We read with great interest the article by de Burbure et al. (2006) on health effects in children who live near nonferrous smelters in France, the Czech Republic, and Poland. We were especially interested in the inverse relationship found between levels of urinary mercury and serum prolactin. We found a similar result in an Italian multicenter crosssectional survey with adult subjects (Alessio et al. 2002) using a different statistical approach based on regression analysis with mixed linear models. We found that serum prolactin decreased as a function of both urinary mercury and occupational exposure to inorganic mercury (Lucchini et al. 2003). In another study (Carta et al. 2003), our group observed the opposite behavior of prolactin in adult individuals with a high dietary intake of mercury-contaminated tuna. In that study, serum prolactin was positively associated with urinary and blood mercury. Our interpretation of this dual behavior was that prolactin may be differently affected by inorganic and organic mercury based on the interference with different neurotransmitters implicated in the regulation of prolactin secretion (Carta et al. 2003).
The article by de Burbure et al. (2006) stimulates futher consideration of the observed effects on serum prolactin after exposure to various metals and other chemical substances. In fact, prolactin can be increased by exposure to lead (Govoni et al. 1987; Lucchini et al. 2000), organic mercury (Carta et al. 2003), and manganese (Ellingsen et al. 2003; Smargiassi and Mutti 1999; Takser et al. 2004), but it can be decreased by exposure to inorganic mercury (de Burbure et al. 2006; Lucchini et al. 2003; Ramalingam et al. 2003), alluminum (Alessio et al. 1989), and cadmium (Calderoni et al. 2005; de Burbure et al. 2006). Subjects exposed to chemicals such as styrene (Bergamaschi et al. 1996; Luderer et al. 2004; Umemura et al. 2005), perchloroethylene (Beliles 2002; Ferroni 1992), and anesthetic gases (Lucchini et al. 1996; (Marana et al. 2003) have shown an increase of serum prolactin, whereas polychlorinated biphenyls (De Krey et al. 1994) and the pesticide lutheinate [U.S. Environmental Protection Agency (EPA) 2002] are known to decrease serum prolactin.
Possible mechanisms, other than direct effects at the cellular level, may be related to different neurotransmitters involved in the modulation of prolactin secretion. For example, the dopaminergic and serotoninergic systems, respectively, are involved in the physiologic regulation of this hormone as a tonic inhibitor and as an excitatory modulator. Different chemicals may interfere with these two systems, resulting in different outcomes regarding serum prolactin. Recent studies have shown that the same chemical may even cause different effects on prolactin depending on the exposure doses (Lafuente et al. 2003).
We would like to know why this neuroendocrine hormone is affected differently by exposure to different chemicals. This is important because of the possible use of prolactin, as described by de Burbure et al. (2006), as a sensitive indicator of early effects in toxicologic research and risk assessment (Mutti and Smargiassi 1998). Negative studies have also been published on the association of prolactin with the exposure to neurotoxicants (Myers et al. 2003; Roels et al. 1992). Therefore, it is vital to assess the causes of the variability that may limit the reproducibility of these tests. Further research should focus on multiple exposure to different chemicals, which may help to explain the lack of association.
Alessio L, Apostoli P, Cortesi I, Lucchini R. eds. 2002. Assessment of Effects Due to Low Doses of Inorganic Mercury Following Environmental and Occupational Exposure: Human and in Vitro Studies on Specific Toxicity Mechanisms. Med Lav 93(3).
Alessio L, Apostoli P, Ferioli A, Di Sipio I, Mussi I, Rigosa C, et al. 1989. Behaviour of biological indicators of internal dose and some neuro-endocrine tests in aluminium workers. Med Lav 80:290-300.
Beliles RP. 2002. Concordance across species in the reproductive and developmental toxicity of tetrachloroethylene. Toxicol Ind Health 18:91-106. [CrossRef].
Bergamaschi E, Mutti A, Cavazzini S, Vettori MV, Renzulli FS, Franchini I. 1996. Peripheral markers of neurochemical effects among styrene-exposed workers. Neurotoxicol 17:753-759.
Calderoni AM, Oliveros L, Jahn G, Anton R, Luco J, Gimenez MS. 2005. Alterations in the lipid content of pituitary gland and serum prolactin and growth hormone in cadmium treated rats. Biometals 18:213-220.
