|
ISOPROPYL ALCOHOL
| Method number: |
109 |
|
| Matrix: |
Air |
|
| Target concentration: |
400 ppm (983 mg/m3) |
|
| OSHA PEL: |
400 ppm (983 mg/m3) TWA |
|
| ACGIH TLV: |
400 ppm (983 mg/m3) TWA |
|
|
500 ppm (1230 mg/m3) ceiling |
|
| Procedure: |
Samples are collected by drawing a known volume of air through two
8-mm o.d. (6-mm i.d.) Anasorb® 747 tubes in series. The front tube
contains 400 mg of adsorbent, and the back tube 200 mg. Samples
are desorbed with a 60/40 N,N-dimethylformamide/carbon disulfide solution and analyzed by GC using a flame ionization detector (FID). |
|
|
Recommended air volume
and sampling rate: |
18 L at 0.05 to 0.2 L/min |
|
| Reliable quantitation limit: |
44.4 ppb (108.9 µg/m3) |
|
Standard error of estimate
at the target concentration: |
5.15% |
|
| Special requirement: |
The air sampler must be separated into its component sampling tubes
as soon as possible after sampling. This will prevent post-sampling migration. |
|
| Status of method: |
Evaluated method. This method has been subjected to the
established evaluation procedures of the Organic Methods Evaluation Branch. |
|
| Date: June 1997 |
Chemist: Mary Eide |
Organic Methods Evaluation Branch
OSHA Salt Lake Technical Center
Salt Lake City, UT 84115-1802
1. General Discussion
1.1 Background
1.1.1 History
The previous method used by OSHA for the collection and analysis of isopropyl alcohol
was based on NIOSH Method 1400 (Ref. 5.1). It required collection of isopropyl alcohol on
coconut shell charcoal tubes, sample refrigeration for shipment and storage, and analysis
to be performed as soon as possible. The samples were desorbed with 99/1 carbon
disulfide/2-butanol, and analyzed by GC/FID. Because 2-butanol is often present in many
formulations with isopropyl alcohol, SLTC developed a different desorbing solution
consisting of 99/1 carbon disulfide/N,N-dimethylformamide
(CS2/DMF). The presence of
DMF effectively facilitated isopropyl alcohol desorption, resulting in a recovery of 92% over
the range of 0.3 to 6 mg, for dry samples. A subsequent study (Ref. 5.2) performed by
spiking charcoal tubes with 6 mg isopropyl alcohol, and drawing 3 L of humid air (80%
relative humidity (RH) at 22°) through the tubes at 0.05 L/min, showed a lower recovery,
whether 99/1 carbon disulfide/2-butanol (77% recovery after desorption correction) or 99/1
CS2/DMF (68% recovery after desorption correction) was used for the desorbing solvent
(Ref. 5.2). This reduction in recovery was apparently due to the observed presence of a
small water phase in these samples. A portion of the isopropyl alcohol had apparently
dissolved in the water phase, thus reducing the amount of isopropyl alcohol present in the
desorption solvent phase, which was injected upon analysis. There was some increase in
recovery when a drying agent, 200 mg of anhydrous MgSO4, was added, using 99/1
CS2/DMF (88% recovery after desorption correction).
Increasing the amount of DMF in the desorbing solvent to a 60/40 DMF/CS2
mixture allowed the water to be dissolved and good recoveries were obtained, 99.7% recovery from tubes that
had 3 L of humid air drawn through them (81% RH at 21°). Storage studies using both the NIOSH and OSHA
charcoal tube methods revealed that isopropyl alcohol migrated from the front section to
the back section upon storage.
The sampling procedures of OSHA Methods 91 and 100 for methyl and ethyl alcohols
respectively require collection on two Anasorb® 747 tubes in series (Refs. 5.3 and 5.4).
The front tube contains 400 mg of adsorbent and the back tube contains 200 mg. The
recommended sampling rate for both procedures is 0.05 L/min, with recommended air
volumes of 3 L for methyl alcohol and 12 L for ethyl alcohol. Because isopropyl alcohol is
often found in formulations with either methyl alcohol or ethyl alcohol or both alcohols, this
collection procedure was explored for isopropyl alcohol. Capacity studies indicated that
isopropyl alcohol can be collected at the 0.05 L/min sampling rate or at a higher sampling
rate of 0.2 L/min for a recommended sample volume of 18 L. Because methyl alcohol and
ethyl alcohol were previously evaluated at 0.05 L/min, additional capacity tests with these
analytes were performed at 0.2 L/min. These tests indicate maximum air volumes of 2 L
for methyl alcohol and 11 L for ethyl alcohol, which are lower than those previously obtained
at 0.05 L/min and these lower maximum air volumes should be used if the higher flow rate
of 0.2 L/min is used. (Section 4.12)
In OSHA Methods 91 and 100, Anasorb® 747 tubes are desorbed with
60/40 DMF/CS2.
The high concentration of DMF effectively prevents a water phase in desorbed samples, by
keeping the collected water in solution. Because
99/1 CS2/DMF is commonly used at SLTC
for charcoal tube analysis, this solvent was first evaluated as a possible desorbing solvent.
A recovery study was performed with samples prepared from humid test atmospheres (80%
RH at 21°) on 400/200 mg Anasorb® 747 tubes, with desorption using 3 mL of 99/1
CS2/DMF. As anticipated, there was a significant
loss in the recovery, and a water layer was observed to be floating on top of the
99/1 CS2/DMF. When the water layer was
removed and analyzed, isopropyl alcohol was found, indicating that the
99/1 CS2/DMF could not be used for the
desorbing solvent. The desorbing solvent of 60/40 DMF/CS2 was
therefore used in this method for all studies, because it puts any collected water into
solution, eliminating any water phase.
