
| |  | |  |
Research Article
|
| Ecotoxicity in the Reconquista River, Province of Buenos Aires, Argentina: A Preliminary Study Jorge Herkovits, Cristina Silvia Perez-Coll, and Francisco Damian Herkovits
Programa de Seguridad Quimica, Instituto de Ciencias Ambientales y Salud, Fundación PROSAMA, Buenos Aires, Argentina Abstract
The Reconquista River in Argentina is considered a "supercritical" river basin due to environmental degradation. Within its valley of 1.547 km2, there are more than 3 million inhabitants and 12,000 industries. Using early-life-stage toxicity tests with Bufo arenarum embryos (the most sensitive of three native species) , we determined the water quality at six sampling stations of the river valley and expressed the results as acute and chronic toxicity units. Along most of the river, the toxicity was higher than the allowable level of whole industrial effluent toxicity recommended by U.S. EPA. In a tributary stream, Arroyo Morón, the water was about 10 times more toxic than the criteria maximum concentration (CMC) recommended by U.S. EPA for industrial effluents. Similar degradation of the water quality was found taking as a reference value an upstream sampling station. In all places where the water quality was worse than the CMC, no macroorganisms were found, and in one of these places, a large number of dead fishes was observed. Our study points out that amphibian early-life-stage toxicity tests could be appropriate for assessing water contamination and water quality, which is essential for diagnosis, protection of environmental services, monitoring, and restoration purposes. Key words: amphibians, aquatic toxicity, Argentina, Bufo arenarum, early-life stage toxicity test, rivers, water quality. Environ Health Perspect 104:186-189 (1996)
Address correspondence to J. Herkovits, Instituto de Ciencias Ambientales y Salud, Nicasio Oroño 710, 1405 Buenos Aires, Argentina. The standardization of the toxicity test with Bufo arenarum embryos was performed with a grant from the Pan American Health Organization. This study received support from the National Council of Science and Technology (CONICET) . We acknowledge Marcelo Merino and Miguel Angel Gomez Peral for their help during the sampling. We thank Daniela Fuentes for secretarial assistance. Received 4 August 1995 ; accepted 20 November 1995. |
|
|
 |
Introduction
Most developing countries face the enormous challenge of improving living standards against a backdrop of serious environmental and health problems. For instance, because streams and rivers have been considered a convenient means of cleaning and carrying wastes away from the discharge points, most rivers in urban areas are highly loaded with urban and industrial wastes (1,2). According to the Pan American Health Organization, less than 10% of the municipalities in Latin America treat sewage adequately before emptying it into natural watercourses, and wastewater treatment plants and sewers for industrial effluents are often not working or nonexistent (3). In addition, pollution prevention, recycling, and chemical safety programs are scarce or nonexistent. As a result of these practices, South America pollutes nearly 11 times more freshwater on a per capita basis than Europe (3). The untreated wastewater dumped into the rivers in many cases becomes part of the water sources downstream.
The Reconquista River Valley is located in the northern area of metropolitan Buenos Aires. It extends 1.547 km2 and has more than 3 million inhabitants. There are also about 12,000 industries located in the area, and therefore the major cause of pollution is the high load of urban and industrial wastes poured into the river. The river used to be dark-colored and bad smelling along most of its extension; it is well known that macroorganisms are alive only upstream. Its flow varies between 69,000 and 1,700,000 m3/day in cases of heavy rain (4-6).
Traditionally, the quality of liquid effluent discharges entering head waters and the river water itself has been controlled by comparing physical and chemical parameters with reference parameters and priority pollutant lists. But such an analytical approach was recognized as unable to protect the aquatic environment against hazardous discharges (2). Regulatory authorities worldwide are showing increasing interest in using laboratory toxicity tests to assess the quality of river waters. For instance, the Organization for Economic Cooperation and Development recommended more than 10 years ago the adoption of toxicity testing as one factor in making decisions about water regulations (7,8).
Amphibians are useful indicators of freshwater contamination (9-11). Embryos of Bufo arenarum, a South American toad, due to their high sensitivity to environmental pollutants (12-15), could be very useful for ecotoxicological studies and for assessing the water quality needs of widlife (16). On the other hand, it has been established that there is a good predictive correlation between embryo-larval tests for measuring in-stream toxicity and estimating chronic effects on aquatic biota (17). These studies indicate a good predictive correlation between embryo-larval survival and independent ecological parameters, especially species richness (r = 0.96) and diversity (r = 0.93).