Carta P, Flore C, Alinovi R, Ibba A, Tocco MG, Aru G, et al. 2003. Sub-clinical neurobehavioral abnormalities associated with low level of mercury exposure through fish consumption. Neurotoxicology 24:617-623.
de Burbure C, Buchet JP, Leroyer A, Nisse C, Haguenoer JM, Mutti A, et al. 2006. Renal and neurologic effects of cadmium, lead, mercury, and arsenic in children: evidence of early effects and multiple interactions at environmental exposure levels. Environ Health Perspect 114:584-590.
De Krey GK, Hollingshead NC, Kerkvliet NI, Smith BB. 1994. Suppression of prolactin and cytotoxic T-lymphocyte activity in PCB-treated mice. Int J Immunopharmacol 16:251-257.
Ellingsen DG, Haug E, Gaarder PI, Bast-Pettersen R, Thomassen Y. 2003. Endocrine and immunologic markers in manganese alloy production workers. Scand J Work Environ Health 29:230-238.
Ferroni C, Selis L, Mutti A, Folli D, Bergamaschi E, Franchini I. 1992. Neurobehavioral and neuroendocrine effects of occupational exposure to perchloroethylene. Neurotoxicology 13:243-247.
Govoni S, Battaini F, Fernicola C, Castelletti L, Trabucchi M. 1987. Plasma prolactin concentrations in lead exposed workers. J Environ Pathol Toxicol Oncol 7:13-15.
Lafuente A, Cano P, Esquifino A. 2003. Are cadmium effects on plasma gonadotropins, prolactin, ACTH, GH and TSH levels, dose-dependent? Biometals 16:243-250.
Lucchini R, Albini E, Cortesi I, Placidi D, Bergamaschi E, Traversa F, et al. 2000. Assessment of neurobehavioral performance as a function of current and cumulative occupational lead exposure. Neurotoxicology 21:805-811.
Lucchini R, Calza S, Camerino D, Carta P, Decarli A, Parrinello G, et al. 2003. Application of a latent variable model for a multi-center study on early effects due to mercury exposure. Neurotoxicology 24(4–5):605-616.
Lucchini R, Placidi D, Toffoletto F, Alessio L. 1996. Neurotoxicity in operating room personnel working with gaseous and non-gaseous anesthesia. Int Arch Occup Environ Health 68:188-192.
Luderer U, Tornero-Velez R, Shay T, Rappaport S, Heyer N, Echeverria D. 2004. Temporal association between serum prolactin concentration and exposure to styrene. Occup Environ Med 61:325-333.
Marana E, Annetta MG, Meo F, Parpaglioni R, Galeone M, Maussier ML, et al. 2003. Sevoflurane improves the neuro-endocrine stress response during laparoscopic pelvic surgery. Can J Anaesth 50:348-354.
Mutti A, Smargiassi A. 1998. Selective vulnerability of dopaminergic systems to industrial chemicals: risk assessment of related neuroendocrine changes. Toxicol Ind Health 14:311-323.
Myers JE, Thompson ML, Naik I, Theodorou P, Esswein E, Tassell H, et al. 2003. The utility of biological monitoring for manganese in ferroalloy smelter workers in South Africa. Neurotoxicology 24:875-883.
Ramalingam V, Vimaladevi V, Rajeswary S, Suryavathi V. 2003. Effect of mercuric chloride on circulating hormones in adult albino rats. J∞Environ Biol 24:401-404.
Roels HA, Ghyselen P, Buchet JP, Ceulemans E, Lauwerys RR. 1992. Assessment of the permissible exposure level to manganese in workers exposed to manganese dioxide dust. Br J Ind Med 49(1):25-34.
Smargiassi A, Mutti A. 1999. Peripheral biomarkers and exposure to manganese. Neurotoxicology 20:401-406.
Takser L, Mergler D, de Grosbois S, Smargiassi A, Lafond J. 2004. Blood manganese content at birth and cord serum prolactin levels. Neurotoxicol Teratol 26:811-815.
Umemura T, Kurahashi N, Kondo T, Katakura Y, Sata F, Kawai T, et al. 2005. Acute effects of styrene inhalation on the neuroendocrinological system of rats and the different effects in male and female rats. Arch Toxicol 79:653-659. doi:10.1007/s00204-005-0684-2. [Online 15 July 2005].
U.S. EPA. 2002. A Review of the Reference Dose and Reference Concentration Processes. EPA/630/P-02/002F. Washington, DC:U.S. Environmental Protection Agency. Available: http://www.epa.gov/IRIS/RFD_FINAL%5B1%5D.pdf [accessed 15 May 2006].
