Despite the controversial nature of multiple chemical
sensitivities (MCS)/environmental sensitivities (ES),
the following consensus criteria (Bartha et al. 1999)
have been proposed to define MCS/ES: The symptoms are
reproducible with repeated chemical exposure; the condition
is chronic; low levels of exposure (lower than previously
tolerated) result in manifestations of the syndrome;
the symptoms improve or resolve when the incitants
are removed; responses occur to multiple chemically
unrelated substances; and symptoms involve multiple
organ systems. These criteria are based upon previous
definitions (Cullen 1987; Nethercott et al. 1993; Randolph
1961).
Some researchers consider another term, “idiopathic
environmental intolerances” (IEI), more appropriate
[International Programme on Chemical Safety (IPCS)
1996] and believe it should be restricted to individuals
with absolutely no abnormalities except a self-reported
abhorrence to chemicals. Many people with chemical
sensitivities have other conditions and symptoms such
as asthma, rhinosinusitis, dermatitis, and headaches
triggered by chemical exposures and would not meet
this restrictive definition of idiopathic environmental
intolerances. Our previous study (Joffres et al. 2001)
identified a common set of consistent symptoms that
follow exposure: difficulty in concentrating, fatigue,
forgetfulness, irritability, itchy or burning eyes,
sneezing, and hoarseness or loss of voice. The prevalence
of ES is not negligible (Kreutzer et al. 1999; Meggs
et al. 1996), and physicians, even those most familiar
with these conditions, often overlook such a syndrome
(Kutsogiannis and Davidoff 2001). Questions remain
unanswered regarding the etiology, associated mechanisms,
and inconsistency in pattern of symptoms. Several reports
(Fiedler and Kipen 1997; Kipen and Fiedler 2002a, 2002b;
Sparks et al. 1994) have outlined some of the different
viewpoints on etiology and physiopathology. Meggs (1995)
and Bascom et al. (1997) have proposed a neurogenic
inflammation model. Sensitization or kindling of olfactory-limbic
pathways by acute or chronic exposure has also been
proposed (Bell et al. 1997, 1999; Miller 2001). Researchers
for the IPCS (1996) proposed that the key experiment
is to determine in a double-blind challenge study whether
subjects with “IEI” successfully discriminate
between exposures to environmental factors (including
to which illness is attributed) and placebo. If the
subjective response (appearance of symptoms) of test
subjects is able to discriminate between exposure to
test chemicals and placebos, in a blinded design, this
would suggest the operation of a toxicological mechanism
in which culpable agents interact with tissue targets
to trigger a receptor-mediated pathophysiological response.
A few studies have attempted to look at experimental
testing of affected individuals (Fiedler et al. 2000;
Fiedler and Kipen 2001; Rea et al. 1991; Staudenmayer
et al. 1993). A critical factor that may have been
overlooked in some of these studies is the adaptation
of subjects to baseline study protocols. Adaptation
is defined here as the period taken by the subjects
in a study to get used to the general study protocols,
obtaining stable physiologic (i.e., skin conductance
and symptomatic) readings before the actual introduction
of substances. We realized that with our patient population
expressing a high level of reactivity, we would need
to get stable readings before introducing any test
substance. Otherwise, we might obtain erratic physiologic
or symptomatic responses because of testing conditions,
which would make it impossible to differentiate challenge
substances from placebos. Surprisingly, we have not
found any psychophysiologic literature discussing the
importance of stabilizing readings before starting
experiments other than the usual caution of a few minutes
of relaxation. Most studies have either used a set
period for adaptation (Fiedler et al. 2000) or not
considered this factor at all (Staudenmayer et al.
1993).
Therefore, we hypothesized that individuals with
ES may require more time to adapt to the experimental
conditions compared with controls without chemical
sensitivities. Also, we hypothesized that each individual
may have a different adaptation period. We also wanted
to explore the type of measure that would be the most
useful to detect change and therefore included several
psychophysiologic measures.
Addressing the issue of symptom development has also
been critical to research on these illnesses. There
is a need to see how symptoms develop and what triggers
are associated with symptoms in individuals with ES.