As in Methods 91 and 100, due to migration problems, it is necessary to sample for
isopropyl alcohol using the two tube sampling train of a 400-mg tube followed by a 200-mg
tube. These tubes need to be separated, capped, and sealed immediately after sampling
to avoid migration of the analyte from the front (400 mg) tube to the back (200 mg) tube.
A migration study at ambient and refrigerated temperatures, and low (11% RH) and high
(80% RH) humidity, using a 400/200 mg tube was performed at the 405-ppm level, for 23
days. The highest amount of migration, 8.7%, occurred with the tubes collected at low
humidity and were stored at ambient temperature. (Section 4.7.2)
1.1.2. Toxic effects (This section is for information only and should not be taken as the basis of
OSHA policy.) (Ref. 5.5)
The OSHA PEL for isopropyl alcohol is 400 ppm based on an 8-h TWA. Workers exposed
to 400 ppm reported mild irritation of the eyes, nose, and throat, with an increase in the
intensity of these symptoms when the exposure level increased to 800 ppm. Isopropyl
alcohol is readily absorbed through mucous membranes and through ingestion, but less
through skin absorption. In rabbits, an eye exposure of 0.1 mL of 70% isopropyl alcohol in
water (the formulation of rubbing alcohol) caused conjunctivitis, iritis, and cornea1 opacity.
In animal studies, isopropyl alcohol was found to be 2-3 times more potent than ethyl
alcohol as a CNS depressant, and its metabolite, acetone, was also a CNS depressant.
Isopropyl alcohol is metabolized by the liver to acetone, which can be smelled on the breath,
and found in the urine. An exposure of 12,000 ppm isopropyl alcohol for 8 hours killed half
of the rats tested. The oral lethal dose of isopropyl alcohol in humans is 240 mL, with
poisoning occurring at 20 mL. Most fatalities from isopropyl alcohol are due to alcoholics
drinking rubbing alcohol, mistaking it for ethyl alcohol. Isopropyl alcohol has moderate
narcotic properties, similar to ethyl alcohol. Symptoms of isopropyl alcohol ingestion
manifest themselves within 30 minutes of ingestion, and include pain in the GI tract,
vomiting, diarrhea, headache, muscular incoordination, ataxia, confusion, and in high doses
coma and death.
1.1.3. Workplace exposure
Isopropyl alcohol was ranked number 50 in the list of the top 50 chemicals produced in the
United States for 1994, with 1.39 billion pounds produced. (Ref. 5.6). Isopropyl alcohol is
used in the synthesis of acetone, glycerin, glycerol, and isopropyl acetate, and as a solvent
for gums, shellac, essential oils, creosote, and resins. It is used in anti-freeze formulations,
quick-drying inks and oils, as a denaturant for ethyl alcohol, cosmetics, after shaves, hand
lotions, and hair care products. A 70% solution in water is known as rubbing alcohol, and
is used as a body rub and disinfectant. (Refs. 5.5 and 5.7)
1.1.4. Physical properties and other descriptive information (Refs. 5.7 and 5.8)
| CAS number: |
67-63-0 |
| molecular weight: |
60.09 |
| boiling point, °: |
82.5 |
| melting point, °: |
-88.5 |
| color: |
clear |
| specific gravity: |
0.78505204 |
| molecular formula: |
C3H8O |
| vapor pressure (@25°): |
6.02 kPa (33 mmHg) |
| vapor density: |
2.1 |
| odor: |
like ethyl alcohol and acetone mixture |
| flash point: |
11.7° (CC) |
| autoignition temperature: |
455.6° (852°) |
| explosive limits: |
upper: 12.7%; lower: 2.5% (v/v) |
| solubility: |
soluble in water, alcohol, ether, chloroform, acetone, benzene,
toluene, carbon disulfide, and many other organic solvents |
| synonyms: |
dimethyl carbinol; isopropanol; Petrohol; 2-propanol |
| structural formula: |
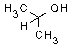 |
The analyte air concentrations throughout this method are based on the recommended sampling
and analytical parameters. Air concentrations listed in ppm and ppb are referenced to 25° and
101.3 kPa (760 mmHg).
1.2 Limit defining parameters
1.2.1 Detection limit of the analytical procedure
The detection limit of the analytical procedure is 7.05 pg. This is the amount of analyte that
will give a response that is significantly different from the background response of a reagent
blank. (Sections 4.1 and 4.2)
1.2.2 Detection limit of the overall procedure
The detection limit of the overall procedure is 0.587 µg per sample
(13.3 ppb or 32.6 µg/m3). This is the amount of
analyte spiked on the sampler that will give a response that is significantly
different from the background response of a sampler blank. (Sections 4.1 and 4.3)
1.2.3 Reliable quantitation limit
The reliable quantitation limit is 1.96 µg per sample (44.4 ppb or
108.9 µg/m3). This is the amount of
analyte spiked on a sampler that will give a signal that is considered the
lower limit for precise quantitative measurements. (Section 4.4)
1.2.4 Precision (analytical procedure)
The precision of the analytical procedure, measured as the pooled relative standard
deviation over a concentration range equivalent to the range of 0.5 to 2 times the target
concentration is 0.61% for 18 L sample loadings. (Section 4.5)
1.2.5 Precision (overall procedure)
The precision of the overall procedure at the 95% confidence level for the ambient
temperature 19-day storage test (at the target concentration) is ±10.1%. This includes an
additional 5% for sampling error. (Section 4.6)
1.2.6 Recovery
The recovery of isopropyl alcohol from samples used in the 19-day storage test remained
above 100.2%. (Section 4.7)
1.2.7 Reproducibility
Six samples collected from a test atmosphere with a draft copy of this procedure, were
submitted for analysis to one of the OSHA Salt Lake Technical Center's service branch
laboratories. The samples were analyzed after 3 days of storage at 4°. No individual
sample result deviated from its theoretical value by more than the precision reported in
Section 1.2.5. (Section 4.8)
2. Sampling Procedure
2.1 Apparatus
2.1.1 Samples are collected using a personal sampling pump calibrated, with the sampling
device attached, to within 25± at the recommended flow rate.
