of the National Survey of Household Income
and Expenditures [in Spanish]. SEDESOL. Available: http://sedesol2006.sedesol.gob.mx/subsecretarias/prospectiva/main_medicionpobreza.htm
[accessed 29 March 2007].
Setty V. 2006. Better Breastfeeding,
Healthier Lives. Publication no. 14.Baltimore, MD:Johns
Hopkins Bloomberg School of Public Health, The INFO Project.
Smith G. 1991. Chlorinated
hydrocarbon insecticides. In: Handbook of Pesticide Toxicology:
Classes of
Pesticides (Hayes WJ Jr., Laws ER Jr., eds). San Diego:Academic
Press, 731–915.
Tomeo CA, Rich-Edwards
JW, Michels KB, Berkey CS, Hunter DJ, Frazier AL, et al. 1999.
Reproducibility
and validity of maternal recall of pregnancy-related events.
Epidemiology 10(6):774–777.
Venners SA, Korrick S,
Xu X, Chen C, Guang W, Huang A, et al. 2005. Preconception serum
DDT and pregnancy
loss: a prospective study using a biomarker of pregnancy. Am
J
Epidemiol 162(8):709–716.
Weldon RH, Webster M, Harley K, Bradman A,
Fenster L, Barr DB, et al. 2006. Exposure to persistent organic
pollutants and duration of lactation in Mexican-American
mothers. Epidemiology 17(6):S193.
WHO. 2006. WHO Gives Indoor Use of DDT A
Clean Bill of Health for Controlling Malaria. Washington, DC:
World Health Organization. Available: http://www.who.int/mediacentre/news/releases/2006/pr50/en/ [accessed
31 January 2007]. |
|
 |
|
| |

