Beginning early in sex determination and
gonadogenesis in fish, communication between
nonadjacent tissues is necessary. This communication
is accomplished through the endocrine system,
which controls sex differentiation through
complex interactions between the central nervous
system and gonads using pituitary-derived gonadotropins
and sex steroids produced in the gonad and
brain (Nagahama 1994). Production of these
sex steroids is very strongly linked with the
early steps of gonadal differentiation, and
they can influence long-term sex determination
choices (Devlin and Nagahama 2002). Further,
it has been demonstrated that exposure to estrogen
or estrogen-mimicking chemicals during a critical
period of development can result in genotypic
XY males developing into fully functional phenotypic
females, whereas exposure of genotypic XX females
to androgenic chemicals can result in development
of phenotypic males (Cheek et al. 2001a, 2001b;
Edmunds et al. 2000; Gronen et al. 1999; Hunter
and Donaldson 1983; Yamamoto 1969).
Because of the critical role of estrogen
in the very early stages of sex determination
and sex differentiation, the estrogen-synthesizing
enzyme is of likely importance. As in all vertebrates,
in fish this function is performed by cytochrome
P450 aromatase, which converts androgens to
estrogens and is expressed in a number of tissues,
including the brain, liver, and gonads. Recently,
a second isoform of aromatase (cyp19b;
Genbank accession no. AY319970; National Center
for Biotechnology Information, National Library
of Medicine, Bethesda, MD) has been found in
the brain of several teleost species (Callard
and Tchoudakova 1997; Chiang et al. 2001; Kuhl
et al. 2005). This isoform has much higher
activity and mRNA levels in the brain than
the ovary has of ovarian aromatase (Kishida
and Callard 2001).
It has been hypothesized that altered expression
of aromatase is important in environmentally
influenced sex differentiation. Increases in
brain aromatase expression occur in 0- to 14-day-posthatch
(dph) medaka concomitantly with xenoestrogen-induced
feminization (Kuhl et al. 2005), whereas ovarian
aromatase mRNA transcripts at this life stage
are not detectable. Conversely, inhibition
of aromatase activity can result in masculinization
of genotypic females (Piferrer et al. 1994).
Whether altered aromatase activity is directly
responsible for sex reversal is not known.
In the present study we aimed to further examine
the critical role aromatase plays in the genesis
of developmental abnormalities in response
to endocrine disruptors.
Most research to date on the effects of endocrine-disrupting
chemicals (EDCs) has focused on abnormalities
induced by exposure to a single compound. However,
in the environment, humans and wildlife are
exposed to diverse mixtures of androgenic,
estrogenic, antiandrogenic, and antiestrogenic
compounds (Bush et al. 1990). Only recently
has focus begun to shift to examining the impact
of mixtures of EDCs on human and wildlife populations.
Most of this research, however, has focused
on the synergism between several weak estrogenic
compounds (Bergeron et al. 1999; Payne et al.
2000). These studies demonstrate that there
is an additive effect of several weakly estrogenic
EDCs. To date, little work has been done on
the impacts of mixtures of antagonistic EDCs.
It is unknown if an androgenic or antiestrogenic
chemical can block the activity of an estrogenic
one. Here we address this question by examining
if xenoestrogen [dichlorodiphenyltrichloroethane
(o,p´-DDT)]-induced feminization
of developing medaka can be prevented by coexposure
to a pharmaceutical [fadrozole (FAD)] and an
environmental aromatase inhibitor (AI), tributyltin
(TBT). We hypothesize that o,p´-DDT
activates the estrogen receptor to induce transcription
of brain aromatase. Increased brain aromatase
activity results in increased estradiol levels
feminizing the development of the fish. We
further hypothesize that inhibiting aromatase
activity will prevent a xenoestrogen-induced
surge of estradiol production and prevent feminization.