2.1.2 Samples are collected using a 400-mg and a 200-mg Anasorb® 747 sampling tube. The
sampling tubes (11-cm × 8-mm o.d. × 6-mm i.d.) are connected in series with silicone tubing
prior to sampling. The adsorbent beds are held in place with a glass wool plug at the front,
and a foam plug at the rear of the adsorbent bed. For this evaluation, commercially prepared
sampling tube sets were purchased from SKC, Inc. (catalog no. 226-82).
2.2 Reagents
None required.
2.3 Technique
2.3.1 Break off both ends of the sampling tubes immediately before sampling. The holes on the
broken ends of the sampling tubes should be approximately one-half the i.d. of the sampling
tube. All tubes should be from the same lot. Connect the outlet end of a 400-mg sampling
tube to the inlet end of a 200-mg tube with a 1-in. length of 1/4-in. i.d. silicone rubber tubing.
The inlet end of a sampling tube is the end with the glass wool plug. Insure that the
connection is secure and that the broken ends of the tubes just touch each other. Be
careful not to cut the silicone tubing with the sharp ends of the sampling tubes.
2.3.2 Attach the sampling tube to the sampling pump with flexible, non-crimpable tubing. It is
desirable to utilize sampling tube holders which have a protective cover to shield the
employee from the sharp, jagged end of the sampling tube. Position the tube so that the
sampled air first passes through the inlet of the larger 400-mg tube.
2.3.3 Sampled air should not pass through any hose or tubing before entering the sampling tube.
2.3.4 Attach the sampler vertically with the larger, 400-mg tube, pointing downward in the
worker's breathing zone to avoid channeling. Position the sampler so it does not impede
work performance or safety.
2.3.5 Remove the sampling tubes after sampling for the appropriate time. Separate the two
sampling tubes and cap the tube ends with plastic end caps. Wrap each sample end-to-end
with a Form OSHA-21 seal. The silicone tubing used to connect the tubes is susceptible
to cuts from the sharp ends of the sampling tubes and should be discarded after one use.
2.3.6 Submit at least one blank sample with each set of samples. Handle the blank sampling tube
in the same manner as the other samples, except draw no air through it.
2.3.7 Record sample air volumes (in liters) for each sample, along with any potential
interferences.
2.3.8 Ship any bulk sample(s) in a container separate from the air samples.
2.3.9 Submit the samples to the laboratory for analysis as soon as possible after sampling. If
delay is unavoidable, store the samples at reduced temperature in a refrigerator or freezer.
2.4 Sampler capacity
Sampler capacity is determined by measuring how much air can be sampled before the analyte
breaks through the sampler, i.e., the sampler capacity is exceeded. Breakthrough is considered to
occur when the effluent from the sampler contains a concentration of analyte that is 5% of the
upstream concentration (5% breakthrough). Testing for breakthrough was performed by using an
FID to monitor the effluent from 400 mg Anasorb® 747 tubes. Dynamically generated test
atmospheres, which were about two times the target concentration, were used for the capacity
tests. The samples were collected at 0.05 and 0.2 L/min and the relative humidity was about 80%
at 22°. The 5% breakthrough air volume was calculated from the data of duplicate
determinations at 0.05 and 0.2 L/min and differing humidities. The breakthrough determinations
at high humidity are 22.6 L for 0.2 L/min at 84% RH and 29.0 L for 0.05 L/min at 77% RH. The
breakthrough determinations at low humidity are 29.8 L for 0.2 L/min at 12% RH and 37.6 L for
0.05 L/min at 13% RH. (Section 4.9)
2.5 Desorption efficiency
2.5.1 The average desorption efficiency for isopropyl alcohol from Anasorb® 747 over the range
of 0.5 to 2.0 times the target concentrations was 103.6%. (Section 4.10)
2.5.2 The average desorption efficiency for isopropyl alcohol from Anasorb® 747 for 0.05, 0.1
and 0.2 times the target concentration were 100.2%, 102.9%, and 103.6% respectively. (Section
4.10)
2.5.3 Desorbed samples remain stable for at least 64 h. (Section 4.10)
2.5.4 Desorption efficiencies should be confirmed periodically because differences may occur
due to variations between sampling media lots, desorption solvent, and operator technique.
2.6 Recommended air volume and sampling rate
2.6.1 For long-term samples, collect 18 L at 0.05 to 0.2 L/min. The 18-L maximum air volume
is based on sampler capacity studies, which indicate that at 0.2 L/min the maximum air
volume is 18 L, and that at the lower flow rate of 0.05 L/min the maximum air volume is 23.8 L.
2.6.2 For short-term samples, collect 3 L at 0.2 L/min.
2.6.3 When short-term samples are collected, the air concentration equivalent to the reliable
quantitation limit becomes larger. For example, the reliable quantitation limit is 0.266 ppm
(0.653 mg/m3) when 3 L is sampled.
