FDA > CDRH >
CDRH Learn > Presentation: 510(k)Third Party Reviews
Presentation: 510(k) Third Party Reviews
(This presentation is also available in video format with captioning and PDF printer-friendly format.)
Eric Rechen
Policy Analyst Program Operations Staff
FDA Office of Device Evaluation
CDRH
Third Party Review Program(a.k.a. “Accredited Persons Program”)
- Gives 510(k) submitters the option of using accredited, non-Federal organizations to review 510(k)s for low and moderate risk devices, in place of FDA’s review
- Authorized by §523 of the FD&C Act
Purpose
- More rapid decisions
- Better allocationof FDA’s resources
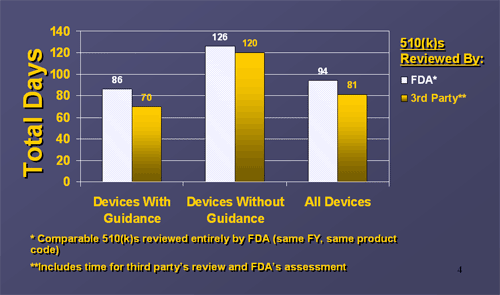
Comparison of Average Total Elapsed Days for 510(k) Reviews- Excluding “Special” 510(k)s -(FY 2005 Receipts)
Use of Third Parties
- Approximately 300 “third party” 510(k)s in FY 2008
- 8% of all 510(k)s
How Does It Work?
Third Party (TP) Review Process
- Applicant may elect to use TP or FDA for eligible devices
- If TP route is chosen:
Applicant contracts with TP
- TP reviews 510(k), makes recom.
- FDA issues final decision (30 days)
- Procedural guidance
- List of eligible devices
- List of Accredited Persons
Which Devices Are Eligible?
Eligible Devices
Statutory Limitation§ 523(a)(3)
Third parties may not review:
- Class III devices
- Class II devices that:
- are permanently implantable
- are life sustaining/supporting, or
- require clinical data in 510(k)s
Statutory Limitation§ 523(a)(3)
Third parties also may not review:
- 510(k)s that require CBER/CDER lead or consulting review. Example:
(e.g., drug/device combination products)
Who Are the Third Parties?
Accredited Organizations
- British Standards Institution (United Kingdom)
- Center for Measurement Standards, ITRI (Taiwan)
- Cheiroon, BV (Netherlands)
- CITECH
- Intertek Testing Services
- KEMA Quality, BV (Netherlands)
- NIOM Scand. Inst. of Dental Materials (Norway)
- Regulatory Technology Services, LLC
- TUV SUD America, Inc.
- TUV Rheinland of North America, Inc.
- Underwriters Laboratories, Inc.
Accreditation of Third Parties
- FDA serves as accreditation body
- Emphasis is on adequacy of:
- Personnel and procedures to ensure competent reviews
- Controls to prevent conflict of interest
Why Consider a Third Party?
- Usually more timely
- Many TPs also have standards expertise and foreign regulatory role
- Accessibility
- No FDA user fee
When to Think Twice
- Complex, precedent-setting submissions
- Device eligibility uncertain
(e.g., may require clinical data)
- “Special” 510(k)s
- TP lacks relevant experience
Questions?
Updated January 8, 2009

CDRH Home Page | CDRH A-Z Index | Contact CDRH | Accessibility | Disclaimer
FDA Home Page | Search FDA Site | FDA A-Z Index | Contact FDA | HHS Home Page
Center for Devices and Radiological Health / CDRH



![]()