EduNeering ComplianceWire Courses
Through a Cooperative Research and Development
Agreement (CRADA) with EduNeering, Inc. of Princeton, NJ, ORA U is offering many technical
competency courses online. The objective of the CRADA is to favorably impact public health
by increasing the skill level and productivity of FDA inspectors and by improving
compliance with government regulations in FDA-regulated industries.
See the online ComplianceWire course catalog for a
complete list of all available courses.
Pre-existing EduNeering courses
As part of the CRADA, FDA has reviewed existing Eduneering online technical and compliance
courses for adherence to FDA policy. FDA personnel will have access to the entire library
of over 100 existing courses. These include such titles as Introduction to GMPs,
Principles of Good Documentation, and Principles of Auditing.
All courses listed with a category of
"General" are EduNeering courses provided for FDA use under the terms of the
CRADA. These courses were developed by EduNeering to train personnel in regulated
industries. Although we expect that the vast majority of information in these courses is
accurate, they have not been reviewed by FDA personnel for consistency with our
regulations and policies. Language used in these courses reflects the target audience --
regulated industry.
FDA students are urged to take "General"
category courses as they keep in mind the industry audience for whom they were developed.
If you find an inconsistency in the content of these courses, please e-mail your concern
to ORA U at orau@ora.fda.gov. Please be sure to
include the course title and topic area in which the problem appears. Be as detailed as
possible in your comment.
Thank you in advance for your help!
FDA-Sponsored Courses
FDA and EduNeering will be developing online versions of existing FDA courses. The first
phase of this initiative will include Food and Drug Law, Basics of HACCP, Food
Microbiological Control, Basics of Investigations, and numerous other titles.
To take the courses on EduNeering’s
ComplianceWire, you must download and install Macromedia’s Shockwave Player. This is
a free download available over the Internet. Go to the Recommended System Requirements
page and click on "Shockwave plug-in" for directions. Be sure to do this BEFORE
you log on to EduNeering’s ComplianceWire site.
On the EduNeering ComplianceWire logon
page, enter the following:
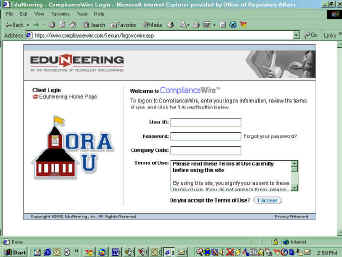
- User ID
You should have received a letter with your user ID included. Accounts in the system use
one of four user ID conventions:
1. User ID: this is your e-mail identifier, usually your first initial and last name
(i.e., jdoe@ora.fda.gov – user ID "jdoe") Use whatever you use before the @
symbol in your official e-mail address.
2. Your first initial and last name - For example: jsmith for Jane Smith
3. Firstname.lastname - For example: jane.smith for Jane Smith
4. Either of the two options above, plus a number at the end if your user ID duplicated
someone else's - For example: jsmith2
- Password
Everyone's initial password has been set as "orau". You will be prompted to
customize it the first time you log in.
- Company code
Use "fdaoraustates" plus your state's two-letter abbreviation. -
- Click "I Accept"
You should now be logged in to the ComplianceWire
system. Your To-Do list will appear first. This is where course titles may be listed if
they have been assigned to you.
If you select "Catalog"
from the drop-down box at the top of the screen, a list of all available courses will
appear. You can do a keyword search for a particular topic. Click on the title of any
course you are interested in taking, and it will be added to your To-Do list as an
elective.
A virtual "Tour of the FDA" has been
developed to give new employees an overview of every FDA center. Available to the public
as well as FDA employees, this course provides a better understanding of the FDA and its
structure. It covers all units within FDA and explains how each works. The course also
describes FDA history, its impact on society, and its mission. Click here to take a
"Tour of the FDA" without logging in to ComplianceWire. If you want to
receive credit for completing this course, access it from the regular course catalog after
you have logged in to ComplianceWire.
Please contact your system adiminstrator for computer
support. For questions/comments about course content, contact ORAU@ORA.FDA.GOV
Last Updated May 29, 2003 |