2.6.4 When sampling for methyl alcohol and/or ethyl alcohol in conjunction with isopropyl
alcohol, the sampling rate and air volumes recommended in Methods 91 and 100 should be used.
Samples collected for methyl alcohol, in conjunction with ethyl alcohol and/or isopropyl
alcohol should be sampled at 0.05 L/min for 3 L. Samples collected for ethyl alcohol, in
conjunction with isopropyl alcohol should be sampled at 0.05 L/min for 12 L.
2.7 Interferences (sampling)
2.7.1 There are no known compounds that will severely interfere with the collection of isopropyl
alcohol on Anasorb® 747. In general, the presence of other contaminant vapors in the air
will reduce the capacity of Anasorb® 747 to collect isopropyl alcohol.
2.7.2 Suspected interferences should be reported to the laboratory with submitted samples.
2.8 Safety precautions (sampling)
2.8.1 The sampling equipment should be attached to the worker in such a manner that it will not
interfere with work performance or safety.
2.8.2 All safety practices that apply to the work area being sampled should be followed.
2.8.3 Protective eyewear should be worn when breaking the ends of the glass sampling tubes.
3. Analytical Procedure
3.1 Apparatus
3.1.1 A gas chromatograph equipped with an FID. For this evaluation, a Hewlett-Packard 5890A
Series II Gas Chromatograph equipped with a 7673A Automatic Sampler was used.
3.1.2 A GC column capable of separating isopropyl alcohol from the desorption solvent, internal
standard and any potential interferences. A 60-m × 0.32-mm i.d. capillary DB-5 with a
1.0-µm df (J&W Scientific, Folsom, CA) was used in the evaluation.
3.1.3 An electronic integrator or some other suitable means of measuring peak areas. A Waters
860 Networking Computer System was used in this evaluation.
3.1.4 Glass vials with poly(tetrafluoroethylene)-lined caps. For this evaluation 4-mL vials were
used to hold the samples for desorption, and 2-mL vials were used in the autosampler for
analysis.
3.1.5 A dispenser capable of delivering 3.0 mL of desorbing solvent to prepare standards and
samples. If a dispenser is not available, a 3.0-mL volumetric pipet may be used.
3.1.6 Disposable pipets, Pasteur-type, for the transfer of samples and standards from the 4-mL
vials to the 2-mL GC autosampler vials for analysis.
3.2 Reagents
3.2.1 Isopropyl alcohol, reagent grade or better. EM OMNISOLV isopropyl alcohol (Lot 9051)
was used in this evaluation.
3.2.2 Desorbing solution, 60/40 (v/v) DMF/CS2, reagent
grade or better. EM OMNISOLV CS2
(Lot 34279) and Baxter B&J Brand, High Purity Solvent DMF (Lot BB087) were used in this
evaluation. p-Cymene (1µL/mL) (Aldrich Lot 05104CP) was added for use as an internal
standard for this method. The large amount of DMF is necessary to eliminate any water
phase from water vapor collected on the air samples.
3.2.3 GC grade nitrogen, air, and hydrogen.
3.3 Standard preparation
3.3.1 Prepare working analytical standards by injecting microliter amounts of isopropyl alcohol
into 4-mL vials containing 3 mL of desorption solvent delivered from the same dispenser used
to desorb samples. An analytical standard equivalent to a 391-ppm air sample, with the
recommended air volume of 18 L, was prepared by placing 22 µL of isopropyl alcohol into
a sealed vial containing 3.0 mL of desorbing solution. An analytical standard equivalent to
a 426-ppm air sample, at the recommended air volume of 3 L, was prepared by placing 4µL of
isopropyl alcohol into a sealed vial containing 3.0 mL of desorbing solution for a concentration
of 1.05 mg/mL.
3.3.2 Bracket sample concentrations with working standard concentrations. If sample
concentrations are higher than the concentration range of prepared standards, prepare and
analyze additional standards, at least as high a concentration as the highest sample, to ascertain
the linearity of response, or dilute the sample with the desorbing solvent to obtain a
concentration within the existing standard range.
3.4 Sample preparation
3.4.1 Remove the plastic end caps from the sample tubes and carefully transfer the adsorbent
to separate 4-mL vials. Discard the glass tube, urethane foam plug and glass wool plug.
3.4.2 Add 3.0 mL of desorption solvent to each vial using the same dispenser as used for
preparation of standards.
3.4.3 Immediately seal the vials with poly(tetrafluoroethylene)-lined caps.
3.4.4 Shake the vials vigorously several times during the next 1 hour.
3.5 Analysis
3.5.1 Transfer an aliquot of each of the standards and samples into separate GC autosampler
vials if necessary.
3.5.2 Analytical conditions
| GC conditions |
|
| zone |
|
| temperatures: |
60° (column), hold 4 min, ramp at 10°/min to 160°, hold 4 min
210° (injector)
225° (detector) |
| run time: |
18 min |
| column gas flow: |
2.9 mL/min (hydrogren) |
| septum purge: |
1.9 mL/min (hydrogen) |
| injection size: |
1.0 µL (19:1 split) |
| column: |
60-m x 0.32-mm i.d. capillary DB-5 (1.0 µm df) |
| retention times: |
4.4 min (isopropyl alcohol)
16.6 min (p-cymene) |
|
| FID conditions |
|
| hydrogen flow: |
38 mL/min |
| air flow: |
450 mL/min |
| makeup flow: |
30 mL/min (nitrogen) |
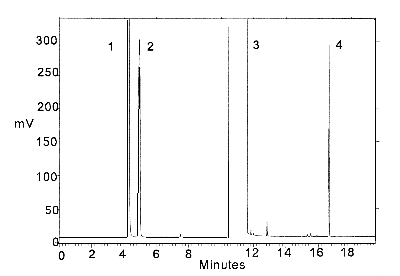
Figure 3.5.2. Chromatogram of a standard.
