|
TRIMELLITIC ANHYDRIDE
| Method no.: |
98 |
|
|
|
|
| Matrix: |
Air |
|
|
|
|
| Target concentration: |
40 µg/m3 |
|
|
|
|
| Procedure: |
Samples are collected open face on glass fiber
filters coated with veratrylamine (3,4-dimethoxybenzylamine)
and di-n-octyl phthalate. Samples are extracted with
aqueous ammonium hydroxide and analyzed by HPLC using a UV
detector. |
|
|
|
|
Recommended air volume
and sampling rate: |
480 L at 2.0 L/min |
|
|
|
|
| Reliable quantitation limit: |
0.623 µg/m3 |
|
|
|
|
Standard error of estimate
at the target
concentration:
(Section 4.7.) |
6.4% |
|
|
|
|
| Status of method: |
Evaluated method. This method has been subjected to
the established evaluation procedures of the Organic Methods Evaluation
Branch. |
|
|
|
|
| Date: November 1992 |
Chemist: Yihlin Chan |
Organic Methods Evaluation Branch
OSHA Salt Lake Technical Center
Salt Lake City, UT 84165-0200
1. General Discussion
1.1. Background
1.1.1. History
Over the years, many authors have reported air sampling and analytical procedures for
trimellitic anhydride (TMA). Purnell and Warwick (1980, Ref. 5.1.) collected TMA on
glass fiber filters, extracted with aqueous sodium hydroxide, and analyzed by HPLC.
Palassis et al. (1981, Ref. 5.2.) collected TMA on DM-800 (PVC-copolymer) membrane
filters, extracted with methanol, converted TMA to trimethyl ester with boron trifluoride,
and analyzed by GC/FID. Geyer et al. (1986, Ref. 5.3.) also used glass fiber filters, but
extracted the samples with 0.05 M sodium hydroxide and analyzed by HPLC using
acidified mobile phase. A similar method was published by Ledbetter et al. (1987, Ref.
5.4.) involving glass fiber filters, acetonitrile/water extraction, and HPLC or GC/FID
analysis. The current (1990) NIOSH Method (Ref. 5.5.) was based on the procedure of
Palassis et al. However, all of these procedures are susceptible to interference from
trimellitic acid that is present in the sampled air because none of them differentiates TMA
from trimellitic acid.
Rushing et al. (1982, Ref. 5.6.) presented an analytical method for simultaneously
determining TMA and trimellitic acid, whereby a sample was dissolved in anhydrous
ether and treated with diazomethane. Under anhydrous conditions the anhydride
functional group was left intact and TMA was converted to monomethyl ester whereas
trimellitic acid was converted to trimetyl ester. However, this procedure can not be
applied to air samples because TMA will react with water in the air to form trimellitic acid
even in the solid state at room temperature (Ref. 5.7.). TMA deposited on the sampling
filter will continually undergo hydrolysis as more air is drawn through the filter.
OSHA has been using a partially evaluated method whereby TMA is collected in 2-propanol bubblers. TMA is converted to mono-2-propyl esters (two isomers) while
trimellitic acid is not affected. They are separated by HPLC. Problems with this
procedure are that the use of a bubbler is inconvenient, and trace amounts of water in
2-propanol, either present originally or collected during sampling, may hydrolyze a
portion of the TMA to the acid before it is converted to the ester.
The method presented here uses coated filters for collecting airborne TMA. Glass fiber
filters are coated with veratrylamine in order to derivatize TMA in situ. di-n-octyl
phthalate is added to improve the contact between the reactants. A high sampling rate
(2 L/min) is recommended in order to obtain reasonable detection limits with short
sampling times. Trimellitic acid does not interfere because it does not react with
veratrylamine. The derivatized TMA (TMAVA) is stable. The sampler has a capacity (at
80% relative humidity and 25°C) of 300 µg or about 15 times the target concentration at
the recommended air volume. Two isomers of TMAVA are formed in about 1:1 ratio.
They are analyzed by HPLC, eluted either as a single peak or two separate peaks,
depending on the column used.
1.1.2. Toxic effects (This section is for information only and should not be taken as the basis
of OSHA policy.) (Refs. 5.8.-5.10.)
