|
Synergistic Effect of 2,2',4,4',5,5'-Hexachlorobiphenyl and 2,3,7,8-Tetrachlorodibenzo-p-Dioxin on Hepatic Porphyrin Levels in the Rat
Angélique P.J.M. van Birgelen,1,2 Kitty M. Fase,1 Jolanda van der Kolk,3 Hermann Poiger,3 Abraham Brouwer,2 Willem Seinen,1 and Martin van den Berg1 1Research Institute of Toxicology, University of Utrecht, 3508 TD Utrecht, The Netherlands; 2Department of Toxicology, Agricultural University, 7600 EA Wageningen, The Netherlands; 3Institute of Toxicology, CH-8603 Schwerzenbach, Switzerland Abstract
We studied the effect of polychlorinated biphenyls (PCBs) on hepatic porphyrin accumulation in female Sprague-Dawley rats by feeding them diets containing 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) , 2,2´,4,4´,5,5´-hexachlorobiphenyl (PCB 153) , 2,3,3´,4,4´,5-hexachlorobiphenyl (PCB 156) , 3,3´,4,4´,5-pentachlorobiphenyl (PCB 126) , or combinations of the single PCB congeners with TCDD for 13 weeks. A dose-dependent increase in hepatic porphyrin accumulation occurred after TCDD, PCB 126, or PCB 156 administration, reaching maximal levels of about twice control values. The lowest dose levels for which a significant increase in hepatic porphyrin accumulation was found were 0.7 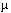 g TCDD/kg diet, 50 g TCDD/kg diet, 50 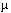 g PCB 126/kg diet, or 6 mg PCB 156/kg diet. These doses are equivalent to 47 ng TCDD/kg/day, 3.2 g PCB 126/kg diet, or 6 mg PCB 156/kg diet. These doses are equivalent to 47 ng TCDD/kg/day, 3.2 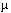 g PCB 126/kg/day, and 365 g PCB 126/kg/day, and 365 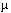 g PCB 156/kg/day. Relative potencies for hepatic porphyrin accumulation, using TCDD as a reference, ranged from 0.015 to 0.06 for PCB 126 and from 0.0001 to 0.0003 for PCB 156. CYP1A2 activities significantly correlated with hepatic porphyrin levels, with coefficients of 0.629, 0.483, or 0.808 for TCDD, PCB 126, or PCB 156, respectively. Administration of PCB 153 alone did not result in hepatic porphyrin accumulation. Co-administration of PCB 153 and TCDD revealed a strong synergistic effect on porphyrin accumulation (about 800 times control levels) . This synergistic effect was significant in rats fed diets containing any combination of PCB 153 with TCDD. Uroporphyrin III and heptacarboxylic porphyrin were accumulated in porphyrinogenic livers. These results suggest that TCDD induction of CYP1A2 may be involved, leading to oxidation of uroporphyrinogen III to uroporphyrin III, in combination with an increase in g PCB 156/kg/day. Relative potencies for hepatic porphyrin accumulation, using TCDD as a reference, ranged from 0.015 to 0.06 for PCB 126 and from 0.0001 to 0.0003 for PCB 156. CYP1A2 activities significantly correlated with hepatic porphyrin levels, with coefficients of 0.629, 0.483, or 0.808 for TCDD, PCB 126, or PCB 156, respectively. Administration of PCB 153 alone did not result in hepatic porphyrin accumulation. Co-administration of PCB 153 and TCDD revealed a strong synergistic effect on porphyrin accumulation (about 800 times control levels) . This synergistic effect was significant in rats fed diets containing any combination of PCB 153 with TCDD. Uroporphyrin III and heptacarboxylic porphyrin were accumulated in porphyrinogenic livers. These results suggest that TCDD induction of CYP1A2 may be involved, leading to oxidation of uroporphyrinogen III to uroporphyrin III, in combination with an increase in 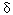 -aminolevulinic acid synthetase induced by PCB 153. Under porphyrinogenic conditions, an inhibitor of CYP1A2 activity may also be formed. The interactive effects on porphyrin accumulation after co-administration of dioxinlike and non-dioxinlike compounds may have significant implications for the risk assessment of these chemicals. Key words: Ah-receptor, chemical synergism, polychlorinated biphenyl, porphyria, porphyrins, 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin. Environ Health Perspect 104:550-557 (1996) -aminolevulinic acid synthetase induced by PCB 153. Under porphyrinogenic conditions, an inhibitor of CYP1A2 activity may also be formed. The interactive effects on porphyrin accumulation after co-administration of dioxinlike and non-dioxinlike compounds may have significant implications for the risk assessment of these chemicals. Key words: Ah-receptor, chemical synergism, polychlorinated biphenyl, porphyria, porphyrins, 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin. Environ Health Perspect 104:550-557 (1996)
Address correspondence to A.P.J.M. van Birgelen, U.S. Environmental Protection Agency, MD-74, Research Triangle Park, NC 27711 USA. J. van der Kolk is currently at Springborn Laboratories, PO Box 9326, Horn, Switzerland. We are grateful to P.R. Sinclair and L.S. Birnbaum for the insightful discussions regarding porphyrin metabolism. We thank D.G. Ross for the skillful assistance with the Western blot analysis of CYP1A2. This work was supported by The Technology Foundation, The Netherlands (grant UDG 92.1869) , The Swiss National Foundation, Switzerland (grant 2-77-330.91) , and the Chemical Manufacturer Association. This work was presented in part at the 15th International Symposium on Chlorinated Dioxins and Related Compounds, Edmonton, Canada, August 1995. Received 30 August 1995 ; accepted 17 January 1996.
The full version of this article is available for free in HTML format. |