o,p´-DDT is a known xenoestrogen
that can negatively affect reproduction and
development in fish, through immersion exposure
(Cheek et al. 2001a, 2001b) and direct injection
into oocytes (Edmunds et al. 2000). FAD has
been used extensively in breast cancer research
as a pharmaceutical inhibitor of aromatase
and has been shown to be a reversible competitive
inhibitor of aromatase in birds (Elbrecht and
Smith 1992), mammals (Steele et al. 1987),
and fish (Afonso et al. 1999, 2001). In Nile
tilapia (Oreochromis niloticus), the
suppression of aromatase with a FAD-treated
diet resulted in a 100% male population Afonso
2001; Kwon et al. 2000). TBT has been used
as an antifouling biocide in paint for boats,
for wood treatment and preservation, and as
a fungicide/bactericide in textile and industrial
water systems (Gehring et al. 1991). At very
low concentrations, TBT can disrupt the endocrine
system, as evidenced by the induction of male
sexual characteristics in female gastropods
(Oberdorster and McClellan-Green 2002; Smith
1981). TBT has also been shown to be an EDC
in fish, as well. A 30-day exposure to 100
ng/L TBT in zebrafish (Danio rerio)
resulted in an almost completely male population
(McAllister and Kime 2003). It is hypothesized
that TBT acts as a competitive inhibitor of
aromatase, which causes accumulation of testosterone
and masculinization of the organism (McAllister
and Kime 2003; Oberdorster and McClellan-Green
2002).
Our results from the present study demonstrate
that an increase in aromatase activity is not
needed for EDC-induced feminization and that
antiestrogenic AIs are unable to prevent xenoestrogen-induced
feminization.
Experimental animals. Medaka
(d-rR strain) used in this study were hatched
from broodstock cultured and maintained at
the Gulf Coast Research Laboratory, University
of Southern Mississippi (Ocean Springs, MS).
The d-rR strain contains a Y-chromosome-linked
gene coding for a red body color phenotype,
allowing for the simple determination of sex
genotype: males have red phenotype, and females
have white. Medaka also have secondary sex
characteristics that are reflective of sexual
phenotype. Females have shorter anal fins,
and males have a notched dorsal fin. With the
d-rR strain and secondary sex characteristics
(fin morphology), sex reversals can be determined
by simple observation of body color and fin
development. Animal care and experimentation
were conducted in accordance with University
of Southern Mississippi guidelines for animal
care and use, and animals were treated humanely
with regard for alleviation of suffering. Broodstock
cultures were maintained in 300-L fiberglass
runs at a 27 ± 1°C. A 16-hr light/8-hr
dark photoperiod was provided by timer-controlled
overhead fluorescent lights. Broodstock cultures
were fed a minimum of twice daily, one feeding
of commercial flake (Ziegler Brothers, Santa
Anna, CA) and one feeding of brine shrimp nauplii, ad
libitum.
EDC exposure. Medaka were exposed
in two consecutive experiments. In the first
experiment medaka were exposed to nominal concentrations
of 0, 10, 50, and 100 µg/L FAD and 0.7 µg/L
TBT. This was designed as a range-finding study
to determine potential masculinizing concentrations
of a pharmaceutical AI and determine the responsiveness
of the system to an environmental AI. The second
exposure consisted of nominal concentrations
of 300 µg/L FAD, 1.5 µg/L TBT,
7.5 µg/L DDT, 50 µg/L FAD with
7.5 µg/L DDT, 300 µg/L FAD with
7.5 µg/L DDT, and 1.5 µg/L TBT
with 7.5 µg/L DDT. DDT concentrations
were selected based on levels previously shown
to induce male-to-female sex inversion (Kuhl
et al. 2005).
Eggs were collected from 15-cm cylindrical
filter sponges used as spawning substrates.
Embryos were transferred to glass hatching
jars containing about 4 L of hatching solution
(1.00 g/L NaCl, 0.030 g/L KCl, 0.040 g/L CaCl2,
0.162 g/L MgSO4 in distilled water)
with the salinity of the hatching solution
brought to 5 g/L with NaCl to control fungus.
Hatching jars were maintained under continuous
fluorescent light in a water bath at 24 ± 1°C
and vigorously aerated to suspend embryos.
At hatch, 75 d-rR fry were housed in three
retention chambers (100-mm Petri dish bottoms
with attached 475-µm nylon collar) with
25 fish in each chamber. For the first exposure,
fry were exposed to four duplicated exposure
treatments (control, three FAD concentrations,
and one TBT concentration) for a total of 8
test aquaria. For the second exposure, fry
were exposed to seven duplicated exposure treatments
(control, carrier control, FAD, two FAD + DDT
combinations, TBT, and TBT + DDT) for a total
of 14 test aquaria. Test aquaria were 35 L
with a water depth of 19 cm maintained by drain
siphon. Test aquaria were housed within a central
water bath kept at 27 ± 1°C and
provided with a 16-hr light/8-hr dark photoperiod
supplied by fluorescent bulbs.