TMA causes severe respiratory irritation and sensitization. Exposure may result in
noncardiac pulmonary edema, immunological sensitization, and irritation of the
pulmonary tract, eyes, nose, and skin. Repeated inhalation of TMA dust or fume over
a period of weeks to years can cause an allergic reaction: TMA binds to a protein (human
serum albumin) forming an antigen. In susceptible individuals, the body then forms one
or more types of antibodies specific to the TMA-antigen. Upon re-exposure, the
antibodies react with the TMA-antigen and an allergic reaction occurs. TMA is listed in
the Table Z-1-A of the Code of Federal Regulations with a Final Rule Limit of 0.04 mg/m3
TWA. ACGIH, in its Notice of Intended Change, has set the TLV for TMA at 0.04 mg/m3
ceiling.
The pulmonary tract, eyes, nose, and skin all present a moist environment, therefore it
is not unreasonable to suspect that the hydrolysis product, trimellitic acid, may be a
contributor to the toxic effects attributed to TMA. Despite the fact that this is not
established, much less addressed in the literature, the exposure burden to trimellitic acid
may be useful supplemental information. Although this method has not been evaluated
for trimellitic acid, the analytical conditions presented allow the detection and quantitation
of the acid.
1.1.3. Workplace exposure (Refs. 5.1., 5.8.-5.9. and 5.11.)
Trimellitic anhydride is used as a curing agent for epoxy and other resins. It is also used
in vinyl chloride plasticizers, various polymers and polyesters, agricultural chemicals,
dyes and pigments, paints and coatings, pharmaceuticals, surface active agents,
numerous modifiers and intermediates, and specialty chemicals. A number of epoxy
resin and surface coating systems, containing between 2-10% TMA, are available as dry
powder formulations and are intended for application either by electrostatic dry powder
spraying or by dipping pre-heated articles into fluidized beds. Primary occupational
exposure to TMA may occur from inhalation of airborne dust and fume when bulk
material is handled or during the application of dry powder formulations to surfaces. In
1978, NIOSH estimated that approximately 20,000 U.S. workers were at risk of exposure
to trimellitic anhydride in its various applications. The NIOSH 1972 National
Occupational Hazard Survey found 475 workers out of 3515 employed in nonmetallic
mineral products and engine electrical equipment industries were exposed to TMA. The
NIOSH 1982 survey found 97 workers of the total payroll of 269 in the printing ink
industry were exposed.
1.1.4. Physical properties and other descriptive information
| TMA |
(Refs. 5.12.-5.14.) |
|
|
|
|
| CAS no.: |
552-30-7 |
| synonyms: |
1,2,4-benzenetricarboxylic acid 1,2-anhydride; anhydro
trimellic acid; 1,2,4-benzenetricarboxylic acid anhydride;
1,2,4-benzenetricarboxylic acid, cyclic 1,2-anhydride; 1,2,4-benzenetricarboxylic anhydride; 4-caroxyphthalic anhydride; 1,3-dihydro-1,3-dioxo-5-isobenzofurancarboxylic
acid; 1,3-dioxo-5-phthalancarboxylic acid; NCI-C56633; 5-phthalanacarboxylic acid, 1,3-dioxo-; TMA; TMAN; trimellic
acid anhydride; trimellic acid 1,2-anhydride; trimellitic acid
cyclic 1,2-anhydride; trimellitic anhydride |
| structural formula: |
 |
| molecular wt: |
192.13 |
| melting point: |
161-163.5°C |
| boiling point: |
240-245°C at 1.9 kPa (14 mmHg); 390°C at 101.3 kPa (760 mmHg) |
| appearance: |
white solid |
| vapor pressure: |
5.3 × 10-7kPa
(4 × 10-6 mmHg) at 25°C; 0.3 kPa at 200°C; 2 kPa at 250°C; 11 kPa at 300°C |
| flash point: |
227°C by the closed-cup method |
| solubility: |
at 25°C, in g/100 g solvent: carbon tetrachloride 0.002;
ligroin 0.06; mixed xylenes 0.4; dimethylformamide 15.5;
acetone 49.6; ethyl acetate 21.6; 2-butanone 36.5;
cyclohexanone 38.4 |
|
|
|
|
| Derivatives (mixture of two isomers) (personal observations) |
|
|
|
|
| chemical name: |
N-(2,4-dicarboxybenzoyl)-N-(3,4-dimethoxybenzyl)amine
N-(2,5-dicarboxybenzoyl)-N-(3,4-dimethoxybenzyl)amine |
| synonyms: |
TMAVA |
| mixture melting point: |
133-138°C |
| solubility: |
soluble in dilute ammonium hydroxide, methanol (reacts slowly), acetone, chloroform |
| appearance: |
white crystalline solid |
| molecular wt: |
359.35 |
| structural formula: |
|
 |
The analyte air concentrations throughout this method are based on the recommended sampling and
analytical parameters.
