| Rodent Cell Transformation and Immediate Early Gene Expression Following 60-Hz Magnetic Field Exposure Elizabeth K. Balcer-Kubiczek,1,2,3 Xiao-Feng Zhang,1 George H. Harrison1,2 Welton A. McCready,1 Zhong-Ming Shi,1 Lin-Huang Han,1 John M. Abraham,3,4 Luther L. Ampey, III,1 Stephen J. Meltzer,2,3,4 Maria C. Jacobs,1,2 and Christopher C. Davis5 1Department of Radiation Oncology; 2Program in Oncology; 3Cell and Molecular Biology Graduate Program; 4Department of Medicine (Gastroenterology Division), University of Maryland School of Medicine, Baltimore, Maryland 21201 USA; 5Electrical Engineering Department, University of Maryland, College Park, MD 20742 USA Abstract
Some epidemiological studies suggest that exposure to power frequency magnetic fields (MFs) may be associated with an elevated risk of human cancer, but the experimental database remains limited and controversial. We investigated the hypothesis that 60-Hz MF action at the cellular level produces changes in gene expression that can result in neoplastic transformation. Twenty-four hour 200 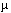 T continuous MF exposure produced negative results in two standard transformation systems (Syrian hamster embryo cells and C3H/10T1/2 murine fibroblasts) with or without postexposure to a chemical promoter. This prompted a reexamination of previously reported MF-induced changes in gene expression in human HL60 cells. Extensive testing using both coded and uncoded analyses was negative for an MF effect. Using the same exposure conditions as in the transformation studies, no MF-induced changes in ornithine decarboxylase expression were observed in C3H/10T1/2 cells, casting doubt on a promotional role of MF for the tested cells and experimental conditions. Key words: ß-actin, C3H/10T1/2 cells, c-fos, c-myc, heat, 60-Hz magnetic field, neoplastic transformation, ODC, ribosomal genes, SHE cells, TPA, X rays. Environ Health Perspect 104:1188-1198 (1996) T continuous MF exposure produced negative results in two standard transformation systems (Syrian hamster embryo cells and C3H/10T1/2 murine fibroblasts) with or without postexposure to a chemical promoter. This prompted a reexamination of previously reported MF-induced changes in gene expression in human HL60 cells. Extensive testing using both coded and uncoded analyses was negative for an MF effect. Using the same exposure conditions as in the transformation studies, no MF-induced changes in ornithine decarboxylase expression were observed in C3H/10T1/2 cells, casting doubt on a promotional role of MF for the tested cells and experimental conditions. Key words: ß-actin, C3H/10T1/2 cells, c-fos, c-myc, heat, 60-Hz magnetic field, neoplastic transformation, ODC, ribosomal genes, SHE cells, TPA, X rays. Environ Health Perspect 104:1188-1198 (1996) Address correspondence to E.K. Balcer-Kubiczek, Department of Radiation Oncology, Division of Radiation Research, 655 W. Baltimore Street BRB 6-015, Baltimore, MD 21201 USA. The authors are grateful to Gary A. Kerckaert, the Procter & Gamble Company, Human and Environmental Safety Division for his valuable help with establishing the SHE (pH 6.7) assay in our laboratories ; we would like to express our appreciation for comprehensive protocols, frozen stocks of SHE cells, and other materials for performing transformation studies with SHE cells reported here. We thank our colleague in the Department of Radiation Oncology, Juong G. Rhee, for the HL60 cell stock. This research was supported in part by the USPHS grants ES06124 (the Shannon award) and ES07120A from the National Institute of Environmental Health Sciences and by University of Maryland Designated Research Initiative Fund. Received 30 May 1996 ; accepted 22 July 1996.
The full version of this article is available for free in HTML format. |