Exposure was conducted in a setup similar
to that described by Walker et al. (1985) and
Manning et al. (1999). Briefly, a water partitioner
delivered 2 L of test solution each cycle to
splitter/mixing boxes that dispensed 1 L to
each duplicate aquarium. The exposure system
cycled between three and five cycles per hour
during the exposure period. Test concentrations
were prepared each cycle by injection of appropriate
stock to the splitter boxes of each treatment
using Hamilton PSD2 liquid injectors (Hamilton
Company, Reno, NV). Stocks were created by
dissolving the compound in the appropriate
solvent. DDT and TBT were dissolved in triethylene
glycol, and FAD was dissolved in well water.
Water quality (pH, temperature, and dissolved
oxygen) was measured twice each week, and water
samples were removed four times (day 0, 5,
8, and 14) for analytical analysis of the test
chemicals. Survival was monitored and recorded
daily, and all dead fry were removed. Six fish
per aquaria (12/treatment) were sampled, weighed,
and archived for molecular analysis on days
5, 9, and 14. Six sampled fish per treatment
were preserved in 200 µL RNAlater (Ambion,
Austin, TX) for mRNA analysis, and six were
preserved in 200 µL phosphate buffer
(100 mM KCl, 10 mM KH2PO4,
1 mM EDTA, 10 mM dithiothreitol, pH 7.4) for
enzyme activity analysis. Upon exposure completion,
fry were transferred to 18.5-L growout aquaria
until sexual maturity so secondary sex characteristics
could be observed. After sex determination,
fish were terminally anesthetized with MS-222
and discarded.
Real-time quantitative RT-PCR. Ovarian
(cyp19a) and brain aromatase (cyp19b)
expression was measured using real-time quantitative
reverse-transcriptase polymerase chain reaction
(RT-PCR). Due to lack of detection of ovarian
aromatase with real-time PCR, further examination
of cyp19a was performed using multiple
primer pair with visualization of expression
using both real-time methods and 2% agarose
gel/ethidium bromide. Total RNA was extracted
from whole fry using a Trizol procedure and
purified with a phenol: chloroform extraction
followed by an ethanol precipitation. Total
RNA concentration was measured using a Beckman
(Fullerton, CA) DU640 spectrophotometer and
treated with DNase H (Invitrogen, Carlsbad,
CA) to remove genomic DNA contamination. cDNA
was synthesized from 1 µg total RNA using
Superscript II reverse transcriptase from Invitrogen
and random decamers. Real-time PCR was accomplished
using Taqman chemistry (Heid et al. 1996).
Table
1
 |
Forward and reverse primers for
cyp19a and
cyp19b amplification
and dual dye-labeled FAM (6-carboxyfluorescein;
excitation, 490 nm; emission, 520 nm)-Black
Hole Quencher (BHQ) were designed from the
ovarian and brain aromatase sequence (Genebank
accession nos. D82968 and AY319970) using Beacon
Designer 3.01 (PREMIER Biosoftware, Palo Alto,
CA) (oligo 1-7) (Table 1). 18S primers
designed from published medaka 18S sequence
and Cy5 (excitation, 596 nm; emission, 615
nm)-Iowa Black RQ dual-labeled Taqman
probes (Integrated DNA Technologies, Coralville,
IA) were used as internal normalization standard
(oligo 8, 9, 10) (Table 1). Integrated DNA
Technologies supplied probes, and we used a
Bio-Rad (Hercules, CA) IQ-Cycler real-time
PCR system to amplify and measure fluorescence
of aromatase and 18S. For the reactions in
the first exposure, conditions consisted of
100 nM probe, 900 nM primer for aromatase,
and 100 nM probe and 50 nM primers for 18S
for all sampling days. Primer concentrations
were tested to ensure equal amplification efficiency
between aromatase and 18S.
Due to differences in the ratio of 18S and
aromatase concentrations in fish collected
at each sample period in the second exposure,
we used different concentrations of probe and
primers to obtain equal amplification efficiencies.
Day 5 conditions consisted of 100 nM probe,
1,200 nM primer for aromatase, and 100 nM probe
and 35 nM primers for 18S. Day 9 conditions
were 100 nM probe, 1,200 nM primer for aromatase,
and 100 nM probe and 40 nM primers for 18S.