The pollution of rivers in developing countries is usually not evaluated from an ecotoxicological point of view. The main purpose of this study was to generate, by means of ecotoxicological data on the Reconquista River basin, quantitative information on the water quality that could be used by the authorities as well as the general public. The results of water quality assessment at six sampling stations were compared to the criteria maximum concentration (CMC) for whole effluent toxicity recommended by the U.S. EPA (17). Based on susceptibility studies to toxicants in the Reconquista River, performed in our laboratory with three native species [a fish (Cnesterodon decemmaculatus), a shrimp, collected at R1, our reference upstream sampling station, and toad embryos (Bufo arenarum)], this early-life toxicity test was the most sensitive and appropriate for evaluating water quality at all sampling stations. Here we focus on the results obtained with Bufo arenarum embryos. In addition, an overview of the presence of macroorganisms (alive or dead) at the sampling stations is reported.
Methods
 |
|
Figure 1. Geographic localization of the Reconquista River basin showing the position of the sampling stations (R1, R2, AM, R3, R4, and R5). |
We selected six sampling stations (Fig. 1): reference point upstream close to Dique Ruggero (R1), a reference point downstream within 100 m of the discharge of the Reconquista River into the Lujan River (R5), three sampling stations between R1 and R5 (R2, R3, and R4), and one sampling station on a stream, Arroyo Moron (AM), about 300 m from its discharge into the Reconquista River. All sampling stations were located far away from industrial or municipal discharge points. Surface water was collected at each site near the shore in June 1994. We used a manual pump to collect 20 l of water at a depth of 0.5 m, transported the water to the laboratory, and refrigerated it at 4°C for processing. At each sampling station, the presence of macroorganisms was recorded by capturing them with a net (diameter of the hole: 0.2 mm) and by direct observation.
Ovulation of Bufo arenarum females was induced by injecting a suspension of a homologous hypophysis. The oocytes were fertilized in vitro with a sperm suspension in 10% Holtfreter's solution (HS). Embryos obtained from three couples of parents were maintained in HS at 20 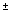 2°C. Experiments were performed with embryos at the following developmental stages: stage 13 (gastrulae), stage 18 (neuromuscular activity), and stage 25 (complete operculum) (18). Starting at these stages, we conducted 7-day embryo-larval survival and growth renewal toxicity test using duplicate batches of 10 embryos placed in 10-cm glass petri dishes containing 40 ml of water from each sampling station. In the case of embryos from the stage 13 and 18, in addition to survival, we recorded malformations and anomalies observed using a binocular estereoscopic microscope. To obtain additional information on the water quality in samples with less toxic effects, the toxicity test performed with embryos at stage 25 was extended up to 14 days. For each sample, eight dilutions ranging between 80% and 30% were prepared using HS, and toxicity tests with embryos were conducted with these concentrations simultaneously with the undiluted samples and with control embryos maintained in HS solution. For dilutions, we used HS, rather than water from the reference sampling point, in accordance with the procedures of FETAX (19), the best-known standard method for developmental toxicity tests with amphibians. 2°C. Experiments were performed with embryos at the following developmental stages: stage 13 (gastrulae), stage 18 (neuromuscular activity), and stage 25 (complete operculum) (18). Starting at these stages, we conducted 7-day embryo-larval survival and growth renewal toxicity test using duplicate batches of 10 embryos placed in 10-cm glass petri dishes containing 40 ml of water from each sampling station. In the case of embryos from the stage 13 and 18, in addition to survival, we recorded malformations and anomalies observed using a binocular estereoscopic microscope. To obtain additional information on the water quality in samples with less toxic effects, the toxicity test performed with embryos at stage 25 was extended up to 14 days. For each sample, eight dilutions ranging between 80% and 30% were prepared using HS, and toxicity tests with embryos were conducted with these concentrations simultaneously with the undiluted samples and with control embryos maintained in HS solution. For dilutions, we used HS, rather than water from the reference sampling point, in accordance with the procedures of FETAX (19), the best-known standard method for developmental toxicity tests with amphibians.
Conductivity was measured with a Luftman C400 conductivity meter, and pH was measured with a Luftman P300 pH meter. These measurements provided information on the water quality needed for the control solution in the toxicity test. Intralaboratory quality control methods for the toxicity test based on reference toxicants were performed with cadmium, and the minimum criteria of test acceptability including the percent coefficient of variation was conducted.