1.2. Limit defining parameters
1.2.1. Detection limit of the analytical procedure
The detection limit of the analytical procedure is 0.30 ng per injection (10-µL injection of
a 0.030 µg/mL solution). This is the amount of analyte that will produce a peak with a
height that is approximately 5 times the baseline noise. (Section 4.1.)
1.2.2. Detection limit of the overall procedure
The detection limit of the overall procedure is 0.106 µg per sample (0.221 µg/m3). This
is the amount of analyte spiked on the sampling device that, upon analysis, produces a
peak similar in size to that of the detection limit of the analytical procedure. (Section 4.2.)
1.2.3. Reliable quantitation limit
The reliable quantitation limit is 0.299 µg per sample (0.623 µg/m3). This is the smallest
amount of analyte which can be quantitated within the requirements of a recovery of at
least 75% and a precision (±1.96 SD) of ±25% or better. (Section 4.3.)
The reliable quantitation limit and detection limits reported in the method are based upon
optimization of the instrument for the smallest possible amount of analyte. When the target
concentration of analyte is exceptionally higher than these limits, they may not be attainable at the
routine operating parameters.
1.2.4. Instrument response to the analyte
The instrument response over concentration ranges representing 0.5 to 2 times the target
concentration is linear. (Section 4.4.)
1.2.5. Recovery
The recovery of TMAVA from samples used in a 16-day ambient storage test remained
above 99%. (Section 4.5.)
1.2.6. Precision (analytical procedure)
The pooled coefficient of variation obtained from replicate determinations of analytical
standards at 0.5, 1 and 2 times the target concentration is 0.032. (Section 4.6.)
1.2.7. Precision (overall procedure)
The precision at the 95% confidence level for the 16-day ambient temperature storage
test is ±12.5%. (Section 4.7.) This includes an additional ±5% for sampling error.
1.2.8. Reproducibility
Six samples, collected from a test atmosphere of trimellitic anhydride, and a draft copy
of this procedure were given to a chemist unassociated with this evaluation. The
samples were analyzed after 2 days of storage at room temperature. No individual
sample result deviated from its theoretical value by more than the precision reported in
Section 1.2.7. (Section 4.8.)
2. Sampling Procedure
2.1. Apparatus
2.1.1. A personal sampling pump that can be calibrated within ±5% of the recommended flow
rate with the sampling device in line.
2.1.2. A four-piece polystyrene cassette
containing two coated glass fiber
filters assembled as shown.

2.1.3. Treated glass fiber filters each coated with 10 mg of veratrylamine (3,4-dimethoxybenzylamine) and 10 mg
of di-n-octyl phthalate (Kodak).
They are prepared as follows:
Weigh 0.4 g of veratrylamine and
0.4 g of di-n-octyl phthalate in a
scintillation vial. Add 16 mL of
methanol and shake well. Place 40
glass fiber filters on a clean glass
plate (a regular TLC plate). Apply
0.4 mL of the methanol solution to each filter. Air dry for 5 minutes. Place filters in a
wide-mouth jar. Dry under vacuum at room temperature overnight. Store the coated
filters in a refrigerator and use within a month. Filters stored longer than this period
developed several small interfering peaks (believed to be decomposition products of
veratrylamine) in the HPLC chromatogram.
2.2. Reagents
No reagents are required for sampling.
2.3. Technique
2.3.1. Prepare the sampler for open-face sampling by removing the top piece and the end plug
from the bottom piece. Attach the sampler to the sampling pump with a piece of flexible
tubing and place it in the worker's breathing zone with the open face of the cassette
facing down.
2.3.2. Replace the top piece and the end plug after sampling. Seal each sample with an official
Form OSHA-21.
2.3.3. Submit at least one blank with each set of samples. Handle the blank the same as the
other samples except draw no air through it.
2.3.4. List any potential interferences on the sample data sheet.
2.4. Sampler capacity
Sampling capacity is approximately 300 µg at a sampling rate of 2.0 L/min. This loading is
equivalent to an air volume of 3750 L at 2 times the target concentration. (Section 4.9.)