Peak identification: (1) isopropyl alcohol,
(2) carbon disulfide, (3) DMF, (4) p-cymene.
3.5.3 An internal standard (ISTD) calibration method is used. A calibration curve can be
constructed by plotting ISTD-corrected response of standard injections versus milligrams of
analyte per sample. Bracket the samples with freshly prepared analytical standards over a range
of concentrations.
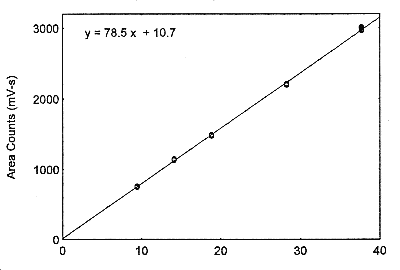
Amount (mg/sample)
Figure 3.5.3. The calibration curve at the target concentration with a 18-L air volume, made from
data of Table 4.5.2.
3.6 Interferences (analytical)
3.6.1 Any compound that produces an FID response and has a similar retention time as the
analyte or internal standard is a potential interference. If any potential interferences were
reported, they should be considered before the samples are desorbed. Generally,
chromatographic conditions can be altered to separate an interference from the analyte.
3.6.2 When necessary, the identity or purity of an analyte peak may be confirmed with
additional analytical data, such as identification by GC/mass spectrometry (Section 4.11).
3.7 Calculations
The amount of isopropyl alcohol per sample is obtained from the appropriate calibration curve in
terms of milligrams per sample, uncorrected for desorption efficiency. The back (200-mg) tube is
analyzed primarily to determine if there was any breakthrough from the front (400-mg) tube
during sampling. If a significant amount of analyte is found on the back tube (e.g., greater than
25% of the amount found on the front section), this fact should be reported with the sample
results. If any analyte is found on the back tube, it is added to the amount on the front tube. This
amount is then corrected by subtracting the total amount (if any) found on the blank. The air
concentration is calculated using the following formulae.
| mg/m3 = |
milligrams of analyte per sample
liters of air sampled × desorption efficiency × m3/1000 L |
| ppm = |
24.46 × mg/m3
molecular weight of analyte |
| where |
24.46 is the molar volume at 25° and 101.3 kPa (760 mmHg)
60.09 = molecular weight of isopropyl alcohol |
3.8 Safety precautions (analytical)
3.8.1 Adhere to the rules set down in your Chemical Hygiene Plan.
3.8.2 Avoid skin contact and inhalation of all chemicals.
3.8.3 Wear safety glasses, gloves and a lab coat while in the laboratory areas and working with
chemicals.
4. Backup Data
4.1 Determination of detection limit
Detection limits, in general, are defined as the amount (or concentration) of analyte that gives a
response (YDL) that is significantly different
(three standard deviations (SDBR)) from the
background response (YBR).
YDL - YBR = 3(SDBR)
The measurement of YBR and SDBR in chromatographic methods is typically inconvenient
and difficult because YBR is usually extremely low. Estimates of these parameters can be made
with data obtained from the analysis of a series of analytical standards or samples whose
responses are in the vicinity of the background response. The regression curve obtained for a
plot of instrument response versus concentration of analyte will usually be linear. Assuming
SDBR and the precision of the data about curve are similar, the standard error of estimate (SEE)
for the regression curve can be substituted for SDBR in the above equation. The following
calculations derive a formula for DL:
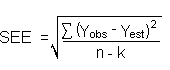
| Yobs |
= |
observed response |
| Yest |
= |
estimated response from regression curve |
| n |
= |
total number of data points |
| k |
= |
2 for linear regression curve |
At point YDL on the regression curve
| YDL = A(DL) + YBR |
A = analytical sensitivity (slope) |
therefore
Substituting 3(SEE) + YBR for YDL gives
4.2 Detection limit of the analytical procedure (DLAP)
The DLAP is measured as the mass of analyte actually introduced into the chromatographic
column. Ten analytical standards were prepared in equal descending increments with the
highest standard containing 3.4 µg/mL of isopropyl alcohol. This is the concentration that
would produce a peak approximately 10 times the background noise of a reagent blank near the
elution time of the analyte. These standards, and the reagent blank, were analyzed with the
recommended analytical parameters (1-µL injection with a 19:l split), and the data obtained
were used to determine the required parameters (A and SEE) for the calculation of the DLAP.
Values of 46.59 and 109.5 were obtained for A and SEE respectively. The DLAP was calculated
to be 7.05 pg.
Table 4.2
Detection Limit of the Analytical Procedure
|
| concentration |
mass on column |
area counts |
| (µg/mL) |
(pg) |
(µV-s) |
|
| 0.34 |
17.89 |
1232 |
| 0.68 |
35.79 |
1917 |
| 1.02 |
53.68 |
2796 |
| 1.36 |
71.58 |
3633 |
| 1.70 |
89.47 |
4335 |
| 2.04 |
107.4 |
5206 |
| 2.38 |
125.3 |
6283 |
| 2.72 |
143.2 |
7001 |
| 3.06 |
161.1 |
7946 |
| 3.40 |
178.9 |
8514 |
|
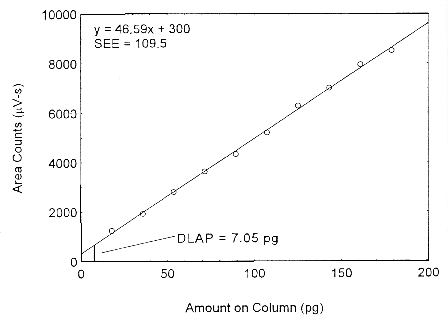
Figure 4.2. Plot of the data from Table 4.2 to determine the DLAP of isopropyl alcohol.