Is there a relation between symptoms and skin conductance
response during exposure to chemicals? Estimating the
level of reactivity of cases and controls to the substances
was another major objective of this pilot study.
Hence, to answer these questions, we conducted a
pilot, blinded, controlled booth study at the Nova
Scotia Environmental Health Centre, a government-funded
facility dedicated to research and management of individuals
with ES.
Inclusion criteria. Subjects with sensitivities
were selected from the last 50 new patients enrolled
at the Nova Scotia Environmental Health Centre who
fulfilled the consensus criteria (Bartha et al. 1999)
and who gave informed consent to participate in the
study. The Dalhousie University Health Sciences Human
Research Ethics Board approved the study protocol.
Controls, without known chemical sensitivities, were
recruited from friends of the patients and from advertisement
in local churches.
Exclusion criteria. Subjects were
excluded from the study if they had any other major
illnesses such as cancer (outside of skin cancer or
past cancer without relapse in the preceding 5 years),
insulin-dependent diabetes, stage 2 or 3 hypertension
(systolic/diastolic blood pressure: stage 2, 160-179/100-109;
stage 3, > 180/> 110), history of myocardial
infarction, angina pectoris, stroke, or psychiatric
disorders such as major depressive disorder, schizophrenia,
shared psychotic disorder, dementia, or drug dependence.
A total of 12 cases (all women; mean age, 40 years;
range, 25-60 years) and 7 controls (all women; mean
age, 42 years; range, 26-59 years) gave an informed
consent to participate and fulfilled the initial inclusion/exclusion
criteria of the study. The subjects were matched within
age, education, and ethnicity categories.
 Figure
1. Challenge booth and testing conditions. Figure
1. Challenge booth and testing conditions.
|
Challenge booth. The study was conducted
in a dedicated room that included the challenge booth
(Figure 1) made from inert materials with no apparent
odors, allowing introduction of materials that release
various chemicals into the environment. The booth is
a glass room with steel framing with dimensions of
2.2 m (7.2 ft) in height by 1.2 m (4 ft) in width by
1.8 m (6 ft) in depth. The construction materials and
procedure used have received careful consideration
toward making it suitable for research work on individuals
with chemical sensitivities. The airflow, temperature,
and lighting in the facility can be varied to increase
the comfort level of the subjects. A side box attached
to the booth was used to introduce chemicals into the
air stream. The door to the side box opens on one side
of the booth so the subjects are unaware of the substance
being introduced into the booth. Air is allowed to
enter the side box into an inlet provided on the frosted
side of the booth. The inlet height is at the breathing
level of the occupant. The air entering the booth is
100% outdoor air that has been filtered before being
distributed throughout the clinic, not recirculated,
and is then directly vented to the outdoors through
an exhaust located in the ceiling of the booth.
Test substances. Test substances for
the study were those commonly reported by affected
people to cause reactions: common glue, a scented body
wash solution, dryer sheet, and for control substances,
unscented shampoo and clean air. These substances were
contained in a closed metal box, introduced through
the side box, and passively released in the booth airflow.
A new dryer sheet was used for each session. Patients
could not smell the substance because of nose plugs.
Outcome measures. Before and after
each booth session, subjects answered a symptom questionnaire
on symptoms reported by Joffres et al. (2001; e.g.,
eye irritation, throat irritation, sleepiness, headache)
as the top symptoms experienced by our patient population
after an exposure. The questionnaire measured irritation
on an ordinal scale of 0-10, with 0 being barely detectable
and 10 being strongest detectable (Joffres et al. 2001).
The booth environment (light, sound, and temperature)
was also rated before and after each session on a scale
of 0-10 (0 = poor, 10 = excellent). The purpose of
the adaptation sessions was to reduce the number and
severity of symptoms reported to the nurse among reactive
subjects. The adaptation sessions identified symptomatic
responses shown by subjects to the baseline protocols
of the study such as wearing nose plugs, wearing respiratory
belt, and the other BIOPAC (BIOPAC Systems Inc., Goleta,
CA, USA) measures.