2.5. Extraction efficiency (Section 4.10.)
2.5.1. The average extraction efficiency of TMAVA from the treated glass fiber filters is 99%.
2.5.2. Extracted samples remain stable for at least 24 h.
2.6. Recommended air volume and sampling rate
2.6.1. For TWA samples the recommended air volume is 480 L at 2.0 L/min.
2.6.2. For short term samples the recommended air volume is 30 L at 2.0 L/min (15-min
samples).
2.6.3. When short term samples are required, the reliable quantitation limit becomes larger.
For example, the reliable quantitation limit is 9.97 µg/m3 when 30 L of air is collected.
2.7. Safety precautions (sampling)
Attach the sampling equipment to the worker in such a manner that it will not interfere with work
performance or safety. Follow all safety practices applicable to the work area.
3. Analytical Procedure
3.1. Apparatus
3.1.1. An HPLC equipped with a UV detector. A BAS 200 HPLC (Bioanalytical Systems) and
Waters WISP auto-sampler were used in this evaluation.
3.1.2. An HPLC column capable of separating TMAVA from any interferences. An Asahipak
C4P-50 column (4.6 × 250 mm) (a polymeric based C4 column, obtained from Keystone
Scientific) was used in this evaluation. A Nova-pak C18 column (8 × 100 mm) (Millipore
Waters) was used in the alternate HPLC conditions.
3.1.3. An electronic integrator or other suitable means of measuring detector response. A
Waters 860 Networking Computer System was used in this evaluation.
3.1.4. Sample vials, 4-mL glass, with poly(tetrafluoroethylene)-lined caps.
3.2. Reagents
3.2.1. Trimellitic anhydride. Trimellitic anhydride, 97%, was obtained from Aldrich. The bottle
was 10 years old, and the TMA content was analyzed to be 93.5%.
3.2.2. Water, HPLC grade. Water was obtained from a Millipore Milli-Q water purification
system.
3.2.3. Extraction solution, 0.02 N ammonium hydroxide. The extraction solution was prepared
by diluting 5.28 mL of concentrated ammonium hydroxide to 4 L with water.
3.2.4. Acetonitrile, HPLC grade. Acetonitrile, Optima grade, was obtained from Fisher
Scientific.
3.2.5. Phosphoric acid. Phosphoric acid, Baker Analyzed Reagent grade, was obtained from
J T Baker.
3.2.6. Veratrylamine. Veratrylamine, 97%, was obtained from Aldrich.
3.2.7. Ethyl acetate. Ethyl acetate, B&J Brand High Purity Solvent, was obtained from Baxter.
3.2.8. Toluene. Toluene, B&J Brand High Purity Solvent, was obtained from Baxter.
3.2.9. Acetone. Acetone, B&J Brand High Purity Solvent, was obtained from Baxter.
3.3. Standard preparation
3.3.1. Synthesis of TMAVA:
Dissolve TMA (97%, 1.98 g, 0.01 mole) in 50 mL of ethyl acetate. Dilute veratrylamine
(97%, 1.72 g, 0.01 mole) with 20 mL of ethyl acetate. With constant stirring, slowly add
the TMA solution to the veratrylamine solution. Sonicate the mixture for 1 h. Collect the
resulting white precipitate. The yield of the first crop should be approximately 81%.
Recrystallize from acetone/toluene. Melting point (capillary tube): 133°C-138°C.
3.3.2. Prepare stock standards by weighing 10-20 mg of TMAVA in 10-mL volumetric flasks
and diluting to volume with the extraction solution. Apply a factor of 0.5347 to the weight
of TMAVA to convert it to that of TMA. For example, 10 mg of TMAVA dissolved in 10
mL will give a standard stock solution representing 534.7 µg/mL of TMA.
(MW TMA)/(MW TMAVA) = 192.13/359.35 = 0.5347
3.3.3. Alternately, TMA of known purity may be weighed and derivatized with excess
veratrylamine in acetonitrile. Weigh 10-15 mg of TMA in a 10-mL volumetric flask. Add
2 mL of acetonitrile and 2 drops of veratrylamine. Sonicate for 15 min. Add the
extraction solvent to mark.
3.3.4. Prepare analytical standards by diluting the stock standards with extraction solvent. A
6.4 µg/mL standard solution corresponds to the target concentration.