Results and Discussion
The biological integrity of the Reconquista River was evaluated by the presence of macroorganisms (alive or dead) at each sampling station. At the upstream reference sample station (R1), a large number of shrimp were collected, while at R2, many dead fish, an average of 10 within 10 m of the shore, were counted. No living macroorganisms were observed from this sampling station downstream. These observations indicate the high degradation of the water quality in this ecosystem. There have been some previous chemical, physical, and biological integrity studies on the Reconquista River which also indicated the poor water quality of this river. For instance, the river is substantially anaerobic, with a mean of biological oxygen demand of approximately 60 mg/l, varying between 30 mg/l and 100 mg/l (5). In addition, some data on ammonia, hardness, pH, cations, and pollutants like heavy metals and pesticides (5,6,20-22), as well as a partial inventory of the biota, are available (23).

Figure 2. Survival of Bufo arenarum embryos maintained in water samples from the Reconquista River basin.

Figure 3. Concentration-response relationships obtained with Bufo arenarum embryos in the Morón stream sample.
Although most EPA chronic toxicity tests have been shortened to 7 days by focusing on the most sensitive life-cycle stages (17), because our study is focused on surface water toxicity and based on our previous experience (24), we extended the observations the stage 25 embryos up to 14 days. Due to the very poor water quality in the Reconquista River, the 7-day toxicity test was sufficient for most of the purposes of this study. In Figure 2, the profile of exposure-toxicity of the water from the six sampling stations is shown. The concentration-exposure-response results obtained for AM, which had the worst water quality of the sampling stations, is shown in Figure 3. These results illustrate the high sensitivity of the amphibian embryo to the toxicity of the river water. It is noteworthy that in a previous study conducted in our laboratory involving the early-life stage Bufo arenarum, we found that, in the case of certain leachates, up to 0.1% of a dilution can be detected (24).
 |
 |
 |
|
Figure 4. Ecotoxicity of Reconquista River basin expressed in acute toxic units. |
|
Figure 5. Ecotoxicity of Reconquista River basin expressed in chronic toxicity units. MRV, EPA maximal reference value; AM, Arroyo Morón stream. |
|
Figure 6. Ecotoxicity of Reconquista River basin expressed in reference chronic toxicity units. AM, Arroyo Morón stream. |
Because toxicity involves an inverse relationship to effective concentration (EC; the lower the EC the higher the toxicity), it is helpful to translate concentration-based toxicity measurements into toxic units. The number of toxic units was defined as 100 divided by the EC measured: in the case of acute toxicity (TUa) = 100/LC50; for chronic toxicity (TUc) = 100/no-observed effect level (17). For whole (industrial) effluent, EPA recommended a maximal value of 0.3 TUa and 1.0 TUc for the most sensitive of at least three test species in both cases. These values were used as references for all the sampling stations studied. The toxicity of the water from the Reconquista River basin on Bufo arenarum embryos (stage 25), expressed as TUa and TUc, is presented in Figures 4 and 5. These data demonstrate that the water sampled was about 10 times more toxic than the maximal value allowed by the EPA for industrial effluents (17). The toxicity of water from the different sampling stations was compared to the "toxicity" of that from a reference sampling station, R1, using the short-term chronic toxicity test (7 days) and was equal to the values reported in Figure 5. When the exposure period is extended, the sensitivity of the test increases (Fig. 6), but in any case it is noteworthy that the profile obtained in extended tests is similar to that obtained based on the EPA criteria for whole industrial effluents.
Although the use of earlier developmental stages (stages 13 and 18), provides meaningful additional information on the teratological potential of the water contamination and on water quality, the results do not reflect a significantly higher susceptibility than the mortality exerted on Bufo arenarum embryos at stage 25. The evaluation of these earlier stages is by far more detailed and includes a precise record of malformations and other developmental anomalies. The main malformations found were axial incurvation, retarded growth, reduced body size, microcephaly, and fin anomalies. These are unespecific malformations produced by different xenobiotics (25-27).