During the testing period, we compared pre- and postsymptom
scores from that day, and coded any change in score
or type of symptom as a positive response. We disregarded
the symptoms that occurred consistently during the
adaptation sessions (baseline symptomatic responses)
while computing a response toward challenge substances.
After the booth session, subjects recorded and reported
to the nurse any changes they observed during the next
8-hr period. They were instructed not to visit malls
or other similar places where they may be exposed to
other substances 8 hr before and 8 hr after each booth
session. Because many of our patients reported effects
lasting up to 4 or 5 days after a booth session, subjects
were exposed to only one substance at a time and had
a minimum of 1 week between two booth sessions.
We used the Brown-Peterson test (Peterson and Peterson
1962) to test a variation in the short-term memory
span of the subjects after a booth exposure. Subjects
were given a series of trigrams of letters; each trigram
was followed by a number countdown challenge, after
which subjects were asked to recall the trigram. Short-term
memory for 9, 18, and 36 sec intervals was examined.
The test was conducted before and after each exposure
session.
Contrast sensitivity (Schreiber et al. 2002), which
provides a detailed assessment of spatial vision and
is sometimes recommended as a test to screen visual
damage caused by chemical exposures such as solvents,
was tested before and after each session. Peak flow
was used as a pre- and postsession measure to determine
impairment in lung function. All these measures were
recorded pre- and postsession during all booth sessions
for each subject.
Physiologic measures. To assess physiologic
measures of skin temperature, skin conductance, respiratory
rate, heart rate, and surface electromyography (EMG),
we used used the BIOPAC MP 100 data acquisition system
(BIOPAC Systems Inc.) during each booth session. Surface
EMG was collected at a rate of 1,000 samples/sec using
the Biopac electrodes placed at the upper trapezius
muscle. Disposable electrocardiogram electrodes were
used to acquire heart rate at a sampling rate of 1,000
samples/sec. The positive electrode was placed on the
right wrist, the negative on the left wrist, and the
ground electrode on the right ankle. Finger temperature
and skin conductance were measured at a rate of 3 sample/sec.
Of the physiologic measures, only skin conductance
was a consistent indicator of adaptation and response
to challenge substances postadaptation. Results and
discussion in this article are therefore restricted
to this physiologic measure.
Skin conductance recording and analysis. We
recorded skin conductance using Ag/AgCl electrodes
filled with isotonic electrolyte jelly and attached
to the fore and middle fingers of the left hand. Subjects
were asked to wash their hands with lukewarm water
before the start of each booth session. Skin conductance
data acquisition and analysis were conducted using
Acknowledge 3.2.4 software (BIOPAC Systems Inc.). The
raw data collected were first smoothed using low-pass
filter. The readings were compared with the baseline
readings from that day.
Skin conductance response has been described in the
literature as having two components: phasic and tonic
responses (e.g., Lim et al. 1997). Phasic responses
may be evoked even by a discrete stimulus such as subtle
variations in environment or even thought processes.
Tonic skin conductance response is the baseline level
of skin conductance in theabsence of any stimulus.
This is known to vary with time in the presence of
a stimulus depending upon the psychological state of
the individual and their autonomic regulation. We considered
only the tonic responses while assessing a positive
response to challenge substances in our pilot study.
Recordings showed variations in the level of conductance
because of artifacts or other factors, and the change
in amplitude and the length of the tonic response cannot
be easily used in a continuous form without arbitrary
criteria about where and how the measures will be made.
Therefore, we adopted a simple criterion that could
be easily reproduced: Tonic responses were considered
positive if there was a change in amplitude from the
preexposure period (of the session) by at least 0.5
microsiemens (µS) about 20 sec after the introduction
of the substance.
Booth session protocol. In an
orientation visit we discussed details of the study
with the subjects and answered questions about the
study. After consent, an adaptation period allowed
subjects to get used to baseline study protocols, such
as cognitive testing (Brown-Peterson test), answering
questionnaires, getting used to the booth, and wearing
nose plugs.