3.3.5. Prepare a sufficient number of analytical standards to generate a calibration curve.
Analytical standard concentrations must bracket sample concentrations.
3.4. Sample preparation
3.4.1. Transfer the two filters to separate 4-mL glass vials.
3.4.2. Add 3.0 mL of the extraction solution to each vial.
3.4.3. Cap the vials and shake them on a mechanical shaker for 30 min.
3.5. Analysis
3.5.1. HPLC conditions
| column: |
Asahipak C4P-50 |
| eluent: |
acetonitrile/water/phosphoric acid, 25/75/0.1 |
| flow rate: |
0.8 mL/min |
| injection vol: |
10 µL |
| UV detector: |
205 nm |
| alternate l: |
232 nm |
| ret. times: |
trimellitic acid 7.1 min
TMAVA 12.8 min |

Figure 3.5.1. Chromatogram of a mixture of TMAVA (6.27 µg/mL) and trimellitic acid
(6.12 µg/mL). Key: (1) trimellitic acid, (2) TMAVA.
3.5.2. Alternate HPLC conditions
| column: |
Novapak C18 |
| eluent: |
acetonitrile/water/phosphoric acid, 22.5/77.5/0.1 |
| flow rate: |
1.0 mL/min |
| injection vol: |
10 µL |
| UV detector: |
205 nm |
| alternate l: |
232 nm |
| ret. times: |
trimellitic acid 1.75 min
TMAVA isomers 5.2 min and 6.6 min |
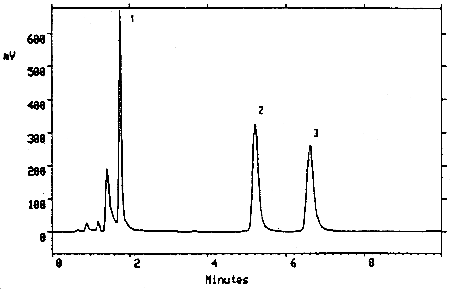
Figure 3.5.2. Chromatogram of a mixture of TMAVA (6.27 µg/mL) and
trimellitic acid (6.12 µg/mL) under the alternate analytical
conditions. Key: (1) trimellitic acid, (2) and (3) TMAVA isomers.
3.5.3. Construct a calibration curve using an external standard method by plotting micrograms
per milliliter versus detector response of standards. If the two isomers are separated,
sum the peak areas.
3.6. Interferences (analytical)
3.6.1. Any compound that absorbs at 205 nm and has a similar retention time as the analyte
is a potential interference. Generally, chromatographic conditions can be altered to
separate an interference from the analyte.
3.6.2. Retention time on a single column is not considered proof of chemical identity. Additional
means of identification could involve: analysis using an alternate HPLC column, detection
at another wavelength (peak ratioing), and GC/MS. The mass spectrum obtained from
GC/MS showed the base peak at m/e 297, representing the cyclic imide, apparently
produced through decarboxylation and cyclization in the GC.
3.7. Calculations
The analyte amount for a sample is obtained from the calibration curve in terms of micrograms
per milliliter uncorrected for extraction efficiency. The analyte amount is corrected by subtracting
the amount found in the blank. The air concentration is obtained by using the following equation.
| where |
A |
= |
micrograms of analyte per milliliter (blank corrected) |
|
B |
= |
extraction volume |
|
C |
= |
liters of air sampled |
|
D |
= |
extraction efficiency |
3.8. Safety precautions (analytical)
3.8.1. Restrict the use of all chemicals to a fume hood.
3.8.2. Avoid skin contact and inhalation of all chemicals.
3.8.3. Wear safety glasses, gloves and a lab coat at all times while working with chemicals.
4. Backup Data
4.1. Detection limit of the analytical procedure
The injection size recommended in the
analytical procedure (10 µL) was used in the
determination of the detection limit of the
analytical procedure. The detection limit of
0.30 ng on-column was determined by
analyzing a dilute standard of TMAVA (0.030
µg/mL). This amount gave a peak with a
height about 5 times the height of the baseline noise.

Figure 4.1. Detection limit of the analytical procedure. Key: (1) TMAVA.
4.2. Detection limit of the overall procedure
The detection limit of the overall procedure
was determined by analyzing filters liquid
spiked with 0.106 µg of TMAVA. This
amount corresponds to an air concentration
of 0.221 µg/m3. The injection size listed in
the analytical procedure (10 µL) was used in
the determination of the detection limit of the
overall procedure.