At the screening level, hazard assessment of environmental toxicants is often based on a few laboratory tests in single species. Safety factors are applied to account for the many uncertainties inherent in extrapolating from one species to another, from acute to chronic exposures, and from single species to ecosystems (7,8,28). Our results seem to confirm that within single-species protocols, amphibian embryos could be among the most sensitive to environmental pollutants (13,14,29). Athough the shrimp we collected in R1 were rather sensitive to the degradation of the water quality in Reconquista River (data not shown), this species seems to be adapted to the high salinity at the reference sampling station (R1) where it was collected and therefore does not survive in low osmolarity conditions found in the lower part of the river (the mixing zone with the water of Lujan River). It is also noteworthy that the test performed with Cnesterodon decemmaculatus only detected toxicity within 7 days for water obtained from AM, the sampling station with highest pollution. The difference in susceptibility between fish and amphibian embryos was more than one order of magnitude. These results are in agreement with the high susceptibility of amphibians to xenobiotics (13,14,29,30) support increasing concerns about the decline of amphibian populations (9,10,31), which seems to be related to the water quality needs of wildlife particularly neglected for these species (16).
The primary objective of this study was to evaluate the possibility of determining pollution by using a relatively simple bioassay procedure performed with native species and to identify the most appropriate test for water quality-based toxicants evaluation (control) for the Reconquista River ecosystem. By focusing on the most sensitive life-cycle stages, the duration of EPA chronic toxicity tests for water quality-based toxicants control has been shortened to 7 days (17). Our study points out that although a 7-day toxicity test with Bufo arenarum embryos seems to be appropriate for water toxicity screening purposes, at least for the high pollution found at most of the sampling stations, it is worthwhile to extend the test to 14 days of exposure to detect toxicity levels that could be significant for wildlife. In this respect, ecotoxicological studies should be customized and interpreted carefully to provide meaningful information (32).
River systems play key roles in ecological interactions, and in the regulation and maintenance of biodiversity in the landscape (33). These interactions include the atmosphere and groundwater. Human societies rely on functions of ecosystems that are essential to the quality of human life, including provision of water and food, the decomposition of sewage, and the replenishment of breatheable air (34). Evaluations of the environmental impact of human activities as well as strategies for river conservation should be considered a main priority for the environmental and human health. The idea that rivers are a convenient means of cleaning and carrying pollution away from the discharge point should be urgently and properly reconsidered worldwide in order to protect water quality, ecosystems, and human health. Our results support the use of toxicity tests with amphibian embryos, as they could accurately indicate water quality for wildlife protection purposes.
|
|
 |
|
References
1. Smith RA, Alexander RB, Wolman MG. Water-quality trends in the nation's rivers. Science 235:1607-1615 (1987).
2. Garric J, Vindimian E, Ferard JF. Ecotoxicology and wastewater: some practical applications. Sci Total Environ (suppl) 1085-1103 (1993).
3. Black H. The price of progress: environmental health in Latin America. Environ Health Perspect 102:1024-1028 (1994).
4. CEAMSE. Investigación sobre la contaminación del Rio Reconquista. Report 1-68. Thames Water Consultancy Services, 1979.
5. CEAMSE. An investigation into the pollution of the River Reconquista (supplementary studies). Document 1-21, Thames Water Consultancy Service, 1980.
6. Federovisky S. Informe sobre la contaminación Rio Reconquista. Document 1-32, Greenpeace, 1988.
7. OECD. Guidelines for testing of chemicals. In: Proceedings of the international workshop on biological testing of effluents and related receiving waters. Duluth, MN:Organization for Economic Cooperation and Development, 1984.
8. OECD. Monographie sur l'environemet. Utilisation des tests biologiques pour l'evaluation et le contrôle de la pollution de l'eau. Organization for Economic Cooperation and Development, 1987.
9. Cooke AS. Tadpoles as indicators of harmful levels of pollution in the field. Environ Pollut 25:123-133 (1981).
10. Dunson WA, Wyman RL, Corbert ES. A symposium on amphibian declines and habitat acidification. J Herpetol 26:349-352 (1992).
11. Blaustein AR, Wake DB, Sousa WP. Amphibian declines:judging stability, persistence and susceptibility of populations to local and global extinctions. Conserv Biol 8:60-71 (1994).
12. Bantle JA, Fort DJ, James BI. Identification of developmental toxicants using the frog embryo teratogenesis assay-xenopus (FETAX). Hydrobiologia 188/189:577-585 (1989).