We appreciate the letter from Alessio and Lucchini concerning the number and variety of toxicants able to affect serum prolactin levels. Reflecting on the wide variability of the currently available data, we would like to make two additional points.
The first point concerns the usefulness of serum prolactin as a potential indicator of neurotoxicity for populations at risk. This biomarker indeed appears to be influenced by a large number of both organic and inorganic chemicals, which have seemingly little in common in terms of mechanistic action (e.g., heavy metals, pesticides, styrene, polychlorinated biphenyls). Moreover, one chemical—cadmium, for example—can have a biphasic dose-dependent effect on serum prolactin (Lafuente et al. 2003), an effect we did not observe in our study (de Burbure et al. 2006) because of low exposure levels; this dose-dependent effect is reminiscent of the biphasic effects of lead on glutamate neurotransmission shown to be dependent on glycine receptor affinity (Marchioro et al. 1996).
As proposed by Alessio and Lucchini in their letter, these data reflect the complexity of the control of prolactin secretion, which is modulated not only by dopamine but also by several other neurotransmitters. These neurotransmitters include serotonin, 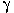 -aminobutyric acid (GABA) [as demonstrated by the hyperprolactinemia developed by GABAB1 knock-out mice (Catalano et al. 2005)], glycine, and glutamate (Fitsanakis and Aschner, 2005; Nagy et al. 2005). In view of these neurotransmitters, serum prolactin—albeit sensitive—appears to be a rather nonspecific biomarker for monitoring populations at risk; therefore, serum prolactin will likely remain a predominantly useful tool in the field of research until the multiple facets of controlling prolactin secretion are unveiled.
-aminobutyric acid (GABA) [as demonstrated by the hyperprolactinemia developed by GABAB1 knock-out mice (Catalano et al. 2005)], glycine, and glutamate (Fitsanakis and Aschner, 2005; Nagy et al. 2005). In view of these neurotransmitters, serum prolactin—albeit sensitive—appears to be a rather nonspecific biomarker for monitoring populations at risk; therefore, serum prolactin will likely remain a predominantly useful tool in the field of research until the multiple facets of controlling prolactin secretion are unveiled.
Another important issue to keep in mind concerns the biological significance of all of the modifications we observed in our study (de Burbure et al. 2006). Despite their statistical significance, are the observed small changes in serum prolactin at all clinically relevant? To what extent do the variations in serum prolactin induced by the various neurotoxicants correlate with changes in brain function? Because prolactin has a large number of potential determinants, probably with different mechanisms of action, it is a rather delicate intellectual exercise to give a correct interpretation of the observed changes in terms of the possible development of neurotoxicity.
Although the lack of specificity of prolactin reduces the immediate usefulness of these dopaminergic biomarkers, the question of the potential clinical impact of the small but significant changes in terms of neurotoxicity (de Burbure et al. 2006) certainly remains an important question that further research will have to address.
Catalano PN, Bonaventura MM, Silveyra P, Bettler B, Libertun C, Lux-Lantos VA. 2005. GABAB1 knockout mice reveal alterations in prolactin levels, gonadotropic axis, and reproductive function. Neuroendocrinology 82(5-6):294–305.
de Burbure C, Buchet JP, Leroyer A, Nisse C, Haguenoer JM, Mutti A, et al. 2006. Renal and neurologic effects of cadmium, lead, mercury, and arsenic in children: evidence of early effects and multiple interactions at environmental exposure levels. Environ Health Perspect 114: 584–590
Fitsanakis VA, Aschner M. 2005. The importance of glutamate, glycine, and gamma-aminobutyric acid transport and regulation in manganese, mercury and lead neurotoxicity. Toxicol Appl Pharmacol 204(3):343–354.
Lafuente A, Cano P, Esquifino A. 2003. Are cadmium effects on plasma gonadotropins, prolactin, ACTH, GH and TSH levels, dose-dependent? Biometals 16(2): 243–250.
Marchioro M, Swanson KL, Aracava Y, Albuquerque EX. 1996. Glycine and calcium-dependent effects of lead on N-methyl-D-aspartate receptor function in rat hippocampal neurons. J Pharmacol Exp Ther 279(1):143–153.
Nagy GM, Bodnar I, Banky Z, Halasz B. 2005. Control of prolactin secretion by excitatory amino acids. Endocrine 28(3):303–308.