Table 4.2.
Detection Limit of the
Overall Procedure
|
| µg |
µg |
percent |
| spiked |
recovered |
recovered |
|
| 0.106 |
0.118 |
111.3 |
| 0.106 |
0.132 |
124.5 |
| 0.106 |
0.184 |
173.6 |
| 0.106 |
0.114 |
107.5 |
| 0.106 |
0.098 |
92.5 |
| 0.106 |
0.118 |
111.3 |
|
4.3. Reliable quantitation limit
The reliable quantitation limit was determined by analyzing filters liquid spiked with
0.299 µg of TMAVA. This amount corresponds to an air concentration of 0.623
µg/m3. The recovery of the analyte from the
spiked samples was greater than 75% with
a precision of ±25% or better.
Table 4.3.
Reliable Quantitation Limit
|
| µg |
µg |
percent |
| spiked |
recovered |
recovered |
|
| 0.299 |
0.296 |
99.0 |
| 0.299 |
0.293 |
98.0 |
| 0.299 |
0.292 |
97.7 |
| 0.299 |
0.294 |
98.3 |
| 0.299 |
0.285 |
95.3 |
| 0.299 |
0.283 |
94.6 |
|
| mean = 97.2% |
| SD = 1.8% |
| precision = 3.5% |
|
4.4. Instrument response to the analyte
The instrument response to TMAVA over the range of 0.5 to 2 times the target concentration was
determined from multiple injections of analytical standards. The response is linear with a slope
of 2.08 × 104 area counts per microgram per milliliter.
Table 4.4.
Instrument Response
|
| × target concn |
0.5× |
1.0× |
2.0× |
| µg/mL |
3.21 |
6.42 |
12.83 |
|
| area counts |
61041 |
131012 |
265178 |
|
66302 |
134085 |
266730 |
|
63616 |
136377 |
264288 |
|
65737 |
137022 |
262804 |
|
65851 |
121815 |
261018 |
|
65042 |
126993 |
265654 |
|
65042 |
126993 |
265654 |
|
| mean |
64598 |
131217 |
264279 |
|

Figure 4.4. Calibration curve for TMAVA.
4.5. Storage data
Storage samples were prepared from a test atmosphere of TMA aerosol. The generation of TMA
aerosol is described in detail in Section 4.9. Thirty-six samples were collected from the chamber
in two sessions. Six samples were analyzed immediately. The rest of the samples were divided
into two groups: 15 were stored in a refrigerator at 5°C, and the other 15 were stored in a closed
drawer at about 22°C. Six samples, three from each group, were analyzed at intervals over a
period of sixteen days. The average concentration of the day 0 samples, which was 53.6 µg/m3,
was set at 100% (no loss due to storage). The recovery of the analyte from the samples stored
at ambient temperature remained above 98.9% (from the regression line).
Table 4.5.
Storage Test
|
days of
storage |
% recovery
(ambient) |
|
% recovery
(refrigerated) |
|
0
0
3
6
9
14
16 |
102.2
98.7
94.3
101.3
105.5
98.7
96.4 |
98.2
104.8
104.1
109.5
102.0
98.9
92.8 |
97.3
98.7
95.2
99.9
100.4
100.8
97.8 |
|
102.2
98.7
102.7
98.2
111.6
97.5
93.6 |
98.2
104.8
94.0
111.6
99.6
97.3
96.8 |
97.3
98.7
108.3
103.4
100.4
99.2
102.9 |
|

Figure 4.5.1. Storage test at ambient temperature.

Figure 4.5.2. Storage test at reduced temperature (5°C).
4.6. Precision (analytical method)
The precision of the analytical procedure is
defined as the pooled coefficient of variation
determined from replicate injections of
analytical standards representing 0.5, 1, and
2 times the target concentration. The coefficients of variation are calculated from the
data in Table 4.4. The pooled coefficient of
variation is 0.032.
Table 4.6.