On day 14, no primer concentrations could be
determined that would express both aromatase
and 18S for all samples in multiplex with equal
amplification efficiency. Therefore, day 14
samples were measured in separate single-plex
reactions using Bio-Rad IQ Real-Time SYBRMix
with SYBRGreen. Multiplex reactions used Bio-Rad
IQ Real-Time Supermix according to manufacturer’s
instructions. Cycle parameters were 50°C
for 120 sec, 95°C for 120 sec, 50 cycles
of 95°C for 15 sec, and 61°C for 30
sec. Relative expression was calculated with
the comparative Ct (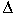
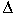 Ct)
method, which involves comparing the threshold
cycle (Ct) values of the
treated samples with the nontreated controls
(calibrator). The Ct values
of both the calibrator and the treated samples
are then normalized to the endogenous housekeeping
gene 18S. Gene expression for each sampling
time is expressed as fold increase over same-day
control.
Ct)
method, which involves comparing the threshold
cycle (Ct) values of the
treated samples with the nontreated controls
(calibrator). The Ct values
of both the calibrator and the treated samples
are then normalized to the endogenous housekeeping
gene 18S. Gene expression for each sampling
time is expressed as fold increase over same-day
control.
Aromatase activity. Aromatase
activity was measured by a tritiated water
release assay based on the work of Thompson
and Siiteri (1974) as adapted to medaka by
Melo and Ramsdell (2001) and Contractor et
al. (2004). Whole medaka fry sampled during
exposure were homogenized in phosphate buffer
(1 M KCl, 0.01 M K2HPO4,
and 0.001 M EDTA, pH 7.4). Protein concentration
of homogenate was determined using a bicinchoninic
acid protein assay kit (Pierce, Rockford, IL).
Homogenate containing about 20 mg of protein
was incubated with 5 nM androst-4-ene-3,17-dione
[1β-3H(N)]
(Perkin Elmer, Boston, MA) in a 200 µL
solution of 1 mM NADPH, 10 mM glucose-6-phosphate,
1 U/mL glucose-6-dehydrogenase, 10 mM potassium
phosphate (dibasic), 1,000 mM potassium chloride,
1 mM EDTA, and 1 mM dithiothreitol at 37°C
for 3 hr. After incubation, reactions were
terminated by immersion in ice-cold water and
adding 100 µL 30% trichloroacetic acid,
and centrifuged at 1,700g for 10 min
to remove precipitated protein. Unconverted
substrate was removed by vortexing vigorously
for 60 sec with 1 mL chloroform followed by
centrifugation at 1,700g for 25 min
at 4°C. Addition of a 5% charcoal/0.5%
dextran slurry followed by a 40 sec vortex
and 30 min centrifugation (10,000g)
was used to remove any residual androst-4-ene-3,17-dione.
Radioactivity of tritiated water was measured
in a Beckman LS6500 liquid scintillation counter
and background subtracted using samples without
homogenate. Sensitivity of detection was set
at two standard deviations above the mean blank
activity to be considered detectable.
Analytical chemistry. Magnolia
Scientific Services Inc. (Hattiesburg, MS)
measured o,p´-DDT and TBT. o,p´-DDT
was measured according to U.S. Environmental
Protection Agency (EPA) method 608 (U.S. EPA
1988), and TBT was measured according to U.S.
EPA method 282.3 (U.S. EPA 1989).
We determined FAD concentration using reverse-phase
high-pressure liquid chromatography (HPLC)
with photo diode array detection. Water samples
(~ 5 mL) were collected twice weekly in glass
vials and injected directly as 1.0 mL aliquots
onto a 4.6 mm  25
cm Beckman Ultrasphere C18 reverse-phase column
connected to a Beckman Gold HPLC system. Samples
were chromatographed using a gradient program
with a mobile phase starting at 60% methanol/40%
50 mM phosphate buffer (pH 7.0) for 4 min and
increased to 80% methanol/20% buffer over 2
min, where it was held for 9 min. Mobile phase
was then returned to 60% methanol/20% phosphate
buffer to prepare the column for the next sample.
FAD in column eluate was detected using a Beckman
System Gold 168 photodiode array detector set
at 229 nm. A five-point standard curve was
developed with FAD dissolved in well water
for sample quantification. Samples were measured
in duplicate in conjunction with standards.
Limit of detection was approximately 5 µg/L.