4.3 Detection limit of the overall procedure (DLOP)
The DLOP is measured as mass per sample and expressed as equivalent air concentration, based
on the recommended sampling parameters. Ten samplers were spiked with equal descending
increments of analyte, such that the highest sampler loading was 10.2 µg/sample. This is the
amount spiked on a sampler that would produce a peak approximately 10 times the background
response for a sample blank. These spiked samplers, and a sample blank, were analyzed with the
recommended analytical parameters, and the data obtained used to calculate the required
parameters (A and SEE) for the calculation of the DLOP. Values of 844.1 and 165.1 were
obtained for A and SEE, respectively. The DLOP was calculated to be 0.587 µg/sample (13.3
ppb or 32.6 µg/m3).
Table 4.3
Detection Limit of the Overall Procedure
|
| mass per |
area counts |
| sample |
(µV-s) |
| (µg) |
|
|
| 1.02 |
1228 |
| 2.04 |
2083 |
| 3.06 |
3036 |
| 4.08 |
3783 |
| 5.10 |
4809 |
| 6.12 |
5505 |
| 7.14 |
6268 |
| 8.16 |
7635 |
| 9.18 |
8199 |
| 10.2 |
8795 |
|
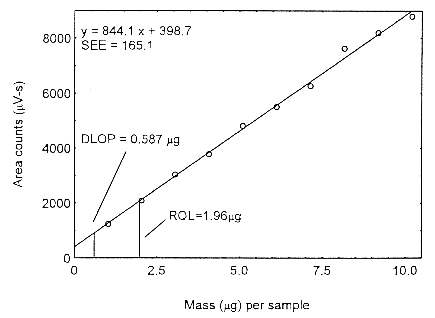
Figure 4.3. Plot of the data to determine the DLOP.
4.4 Reliable quantitation limit (RQL)
The RQL is considered the lower limit for precise quantitative measurements. It is determined
from the regression line parameters obtained for the calculations of the DLOP (Section 4.3),
providing at least 75% of the analyte is recovered. The RQL is defined as the amount of analyte
that gives a response (YRQL) such that
YRQL - YBR = 10(SDBR)
therefore

Figure 4.4. Chromatogram of the RQL for isopropyl alcohol.
The RQL for Isopropyl alcohol was calculated to be 1.96 µg/sample (44.4 ppb or 108.9
µg/m3). The recovery at this concentration is 98.9%.
4.5 Precision (analytical method)
The precision of the analytical procedure is measured as the pooled relative standard deviation
(RSDP). Relative standard deviations are determined from six replicate injections of isopropyl
alcohol standards at 0.5, 0.75, 1, 1.5 and 2 times the target concentrations. After assuring that the
RSDs satisfy the Cochran test for homogeneity at the 95% confidence level, the RSDP is
calculated to be 0.71% and 0.61% for the target concentration with 3 L and 18 L, respectively.
Table 4.5.1
Instrument Response to Isopropyl Alcohol at Target Concentration with
3-L Air Volume
|
| × target concn |
0.5× |
0.75× |
1× |
1.5× |
2× |
| (mg/sample) |
0.50 |
0.74 |
0.99 |
1.48 |
1.98 |
|
| area counts |
121.99 |
178.51 |
245.88 |
364.21 |
483.80 |
| (mV-s) |
121.26 |
181.68 |
245.15 |
364.87 |
487.07 |
|
120.41 |
177.93 |
248.78 |
365.39 |
480.73 |
|
120.65 |
177.72 |
246.55 |
364.89 |
487.10 |
|
121.23 |
178.73 |
246.43 |
363.01 |
485.35 |
|
120.36 |
182.93 |
244.52 |
362.02 |
481.28 |
|
 |
120.98 |
179.58 |
246.22 |
364.06 |
484.22 |
| SD |
0.63 |
2.17 |
1.47 |
1.30 |
2.78 |
| RSD (%) |
0.52 |
1.21 |
0.60 |
0.36 |
0.57 |
|
Table 4.5.2
Instrument Response to Isopropyl Alcohol at Target Concentration with
18-L Air Volume
|
| × target concn |
0.5× |
0.75× |
1× |
1.5× |
2× |
| (mg/sample) |
3.14 |
4.71 |
6.28 |
9.42 |
12.56 |
|
| area counts |
743.30 |
1149.75 |
1488.82 |
2208.53 |
2962.57 |
| (mV-s) |
754.60 |
1126.90 |
1484.71 |
2212.38 |
2981.80 |
|
752.61 |
1132.79 |
1478.24 |
2195.77 |
2975.65 |
|
759.51 |
1134.72 |
1473.01 |
2196.31 |
2982.96 |
|
747.94 |
1133.04 |
1490.48 |
2203.07 |
3001.76 |
|
752.42 |
1135.35 |
1470.78 |
2191.86 |
3014.54 |
|
 |
751.73 |
1135.43 |
1481.0l |
2201.32 |
2986.55 |
| SD |
5.57 |
7.63 |
8.25 |
8.04 |
18.68 |
| RSD (%) |
0.74 |
0.67 |
0.56 |
0.37 |
0.63 |
|
The Cochran test for homogeneity:
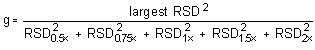
The critical value of the g-statistic, at the 95% confidence level, for five variances, each
associated with six observations is 0.5065. The g-statistics are 0.3915 and 0.3773 for the low and
high target concentrations respectively. Because the g-statistic does not exceed this value, the
RSDs can be considered equal and they can be pooled (RSDP) to
give an estimated RSD for the concentration range studied.