 Figure
2. Time line for booth session. Sequence of changes:
1 and 2, opening and closing of side door of
booth; 3, exhaust fan on low speed; 4, opening
and closing of side door of booth; 5, exhaust
fan on high speed; 6, exhaust fan off. Figure
2. Time line for booth session. Sequence of changes:
1 and 2, opening and closing of side door of
booth; 3, exhaust fan on low speed; 4, opening
and closing of side door of booth; 5, exhaust
fan on high speed; 6, exhaust fan off.
|
Each subject was given up to 10 individual booth
sessions with a maximum of four sessions for adaptation
to the baseline study protocols. Each booth session
consisted of the same set of changes occurring at the
same time, which included opening and closing of the
side door through which substances could be introduced
(2.5, 5, and 10 min), exhaust fan going on low speed
(7 min), high speed (11.5 min), and then being turned
off (12.5 min) (Figure 2). The stability in readings
was judged only by the stability of tonic skin conductance
responses (see above for criteria) and in reduction
of symptomatic responses (symptom scores) based on
the interview with the nurse.
After adaptation, the subjects were blindly challenged
to the test substances, clean air, glue, body wash
solution, and dryer sheet in a randomized sequence.
Subjects received test substances only if an “open
and close” door sequence (time, 2.5 min) in the
pretest period, also done during the adaptation phase,
did not elicit any change in conductance greater than
our defined threshold of 0.5 µS approximately
20 sec after the introduction of the substance. The
subjects received only one challenge substance (placebo,
control, or test substances) in a session. Each subject
was retested to at least one substance that they reacted
to in a randomized sequence. Even if they did not react
to any substance (as in the case of our control subjects),
they were still retested on at least one substance
to confirm their nonreactivity.
 Figure
3. Number of sessions required for each individual
to reach stable baseline skin conductance readings
(adaptation) among (A) cases and (B) controls.
Subjects 3 and 9 (black bars) did not adapt after
four sessions. Figure
3. Number of sessions required for each individual
to reach stable baseline skin conductance readings
(adaptation) among (A) cases and (B) controls.
Subjects 3 and 9 (black bars) did not adapt after
four sessions.
|
Of the 12 subjects with ES, two did not adapt to
the baseline protocols and were excluded from further
study. All seven controls completed the adaptation
phase and were able to participate in the next phase
of the study (Figure 3).
This pilot study was single blinded. Subjects were
not aware of what substance was introduced and could
not smell the substance because of nose plugs. The
nurse monitoring subjects was not aware of what was
being introduced into the booth. The researcher introducing
the substance also monitored data recording and analyzed
the results and was separated from the nurse and the
subject by a partition. The order of administration
of the three test substances (glue, body wash lotion,
dryer sheet) and a control substance (unscented shampoo)
was randomized using a table of random numbers.
Data analyses. Heart rate variability
was analyzed by the Institute of Heart Math (McCraty
et al. 1995) and did not differ between test and control
substances or between patients and controls. Respiratory
rate showed erratic patterns that seemed to be influenced
by presence of the nose plug, and we could not identify
any specific patterns.
In addition to skin conductance, symptom scores before
and after booth sessions were the only other measure
that indicated completion of an adaptation period and
responses to challenge period. No other measures are
discussed in this article. Data were dichotomized into
reaction versus no reaction to simplify presentation
and allow the study to be easily reproduced.
We used SAS (version 9.1; SAS Institute Inc., Cary,
NC, USA) for the statistical analyses. Fisher’s
exact test statistic (two sided) was used to test differences
in proportion using an alpha level of 0.05 between
cases and controls, and McNemar’s statistic was
used for paired data (between placebo and substance
among cases and among controls). Because one column
in the 2  2
table had 0 frequencies, we used a frequency of 0.001
to be able to estimate a p-value.
2
table had 0 frequencies, we used a frequency of 0.001
to be able to estimate a p-value.