25
cm Beckman Ultrasphere C18 reverse-phase column
connected to a Beckman Gold HPLC system. Samples
were chromatographed using a gradient program
with a mobile phase starting at 60% methanol/40%
50 mM phosphate buffer (pH 7.0) for 4 min and
increased to 80% methanol/20% buffer over 2
min, where it was held for 9 min. Mobile phase
was then returned to 60% methanol/20% phosphate
buffer to prepare the column for the next sample.
FAD in column eluate was detected using a Beckman
System Gold 168 photodiode array detector set
at 229 nm. A five-point standard curve was
developed with FAD dissolved in well water
for sample quantification. Samples were measured
in duplicate in conjunction with standards.
Limit of detection was approximately 5 µg/L.
Statistical analysis. We compared
treatments and controls for percent survival
with the chi-square test after transformation
of percentages by the arcsine square root procedure.
Deviations from a 1:1 sex ratio were analyzed
by the replicated goodness-of-fit test (G-test)
followed by the unplanned test of the homogeneity
of replicates. In treatments were no males
remained after exposure, we added 0.05 to values
for all males and females for all treatments
so that the natural logarithm could be calculated.
Throughout all experiments, each fish was treated
as a replicate for both aromatase activity
and cyp19b expression (n = 6)
because space for individual experimental units
was limited. We compared cyp19b expression
and aromatase activity among treatments by
one-way analysis of variance (ANOVA). A Kolmogorov-Smirnov
one-sample test was used to test for normality,
and Levine’s test was used to test for
homogeneity of variance. If data failed either
test, the we used the nonparametric Kruskal-Wallis
ANOVA to examine differences. If significant
difference among groups was observed, a Dunn’s
multiple comparisons test on each day separately
was used to determine where significance occurred.
Statistical significance was accepted at p < 0.05.
All statistical analyses and graphing were
completed using Sigmastat 3.1 (Systat Software,
Inc., Point Richmond, CA) and SPSS 11.5 (SPSS,
Inc., Chicago, IL).
Table
2
 |
Table
3
 |
Table
4
 |
Table
5
 |
Juvenile medaka exposure. In
the first exposure, juvenile medaka were
exposed in a flow-through system to FAD and
TBT for
2 weeks beginning at hatch. Measured concentrations
of the test chemicals were approximately
90-141%
of nominal concentrations. Survival ranged
from 51 to 63%, and according to the goodness-of-fit
chi-square test, no significant difference
in survival between controls and exposed
groups existed (Table 2). Developmental exposure
to
FAD and TBT did not significantly alter adult
sex ratios at concentrations chosen. In d-rR
medaka, white males and red females indicate
a phenotypic sex inversion. At this exposure,
there were no sex inversions as evidenced
by the lack of any white males or red females
(Table 3).
In the second exposure, medaka were exposed
to FAD, TBT, and o,p´-DDT for
2 weeks beginning at hatch. Measured doses
ranged between 81 and 101% of nominal concentrations.
Survival ranged from 39 to 50%, and there was
no significant difference in survival between
controls and exposed groups (Table 4). According
to a goodness-of-fit G-test, developmental
exposure to FAD and TBT did not alter sex distributions
or induce sex inversion. However, o,p´-DDT
significantly altered sex distributions in
all o,p´-DDT exposures regardless
of inhibitor cotreatment (Table 5).
Aromatase expression. Gene
expression was measured in individual whole
fry on days 5, 9, and 14 for each treatment
and quantitated using the 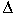
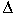 Ct method.
18S was used as the internal normalization
standard, and expression data for each time
point are expressed as fold change relative
to the mean of the same-day controls. Ovarian
aromatase was not detected in any sample through
measurement with real-time PCR or traditional
PCR. Further multiple primer sets were tested
in multiple conditions (data not shown). Therefore,
all following data represent brain aromatase
expression.
Ct method.
18S was used as the internal normalization
standard, and expression data for each time
point are expressed as fold change relative
to the mean of the same-day controls. Ovarian
aromatase was not detected in any sample through
measurement with real-time PCR or traditional
PCR. Further multiple primer sets were tested
in multiple conditions (data not shown). Therefore,
all following data represent brain aromatase
expression.

Figure 1. Effects of
FAD and TBT on brain aromatase expression
on day 5 shown as fold change relative
to same-day control as measured by
real-time PCR (mean ± SE).