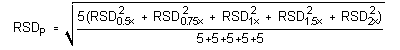
The (RSDP)s are 0.71% and 0.61% for the target
concentration with 3 L and 18 L air volumes respectively.
4.6 Precision (overall procedure)
The precision of the overall procedure is determined from the storage data in Section 4.7. The
determination of the standard error of estimate (SEER) for
a regression line plotted through the graphed storage data allows the inclusion of storage time as
one of the factors affecting overall precision. The SEER is
similar to the standard deviation, except it is a measure of the dispersion of data about
a regression line instead of about a mean. It is determined with the following equation:
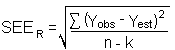
| Yobs |
= |
observed % recovery at a given time |
| Yest |
= |
estimated % recovery from the regression line at the same given time |
| n |
= |
total number of data points |
| k |
= |
2 for linear regression |
| k |
= |
3 for quadratic regression |
An additional 5% for pump error (SP) is added to the SEER
by the addition of variances to obtain the total standard error of the estimate.

The precision at the 95% confidence level is obtained by multiplying the standard error of
estimate (with pump error included) by 1.96 (the z-statistic from the standard normal distribution
at the 95% confidence level). The 95% confidence intervals are drawn about their respective
regression lines in the storage graphs, as shown in Figures 4.7.1.1 through 4.7.1.2. The
precisions of the overall procedure are 10.1% and 10.4% for the ambient and refrigerated
samples, respectively.
4.7 Storage test
4.7.1 Storage test at target concentration with 400-mg tube and 18 L
Storage samples for isopropyl alcohol were prepared by drawing samples from a controlled
test atmosphere of 405 ppm at 0.2 L/min for 90 minutes, to obtain an 18 L air volume. The
relative humidity was approximately 80% at 22°. Thirty-six storage samples were
prepared. Six samples were analyzed immediately after generation, fifteen tubes were
stored at reduced temperature (4°) and the other fifteen were stored in the dark at
ambient temperature (about 22°). At 3-5 day intervals, three samples were selected from
each of the two sets and analyzed.
Table 4.7.1
Storage Test with 400-mg Tubes
|
| time |
ambient storage |
refrigerated storage |
| (days) |
recovery (%) |
recovery (%) |
|
| 0 |
103.6 |
104.6 |
105.0 |
103.6 |
104.6 |
105.0 |
| 0 |
103.1 |
104.3 |
103.2 |
103.1 |
104.3 |
103.2 |
| 3 |
105.8 |
105.7 |
104.9 |
104.0 |
104.3 |
103.8 |
| 7 |
103.0 |
102.3 |
102.8 |
107.8 |
108.5 |
106.6 |
| 10 |
104.9 |
105.2 |
104.6 |
103.9 |
104.2 |
102.0 |
| 14 |
102.4 |
102.4 |
102.3 |
102.5 |
102.7 |
103.5 |
| 19 |
102.1 |
100.3 |
100.2 |
100.9 |
100.3 |
101.8 |
|
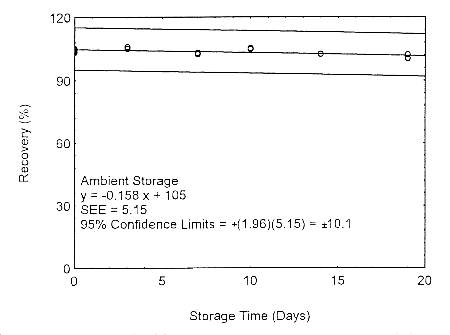
Figure 4.7.1.1. Ambient storage test using 400-mg tubes at the target concentration.
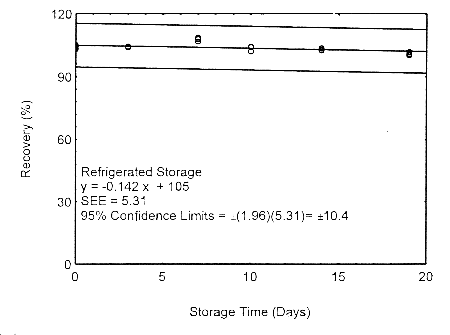
Figure 4.7.1.2. Refrigerated storage test using 400-mg tubes at the target concentration.
4.7.2 Storage and migration test with 400/200 mg Anasorb® 747 tubes
Migration samples for isopropyl alcohol were prepared by drawing samples from a
controlled test atmosphere of 405 ppm at 0.2 L/min for 90 minutes onto 400/200 mg
Anasorb® 747 tubes (Lot 263). Samples were generated at both a relative humidity of
approximately 80% at 22° and 11% at 22°. Forty-eight storage samples were prepared
at each relative humidity. For each relative humidity, 11% or 80%, six samples were
analyzed immediately after generation, eighteen tubes were stored at reduced temperature
(4°) and the other fifteen were stored in the dark at ambient temperature (about 22°).