Table
1

|
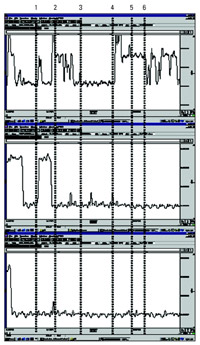
Figure 4. Example of tonic skin conductance
responses observed in a case during adaptation,
by stimuli and by session. Sequence of changes:
1 and 2, opening and closing of booth side
door; 3, exhaust fan on low speed; 4, opening
and closing of booth side door; 5, exhaust
fan on high speed; 6, exhaust fan off.
|

Figure 5. Percentage of individuals with ES presenting
responses to challenge substances, by type of symptom
and substance (sub). |
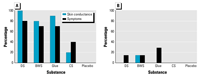
Figure 6. Percentage of (A) cases and (B) controls
presenting skin conductance or any symptom response
by test substance. Placebo was clean air. Abbreviations:
BWS, body wash solution; CS, control substance;
DS, dryer sheet. |
Table 2 |
Table 3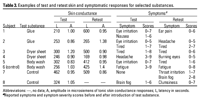
|
Table 1 presents the measures that were collected
during the booth sessions, and Figure 2 shows the
timeline for the different sequences of the preexposure,
exposure
period, and postexposure period.
Figure 3 shows skin conductance responses of cases
and controls to baseline study protocols. The proportion
of cases reacting to the different testing conditions
(different sounds) using set criteria described above
decreased with the number of sessions and was much
higher in cases than in controls. In the ES group,
83% (10 of 12) adapted after four sessions, whereas
17% (2 of 12) did not adapt after four sessions. Most
of the controls adapted in a single session (86%, 6
of 7).
An example of a tonic skin conductance response during
the adaptation period is presented in Figure 4 by stimuli
and by session. Although there were variations in skin
conductance after the different stimuli in the first
and second sessions (first and second window), there
were no longer tonic responses in the third session.
Figure 5 presents the percentage of individuals
with ES having a specific symptomatic response to each
of three challenge substances: glue, body wash solution,
and dryer sheet. The most common type of reaction was
burning eyes and headaches after exposure to the dryer
sheet and glue.
The percentages of cases and controls presenting
a skin conductance response or any specific symptom
to the test substance are shown in Figure 6. The level
of response was higher for all test substances in cases
than in controls and higher for test substances (glue
and dryer sheet) than for control substances (unscented
shampoo and clean air) in cases. There was a relatively
close match between physiologic and symptomatic responses
during exposure to challenge substances. Only one control
displayed a response in skin conductance to a test
substance, and two controls showed symptom responses
to test substances; none showed symptom responses to
the control substances.
The most significant difference in skin conductance
between cases and controls was for dryer sheets (p < 0.0001),
followed by glue (p = 0.0004) and body wash
solution (p = 0.02). In cases, comparing skin
conductance between substances and control substances,
there were similar patterns showing statistical differences
between control substance and dryer sheets (p =
0.0007), glue (p = 0.006), and body wash solution
(p = 0.02).
For symptomatic responses, there were no statistically
significant differences among cases between any substances
and the control substances, but there were statistically
significant differences between cases and controls
for the dryer sheet (p = 0.02) and for glue
(p < 0.05). There were no statistically
significant differences among controls.
Table 2 shows symptomatic and skin conductance test-retest
results in selected cases and controls by subject and
by substance. Table 3 presents all our test-retest
data on selected individuals. There was an overall
good agreement between test and retest for both symptoms
and skin conductance responses (McNemar’s test, p =
0.32 and p = 0.33, respectively).
The purpose of this pilot study was to shed some
light on the experimental conditions, substances, and
measures that could lead to better trials in chemically
sensitive individuals. There was a clear difference
in the time taken by cases to adapt to the experimental
conditions, and two cases did not adapt after four
sessions; however, more than half adapted after the
second session, and six of seven controls adapted in
only one session. We have not found any data that emphasize
the need for an adaptation period in studies that are
measuring physiologic responses outside of the few
minutes used to stabilize readings. There is also no
mention of the importance of stabilizing the reactions
to the experimental conditions such as being observed,
changes in temperature and airflow, and sounds such
as opening and closing a door to introduce a substance.