*Significant difference (p < 0.05). |
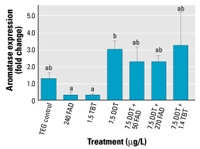
Figure 2. Effects of FAD, TBT,
and o,p´-DDT, as well as
FAD/DDT and TBT/DDT coexposure, on brain
aromatase expression on day 14 shown
as fold change (± SE) relative
to same-day control as measured by real-time
PCR. Bars with different letters are
significantly different from each other
based on Dunn’s multiple comparison
test (p < 0.05). |
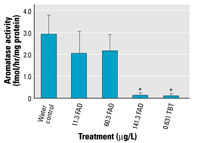
Figure 3. Effects of FAD
and TBT exposure on aromatase enzyme
activity (mean± SE, fmol/hr/mg
protein) on day 14. Values for 141.3
FAD and 0.631 TBT were < LOD.
*Significant difference (p < 0.05). |
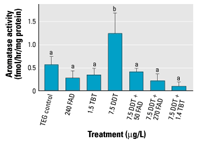
Figure 4. Effects of FAD, TBT, o,p´-DDT,
FAD/DDT, and TBT/DDT on aromatase enzyme
activity (mean ± SE, fmol/hr/mg
protein) on day 14. Bars with different
letters are significantly different from
each other (p < 0.05). |
In the first exposure, day 9 samples were
lost because of defective extraction reagent.
A Kruskal-Wallis ANOVA demonstrated that brain
aromatase expression levels showed a significant
decrease in the TBT treatment versus control
treatment at sampling day 5 (Figure 1). However,
this significance was lost by sampling day
14 (data not shown).
In the second exposure, the day 14 sampling
period demonstrated a significant difference
in cyp19b expression between treatments
according to a Kruskal-Wallis ANOVA. Pairwise
multiple comparisons between treatments revealed
that this difference is between FAD- and TBT-only
treatments and DDT only (Dunn’s method, p < 0.05).
Fish exposed to FAD- and TBT-only treatments
also had lower expression levels than those
exposed to the AI and o,p´-DDT
cotreatments; however, these differences are
not significant (Figure 2). Day 5 and day 9
sampling periods show similar trends but are
not significant (results not shown).
Aromatase activity. Aromatase
activity was measured using a tritiated water
release assay. In the first exposure, assay
sensitivity was 1.49, 1.20, and 0.86 fmol/hr/mg
protein for sampling days 5, 9, and 14, respectively.
The highest concentration of FAD and TBT treatment
levels consistently resulted in aromatase activity
levels below level of sensitivity for this
assay and can be considered nondetectable (Figure
3).
In the first experiment, changes in activity
followed the same pattern as changes in gene
expression on all sampling days, with decreasing
activity at increasing concentrations of FAD
and in the TBT treatment. Although day 5 and
day 9 showed no significant difference between
any treatments (results not shown), day 14
showed a significant decrease in activity between
the controls and the highest concentrations
of FAD and TBT (Figure 3).
For the second exposure, the limit of detection
(LOD) was 2.30, 1.37, and 0.26 fmol/hr/mg protein
for days 5, 9, and 14, respectively. Only the
control treatment had measurable levels of
aromatase activity on day 5, and all day 9
samples were < LOD. On day 14, only the o,p´-DDT/high
FAD and o,p´-DDT/TBT cotreatments
had enzyme activity levels below assay sensitivity
(Figure 4).
Aromatase activity demonstrated a pattern
different from aromatase expression. Unlike cyp19b expression
data, which showed an increase in aromatase
expression in all o,p´-DDT treatments
regardless of presence of inhibitors, aromatase
activity showed an increase only in the o,p´-DDT
control treatment (Figure 4). A Kruskal-Wallis
test demonstrated a significant difference
in aromatase activity between treatments on
day 14. Pairwise multiple comparison procedure
revealed that the o,p´-DDT-only
treatment is significantly different from all
other treatments. The cotreatments of o,p´-DDT
and AIs had reduced or nondetectable aromatase
activity with no corresponding decrease in
aromatase gene expression. However, this decrease
in activity was not significant. This trend
was not observed on sampling day 5 or day 9
(results not shown).