13. Herkovits J, Perez-Coll CS. Amphibian embryos as bioassays to evaluate environmental pollutants and to reduce their toxic effects. In: Proceedings of the environmental contamination 4th international conference, Barcelona (Barceló E, ed). 1990;623-626.
14. Hall RJ, Henry PFP. Review: assessing effects of pesticides on amphibians and reptiles: status and needs. Herpetol J 2:65-71 (1992).
15. Herkovits J, Perez-Coll CS. Stage dependent susceptibility to cadmium on Bufo arenarum embryos. Bull Environ Contam Toxicol 50:608-611 (1993).
16. Boyer R, Grue CE. The need for water quality criteria for frogs. Environ Health Perspect 103:352-357 (1995).
17. U.S. EPA. Technical support document for water quality-based toxics control. EPA 505-2-90-001. Washington, DC:Environmental Protection Agency, 1991.
18. Del Conte E, Sirlin L. The first stages of Bufo arenarum development. Acta Zool Lilloana 12:495-499 (1951).
19. ASTM. Standard guide for conducting the frog embryo teratogenesis assay-Xenopus (Fetax). Designation: E1439-91. In: Annual book of ASTM standards, vol 11.04. Philadelphia, PA:American Society for Testing and Materials, 1994;1286-1296.
20. Castañe PM, Topalian ML, dela Torre F, Demichelis SO, Garcia ME. Nutrientes y caracterización físico-química en el agua del Río Reconquista. Monitoreo de otoño 1994. In: Proceedings of the simposio internacional evaluación de riesgo y saneamiento de cuencas hídricas (in press).
21. Merino MH, Nicolli HB, Gomez Peral MA, Herkovits J, Perez-Coll CS, Herkovits FD. Evaluación ecotoxicologicay características geoquímicas de la cuenca del Río Reconquísta, Provincia de Buenos Aires: Estudio preliminar. II. Características Geoquímicas. In: Proceedings of simposio internacional evaluación de riesgo y saneamiento de cuencas hídricas 1994 (in press).
22. Kuczynski D. Atlas ecológico del Arroyo Morón. Report 7-98. Universidad de Morón, 1991.
23. Loes CR, Salibian A. Premieres donneêf sur le phytoplacton et les caracteristiqués physico-chimiques du Rio Reconquista (Buenos Aires, Argentina). Une riviere urbaine polluee. Rev Hydrobiol Trop 23:283-296 (1990).
24. Herkovits J. Bioassays for ecotoxicological and chemical safety studies. Ingenieria Sanitaria y Ambiental 19:42-48 (1995).
25. Vismara C, Bernardini G, Bonfanti P, Colombo A, Camatini M. The use of in vitro fertilization in the frog embryo teratogenesis assay in Xenopus (FETAX) and its applications to ecotoxicology. Sci Total Environ (Suppl) 787-790 (1993).
26. Herkovits J, Perez-Coll CS. Zinc protection against delayed development produced by cadmium. Biol Trace Elem Res 24:217-221 (1990).
27. Perez-Coll CS, Herkovits J. Stage dependent susceptibility to lead in Bufo arenarum embryos. Environ Pollut 63:239-245 (1990).
28. Sloof W, van Oers JAM, de Zwart D. Margins of uncertainty in ecotoxicological hazard assessment. Environ Toxicol Chem 5:841-852 (1986).
29. Wesley J, Birge J, Black A, Westerman A. Short term fish and amphibian embryo-larval test for determining the effects of toxicant stress on early life stages and estimating chronic values for single compounds and effluents. Environ Toxicol Chem 4:807-821 (1985).
30. Holcombe GW, Phipps GL, Sulaiman AH, Hoffman AD. Simultaneous multiple species testing: acute toxicity of 13 chemicals to 12 diverse freshwater amphibian, fish and invertebrate families. Arch Environ Contam Toxicol 16:697-710 (1987).
31. Wake DB. Declining amphibian population. Science 253:860 (1991).
32. Cairns J Jr. Third wave ecotoxicology. Ecotoxicology 3:1-3 (1994).
33. Dynesius M, Nilsson Ch. Fragmentation and flow regulation of river systems in the northern third of the world. Science 266:753-762.
34. Cairns J, Niederlehner BR. Estimating the effects of toxicants on ecosystem services. Environ Health Perspect 102:936-939 (1994).
Last Update: May 8, 1997
|
|
 |
|
| |