Although adaptation may not be an important issue in “normal” people,
it is certainly an issue in people with ES who are
usually in a state of hyper-reactivity. Reactions to
test substances need to be differentiated from the
effect of the experimental conditions. Otherwise, the
experimental “noise” will result in misclassification,
reducing statistical significance and resulting in
negative findings.
Because we removed most of the noise due to the experimental
conditions, the ability of individuals to detect the
test substances, compared with placebo and compared
with controls, raises several questions. First, EMG,
heart rate, respiratory rate, skin temperature, cognition,
and contrast sensitivity did not show any consistent
patterns of reaction. Therefore, these measures may
not be very sensitive or relevant to the pathophysiology
of reactions in individuals with ES. Because this was
a pilot study, a larger sample size might have shown
some more subtle differences, and therefore we cannot
totally reject the usefulness of these measures in
their ability to discriminate between test and control
substances.
Second, differences in skin conductance response
bring into question what type of reaction takes place
after exposure to a very low-level substance. Skin
conductance responses have been widely used in the
psychosocial field. Since the work of Edelberg (1972),
research has shown that changes in skin conductance
(or, conversely, resistance) are affected by the filling
of sweat ducts and the number of sweat glands affected.
This mechanism, under the autonomic nervous system
control, seems cholinergic, regulated by the premotor
cortex, the hypothalamus and limbic systems, and the
reticular formation. The role of the hypothalamus/limbic
system has been previously suggested in the pathophysiology
of MCS (Bell et al. 1997). Nevertheless, as Bell and
colleagues pointed out, there have been no controlled
experiments to determine whether or not sensitization
to low-level chemical exposures occurs in MCS patients.
Third, because the hypothalamus/limbic system is
closely linked with emotions, is it possible that the
perception of chemicals through the eyes (Millqvist
et al, 1999) or the upper respiratory system (Shusterman
2002) provoked a rise in anxiety, activating the autonomic
nervous system, resulting in a skin conductance response?
In future experiments, we will be studying patterns
of reactions to see if we can differentiate anxiety
responses from the type of response observed in this
study.
Fourth, in contrast to a pure anxiety response hypothesis,
it could be argued that irritation of these chemosensitive
structures (eyes and respiratory tract) could lead
to neurogenic inflammation as hypothesized by Meggs
(1995) and Bascom et al. (1997). Stimulation of the
glossopharyngeal and vagal nerves via the hypopharynx
and larynx could result in the type of symptoms described
by patients and the observer. Most cases reported burning
eyes, eye irritation, headaches, or sleepy or drowsy
feelings, which fit with our previous study of symptoms
after exposure in chemically sensitive patients (Joffres
et al. 2001).
Some of our patients were not able to adapt to baseline
protocols, showing erratic responses to the testing
conditions. These subjects are therefore in a state
of hyperreactivity and should not be included in such
experiments. Some cases reacted to the unscented shampoo
control substance, but none reacted to clean air. This
suggests that clean air should be used as the control
substance.
In subjects who adapted to the experimental setup,
the most irritant substances (dryer sheets and glue)
triggered a physiologic (skin conductance) response
accompanied with symptomatic responses in many cases
(Figure 6). When we looked at whether symptoms preceded
or followed the skin conductance reaction, we found
that most symptoms occurred at the same time or followed
rather than preceded electrodermal response. Only 20%
of subjects had symptoms preceding changes in skin
conductance.
Several studies have shown that undetected chemicals
can still induce brain activations (Lorig 1994; Sobel
et al. 1999). Staudenmayer et al. (1993) used an olfactory
masker with the test substances and as a control substance.
Therefore, if the olfactory masker is not perceived
by the sense of smell but is still able to alter neurophysiology,
there should be no detectable differences between test
and control substance (olfactory masker) because of
the added noise of the olfactory masker. If patients
had adapted to the olfactory masker, they might have
been able to detect a difference, but the study protocol
did not take this problem into account. Lorig (1994)
and Sobel et al. (1999) also raised the question of
why our controls did not detect the active substances.