Most laboratory research to date on EDCs
has focused on the effects of a single compound
on an organism. However, in the environment,
organisms are exposed to complex mixtures of
potentially synergistic and antagonistic compounds,
and it is unknown how these chemicals interact in
vivo and whether such interactions diminish
or exacerbate their individual effects on the
health of the organism. To begin addressing
these questions, our objectives in the present
study were 2-fold: to examine the role of brain
aromatase in o,p´-DDT-induced
sex reversal in medaka and to determine if
an environmentally relevant mixture of both
estrogenic (o,p´-DDT) and apparent
antiestrogenic (TBT) chemicals, which have
opposing effects on aromatase activity, can
block each other’s effects on reproductive
development. Extensive experiments showed that
ovarian aromatase transcripts could not be
detected in medaka fry (0-14 dph) by
real-time or traditional RT-PCR. Further, ovarian
aromatase could not be detected upon stimulation
of the estrogen response system after exposure
to estradiol and o,p´-DDT. It
appears, therefore, that the brain isoform
of aromatase accounts for most, if not all,
estradiol production during this life stage
of the medaka and that the aromatase activity
measured in this study represents the brain
form of this enzyme.
To accomplish our stated objectives, we exposed
juvenile medaka to an environmental estrogen
in the presence and absence of two AIs and
examined the effects on aromatase expression
and activity and sex reversal. After a 2-week
exposure, all fish developed female secondary
sex characteristics regardless of the presence
of AIs and independent of levels of brain aromatase
activity, indicating that an increase in brain
aromatase activity, as observed with o,p´-DDT
exposure only, is not required for a male-to-female
sex reversal resulting from exposure to an
environmental estrogen. The results of the
study presented here also show that the effects
(feminization) of an environmental estrogen
(o,p´-DDT) are not negated by
the antagonistic effects of an environmental
antiestrogen (TBT) at the exposure conditions
and developmental stage examined. Whether the
results of this specific case are of general
validity for mixtures with predicted antagonistic
effects deserves further study.
Additionally, results from this study demonstrate
that immersion exposure to AIs alone did not
result in any female-to-male inversions, even
though aromatase activity was inhibited. These
results contrast with several published studies
in which inhibition of aromatase by both pharmaceutical
(FAD) and environmental (TBT) AI exposure (McAllister
and Kime 2003; Uchida et al. 2004) can result
in masculinization of several fish species,
including Japanese flounder, zebrafish, and
salmon (Kitano et al. 2000; Kwon et al. 2000;
Piferrer et al. 1993; Uchida et al. 2004).
Masculinization occurs concomitantly with a
decrease in aromatase expression, suggesting
that manipulation of the aromatase system during
development can alter gonadal differentiation
(Fenske and Segner 2004).
These studies on the role of aromatase in
sex reversal during development support the
theory that androgens and estrogens are the
natural sex inducers in fish and play pivotal
roles in sex differentiation (Devlin and Nagahama
2002). Other evidence such as sexually dimorphic
expression of aromatase in developing zebrafish
(Trant et al. 2001) and the ability of exogenous
steroid treatment to influence sex differentiation
(Yamamoto 1958) suggest the importance of sex
steroid production in the very early steps
in gonad differentiation. Also, in medaka,
inhibition of aromatase can result in the prevention
of ovarian cavity formation, suggesting the
importance of endogenous estrogen in gonadal
development (Suzuki et al. 2004).
However, other evidence exists that suggests
sex steroids may not play such an important
role. For example, in medaka, germ cell differentiation
appears to occur before somatic steroid-producing
cells are observed (Satoh 1974). Further, in
a study by Kawahara and Yamashita (2000), medaka
eggs incubated with an AI resulted in no abnormal
sex ratios. From these observations, the authors
concluded that female sex determination in
medaka is not estrogen dependent. They also
suggested that estrogen-independent activation
of the estrogen receptor may be a primary pathway
in female gonadal development. They emphasized
that the observations of female-to-male sex
inversion after treatment with AI in other
vertebrates and fish suggests a role of aromatase
only during the sex determination period and
not the importance of estrogen and aromatase
as the natural sex inducer in gonadal differentiation.