Precision of the Analytical Method
(Based on the Data of Table 4.4.)
|
| × target concn |
0.5× |
1.0× |
2.0× |
| µg/mL |
3.21 |
6.42 |
12.83 |
|
| SD1 |
1979 |
5913 |
2074 |
| CV |
0.031 |
0.045 |
0.008 |
|
| 1 - in area counts |
4.7. Precision (overall procedure)
The precision of the overall procedure is determined from the storage data. The determination
of the standard error of estimate (SEE) for a regression line plotted through the graphed storage
data allows the inclusion of storage time as one of the factors affecting overall precision. The SEE
is similar to the standard deviation, except it is a measure of dispersion of data about a regression
line instead of about a mean. It is determined with the following equation:

| where |
n |
= |
total number of data points |
|
k |
= |
2 for linear regression |
|
k |
= |
3 for quadratic regression |
|
Yobs |
= |
observed percent recovery at a given time |
|
Yest |
= |
estimated percent recovery from the regression line at the same given time |
An additional 5% for pump error is added to the SEE by the addition of variances. The precision
at the 95% confidence level is obtained by multiplying the SEE (with pump error included) by 1.96
(the z-statistic from the standard normal distribution at the 95% confidence level). The 95%
confidence intervals are drawn about their respective regression line in the storage graph as
shown in Figure 4.5.1. The data for Figure 4.5.1. were used to determine the SEE of ±6.39% and
the precision of the overall procedure of ±12.5%.
4.8. Reproducibility data
Six samples were collected from a test
atmosphere of trimellitic anhydride aerosol
at 25°C and 80% RH. A draft copy of this
method and the samples were submitted to
a chemist unassociated with this evaluation
for analysis. All of the sample results were
within the range of ±12.5%, the precision of
the overall procedure.
Table 4.8.
Reproducibility Data
|
| µg/m3 |
µg/m3 |
percent |
percent |
| expected |
found |
found |
deviation |
|
| 40.9 |
40.8 |
99.8 |
-0.2 |
| 40.9 |
41.4 |
101.2 |
+ 1.2 |
| 40.9 |
40.9 |
100.0 |
0.0 |
| 40.9 |
41.0 |
100.2 |
+ 0.2 |
| 40.9 |
40.4 |
98.8 |
-1.2 |
| 40.9 |
40.7 |
99.5 |
-0.5 |
|
4.9. Sampler capacity
The sampler capacity was assessed by
sampling from a dynamically generated
test atmosphere of TMA at 25°C and 80%
RH. The test atmosphere of TMA aerosol
was generated by pumping an ethyl
acetate solution of TMA at a rate of 0.8
mL/min through a TSI Model 3076
atomizer (TSI Incorporated, St. Paul, MN),
where it was dispersed with an air stream
of 3.5 L/min. The aerosol passed through
an electrostatic charge neutralizer and
was mixed with a dilution air stream of 97
L/min (25°C, 80% RH). The diluted aerosol flowed into a chamber containing 18
sampling ports. Each port was fitted with
a sampler. After an appropriate
equilibration time, sampling was started at
2 L/min. Three samplers were removed
every hour. Both the front and the back
filters were analyzed for TMAVA and trimellitic acid. Many experiments were run, with different
concentrations of TMA feed solution ranging from 60 µg/mL to 1500 µg/mL (actual TMA content
of the feed solution was 93.5%, with 6.5% trimellitic acid). The "total" aerosol concentration (sum
of trimellitic acid and trimellitic anhydride) obtained ranged from 0.08 to 2.3 mg/m3. No TMAVA
or trimellitic acid was detected on the back filter after 6 h of sampling at a total concentration of
2.3 mg/m3. The front filters contained TMAVA and trimellitic acid in a ratio of 90/10 indicating that
the aerosol contained approximately 90% trimellitic anhydride and 10% trimellitic acid. Apparently
some of the TMA (about 3.5%) in the feed solution was hydrolyzed by the humid air during the
short path from the atomizer to the test chamber. This 90/10 ratio remained constant at lower
concentrations, but began to decrease at higher concentrations and longer sampling time,
indicating that some of the TMA collected on the filter at later stages failed to be derivatized. The
plot of the amount of TMAVA collected against the total amount collected (TMAVA and trimellitic
acid) showed that the "failure point" occurred at about 300 µg of TMAVA. Beyond this point, some
of the TMA deposited on the filter was hydrolyzed by the water in the air before it could react with
the derivatizing agent. This failure point is equivalent to 3750 L at 2 times the target
concentration.

Figure 4.9. Sampler capacity. Data from five different runs covering
high and low concentration ranges.
4.10. Extraction efficiency and stability of extracted samples
4.10.1. Extraction efficiency
To determine the extraction efficiency, six glass fiber filters were
liquid spiked with TMAVA at the
target concentration. These samples were stored overnight at ambient temperature and then extracted
with the extraction solvent and analyzed. The average recovery was
99.0%.