Perhaps they did not because we considered only tonic
skin conductance responses in our study. All but one
of our control subjects displayed only phasic responses,
whereas almost all the cases showed tonic variations.
We will be looking at the importance of phasic changes
in our next study. In addition, we did not use measures
as sensitive as the electroencephalogram. The pilot
study has also helped us identify skin conductance
ranges in which a specific type of response, phasic
or tonic, is displayed if a stimulus is perceived as
a stressor. This will be critical for such studies
because these ranges will help us identify what type
of response can be considered given a specific range
of baseline. We will be confirming this with a larger
sample size.
Reaction times varied by substance and were fastest
with the dryer sheet, where most of the individuals
(8 of 10) reacted in < 200 sec after the introduction
of the substance. It is therefore important to allow
enough time to observe a response and not introduce
other substances that could create experimental noise.
Introducing more than one challenge substance in one
session, sham exposures, or maskers may not allow isolation
of the delayed responses that we observed in a few
cases for substances other than the dryer sheet.
Test-retest performed on five cases reacting to glue
showed that in four cases the level of reaction was
higher in the retest session (similar trends were also
observed while retesting on two other test substances),
and this fits with Bell et al.’s (1997, 1999)
theory regarding sensitization of olfactory-limbic
pathways and what Sorg and Newlin (2002) observed in
rats, where repeated chemical exposure produced sensitization
of the central nervous system circuitry. What was also
interesting in the Sorg and Newlin (2002) study was
that rats given repeated formaldehyde demonstrated
increased fear conditioning to odor paired with footshock,
suggesting amplification of neural circuitry guiding
fear responding to a conditioned odor cue.
Therefore, whether or not these reactions are triggered
by an unconscious anxiety response after the awareness
by the nervous system of a situation perceived as threatening
can still be argued. We tend to agree with Spurgeon
(2002) that the reporting of symptoms may result from
a complex set of interactions between aspects of personality,
attitudes, culture, and social climate as well as any
pathologic changes. Fiedler et al. (2000) noted that
reactions to specific substances in individuals do
not necessarily elicit physiologic responses. We noted
this in a few cases in our study as well. The fact
that, in other cases, the physiologic reaction was
not followed by any symptomatic response raises the
question of whether an anxiety response, recorded at
the unconscious level, could still result in such an
isolated physiologic reaction.
Symptomatic responses in sensitive individuals and
not in controls also correspond to Fiedler et al.’s
(2000) results. We are currently investigating whether
and how we can differentiate anxiety responses from
other types of responses using phasic and tonic responses.
Even if we assume an anxiety response, the question
remains of whether or not it is an initial, secondary
(conditioning), or mixed response to low-level chemical
exposure.
There are several limitations to this study. First,
because it was a pilot study, the number of cases and
controls was relatively small, and results need to
be confirmed with a larger study. Then, to ensure a
double-blind design, it will be necessary to have two
separate individuals, one doing the data analysis and
the other in charge of introducing the different substances.
In addition, the observer, the nurse, might have been
able to pick up the smell of some substances and give
unconscious nonverbal cues to the patient inside the
booth. We have since installed a video camera that
allows remote observation of the patient inside the
booth but have not detected any difference in the results
due to this change. Although the air flow in the booth
was maintained constant and substances were introduced
every time in the same manner, it would be essential
to measure (with gas chromatography) the actual levels
reaching the individuals and ensure that delivery of
the substances is constant through methods such as
those described by Fiedler et al. (2000).
In terms of experimental design, this pilot study
raised the significance of including an adaptation
phase to get stable physiologic measurements and minimize
noise in the results. This study also brings up questions
regarding the significance of an electrodermal response
to low-level chemical substances. Are we observing
an unconscious anxiety response, or another type of
response such as neurogenic inflammation? Other measures
such as functional magnetic resonance imaging would
certainly add understanding of the brain regions involved
in the reactivity, but this will require unique laboratory
conditions. Reproducibility of these results and understanding
the pathophysiologic mechanism should be the next priorities.