In the present study, exposure to AIs significantly
reduced aromatase activity between days 9 and
14 yet did not induce a sex inversion. This
could be because male sex differentiation is
irreversibly determined before day 9. However,
it is more likely that our treatment period
terminated too early, because male germ cell
proliferation does not occur until 14 dph (Satoh
1974). Sensitivity of sex differentiation to
endocrine disruptors has been shown to be dependent
on duration and dose of exposure and on developmental
stage. Insufficient exposure length or dosage
concentrations can result in lack of response
(Cheek et al. 2001b). The lack of response
to aromatase inhibition reported by Kawahara
and Yamashita (2000) may therefore be due to
the developmental period of exposure. In most
cases, the most sensitive period is just before
or at the same time as histologic differentiation
of the primitive gonad (Hunter and Donaldson
1983). In medaka, differentiation occurs at
hatch, with the proliferation of ovarian cells
beginning shortly after hatch (about 6-10
dph) (Satoh 1974). Medaka have been shown to
be sensitive to feminization by estradiol during
this proliferation period (Cheek et al. 2001a;
Nimrod and Benson 1998). Unlike female germ
cells, male germ cells cease to divide immediately
after hatching, and proliferation is delayed
until about the 9-10 mm larval stage
(about 14-20 dph) (Satoh 1974). This
is also the stage of development in which male
gonial cells first appear in the female gonad
upon androgen treatment (Kobayashi and Hishida
1985). Thus, lack of masculinization in response
to aromatase inhibition observed in this study
might be due to termination of exposure before
male germ cell proliferation begins.
In the present study, the inhibition of aromatase
did not induce a female-to-male inversion,
i nor did it prevent a male-to-female inversion
induced by o,p´-DDT. Several studies
have demonstrated a correlation between an
increase in aromatase expression and activity
upon exposure to environmental estrogens and
sex differentiation in fish (Chiang et al.
2001; Fenske and Senger 2004; Kitano et al.
2000; Scholz and Gutzeit 2000; Tanaka et al.
1995). Previous work in this laboratory (Kuhl
et al. 2005) also demonstrated the importance
of the aromatase system in fish gonadal development
by observing a significant 5-fold increases
in aromatase expression and activity at o,p´-DDT
concentrations that induce a male-to-female
sex inversion.
Results from the present study, however,
demonstrate that sex inversion can be induced
without a corresponding increase in aromatase
activity. Treatment with o,p´-DDT
alone induces both aromatase activity and brain
aromatase expression, while resulting in a
complete male-to-female sex inversion. However,
inhibition of aromatase activity in o,p´-DDT/AI-cotreated
fish also results in a complete male-to-female
sex inversion. In these fish, there is an increase
in brain aromatase expression while activity
is reduced to nondetectable levels. Altered
aromatase activity levels are therefore not
a requirement for sex inversion in fish.
The apparent lack of aromatase involvement
suggests that alternative, non-aromatase-dependent
pathways exist through which o,p´-DDT
may bring about sex reversal. For example,
exogenous estrogen treatment can also result
in reduction of expression of several steroidogenic
enzymes, including P450c17, 3βHSD,
and P45011β in
the differentiating testis (Govoroun et al.
2001). This would decrease the synthesis of
11-oxygenated androgen and may be an important
step in exogenous estrogen feminization. Further,
xenobiotics can induce steroid-metabolizing
enzymes, including steroid sulfotransferases,
steroid glucuronidation enzymes, and steroid
hydroxylases (You 2004). Because the ratio
of androgens to estrogens may be more important
in differentiating gonads than their absolute
values (Bogart 1987), xenobiotic alterations
of androgen levels may also play a role in
the cotreatments conducted in this study. Steroidogenic
enzyme levels and androgen to estrogen ratios
during cotreatments must be measured to examine
the possibility of this mechanism. Finally,
nongenomic actions mediated through membrane-bound
hormone receptors also can influence steroid
production. Loomis and Thomas (2000) demonstrated
that estrogens, and likely xenoestrogens including o,p´-DDT,
can cause a decrease in gonadotropin-stimulated
androgen production through a nongenomic mechanism
in the Atlantic croaker (Micropogonias undulatus).
This may further interfere with steroid production
and the ratios between androgens and estrogens.
In conclusion, the present study demonstrates
that the xenoestrogen o,p´-DDT
increases brain aromatase activity accompanied
by complete male-to-female sex inversion. Coexposure
to o,p´-DDT and AIs does suppress
aromatase activity but does not prevent sex
reversal. Thus, increased aromatase activity
is not necessary for sex inversion, and alternatively,
aromatase-independent pathways for sex reversal
resulting from xenobiotic treatment must exist.
This study shows that exposure to AIs during
days 0-14 fails to induce female-to-male
sex inversion. This suggests that testis differentiation
in d-rR medaka does not occur until after 14
dph. The observation that exposure to a mixture
of estrogenic and antiestrogenic compounds
does not block estrogen-induced sex reversal
suggests that, in the environment, exposure
to antagonistic EDCs may not necessarily lessen
the harmful impacts of these compounds.