Table 4.10.1.
Extraction Efficiency
|
sample
no. |
µg
spiked |
µg
recovered |
% recovery |
|
| 1 |
19.09 |
19.21 |
100.6 |
| 2 |
19.09 |
18.84 |
98.7 |
| 3 |
19.09 |
18.67 |
97.8 |
| 4 |
19.09 |
19.12 |
100.2 |
| 5 |
19.09 |
17.94 |
94.0 |
| 6 |
19.09 |
19.54 |
102.4 |
|
| mean |
|
99.0 |
|
4.10.2. Stability of extracted samples
The stability of extracted samples
was ascertained by reanalyzing the
above samples 24 h later with fresh
standards. The samples were
stored at room temperature and
were not recapped. The average
recovery was 99.2%.
Table 4.10.2.
Stability of Extracted Samples
|
initial
recovery (%) |
recovery after
24 h (%) |
percent
change |
|
| 100.6 |
100.4 |
-0.2 |
| 98.7 |
100.1 |
+ 1.4 |
| 97.8 |
99.7 |
+ 1.9 |
| 100.2 |
99.9 |
-0.3 |
| 94.0 |
97.4 |
+ 3.4 |
| 102.4 |
97.6 |
-4.8 |
|
5. References
5.1. Purnell, C.J. and C.J. Warwick, J. High Resolut. Chromatogr. Chromatogr. Commun., 1980, 3,
482-484.
5.2. Palassis, J., J.C. Posner, E. Slick and K. Schulte, Am. Ind. Hyg. Assoc. J., 1981, 42(11), 785.
5.3. Geyer, R., R.C. Jones, and N. Mezin, J. High Resolut. Chromatogr. Chromatogr. Commun., 1986,
9(5), 308-309.
5.4. Ledbetter, A.D., C.L. Leach, N.S. Hatoum and J-C. Roger, Am. Ind. Hyg. Assoc. J., 1987, 48(1),
35-38.
5.5. NIOSH Manual of Analytical Methods, 3rd ed., U.S. Department of Health and Human Services,
Center for Disease Control, National Institute of Occupational Safety and Health; DHHS (NIOSH)
Publication No. 90-121, U.S. Government Printing Office, Washington, DC, 1990; Method No.
P&CAM 322.
5.6. Rushing, L.G., J.R. Althaus, and H.C. Thompson, Jr., J. Anal. Toxicol., 1982, 6, 290-293.
5.7. Towle, P.H., et al., in Kirk-Othmer Encyclopedia of Chemical Technology, Standen, A., Ed., 2nd
ed., John Wiley & Sons, New York, NY, 1968; Vol. 15, p 473.
5.8. "Notice of Intended Change - Trimellitic Anhydride", Appl. Occup. Environ. Hyg., 1991, 6(7), 625.
5.9. Current Intelligence Bulletin 21: Trimellitic Anhydride (TMA), U.S. Department of Health,
Education, and Welfare, Center for Disease Control, National Institute of Occupational Safety and
Health, DHEW (NIOSH) Publication No. 78-121, U.S. Government Printing Office, Washington,
DC, 1978.
5.10. "Table Z-1-A -- Limits for Air Contaminants", Code of Federal Regulations, Title 29; 1910.1000,
U.S. Office of the Federal Register National Archives and Records Administration, Washington,
DC; 1991.
5.11. OSHA Computerized Information System Database, OSHA Regulated Substances (PEL
Standard) Profiles, Trimellitic Anhydride, Revision Date: 08/15/88, OSHA SLTC, Salt Lake City,
UT.
5.12. Merck Index, Budavari, S., Ed., 11th ed., Merck & Co., Rahway, NJ, 1989; p 1386.
5.13. Registry of Toxic Effects of Chemical Substances, Sweet, D.V., Ed.,1985-86 edition, U.S.
Department of Health and Human Services, DHHS (NIOSH) Publication No. 87-114, Government
Printing Office, Washington, DC, 1987; Vol. 1, p 851.
5.14. Bemis, A.G., et al., in Kirk-Othmer Encyclopedia of Chemical Technology, Grayson, M., et al., Ed.,
3rd ed., John Wiley & Sons, New York, NY, 1982; Vol. 17, p 764.
|