At 3-5 day intervals, three samples were selected from each of the four sets and analyzed.
| Table 4.7.2 |
| Storage and Migration Test on 400/200 mg Anasorb® 747 Tubes |
|
|
11% RH |
80% RH |
| time |
ambient storage |
refrigerated storage |
ambient storage |
refrigerated storage |
| (days) |
recovery (%) |
recovery (%) |
recovery (%) |
recovery (%) |
|
front |
back |
front |
back |
front |
back |
front |
back |
|
section |
section |
section |
section |
section |
section |
section |
section |
|
| 0 |
104.5 |
0.0 |
104.5 |
0.0 |
104.5 |
0.0 |
104.5 |
0.0 |
| 0 |
103.6 |
0.0 |
103.6 |
0.0 |
103.6 |
0.0 |
103.6 |
0.0 |
| 0 |
103.8 |
0.0 |
103.8 |
0.0 |
103.8 |
0.0 |
103.8 |
0.0 |
| 0 |
104.6 |
0.0 |
104.6 |
0.0 |
104.6 |
0.0 |
104.6 |
0.0 |
| 0 |
105.6 |
0.0 |
105.6 |
0.0 |
105.6 |
0.0 |
105.6 |
0.0 |
| 0 |
104.1 |
0.0 |
104.1 |
0.0 |
104.1 |
0.0 |
104.1 |
0.0 |
| 5 |
102.1 |
1.1 |
103.0 |
0.0 |
102.9 |
0.0 |
103.5 |
0.0 |
| 5 |
103.1 |
0.0 |
102.8 |
0.0 |
102.6 |
0.0 |
102.8 |
0.0 |
| 5 |
102.1 |
1.2 |
103.6 |
0.0 |
100.3 |
0.0 |
103.9 |
0.0 |
| 8 |
98.1 |
2.4 |
105.0 |
0.0 |
103.7 |
0.0 |
103.4 |
0.0 |
| 8 |
100.0 |
3.7 |
103.6 |
0.0 |
104.0 |
0.0 |
102.1 |
0.0 |
| 8 |
101.4 |
1.0 |
102.3 |
0.0 |
104.0 |
0.0 |
102.5 |
0.0 |
| 12 |
101.5 |
2.5 |
103.9 |
0.5 |
103.9 |
0.1 |
105.3 |
0.0 |
| 12 |
100.6 |
4.6 |
103.3 |
1.0 |
102.2 |
0.0 |
103.8 |
0.0 |
| 12 |
98.6 |
3.7 |
104.2 |
1.2 |
104.2 |
0.0 |
103.3 |
0.0 |
| 15 |
100.4 |
4.5 |
103.7 |
0.2 |
103.4 |
0.0 |
103.1 |
0.0 |
| 15 |
101.2 |
3.1 |
104.8 |
0.8 |
104.1 |
0.0 |
102.8 |
0.0 |
| 15 |
101.3 |
3.1 |
102.3 |
0.0 |
102.2 |
0.0 |
103.1 |
0.0 |
| 20 |
98.7 |
7.4 |
104.5 |
2.2 |
103.6 |
0.0 |
103.9 |
0.0 |
| 20 |
96.0 |
9.0 |
104.3 |
0.1 |
101.9 |
0.3 |
100.8 |
0.0 |
| 20 |
95.6 |
9.3 |
102.8 |
3.0 |
103.6 |
0.0 |
101.6 |
0.0 |
| 23 |
94.7 |
8.1 |
104.2 |
2.2 |
103.8 |
0.3 |
102.2 |
0.0 |
| 23 |
95.9 |
10.0 |
101.4 |
4.3 |
101.5 |
0.2 |
103.6 |
0.0 |
| 23 |
94.4 |
8.1 |
105.3 |
0.4 |
101.8 |
0.1 |
103.4 |
0.0 |
|
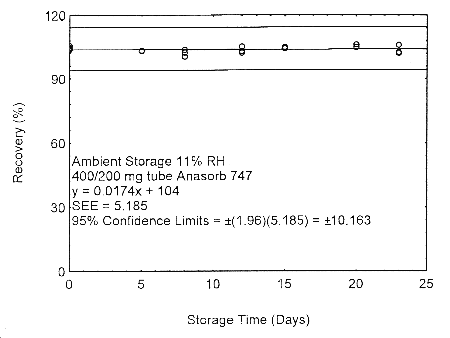
Figure 4.7.2.1. Ambient storage test of 400/200 mg Anasorb® 747 tube generated with 11% RH air.
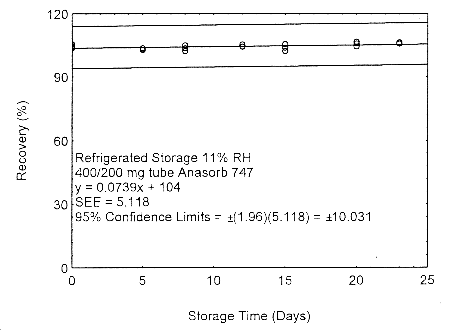
Figure 4.7.2.2. Refrigerated storage test of 400/200 mg Anasorb® 747 tube generated with 11% RH air.
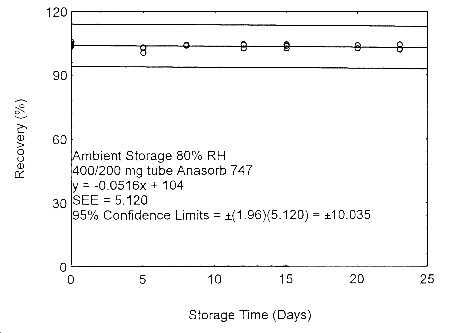
Figure 4.7.2.3. Ambient storage test of 400/200 mg Anasorb® 747 tube generated with 80% RH air.
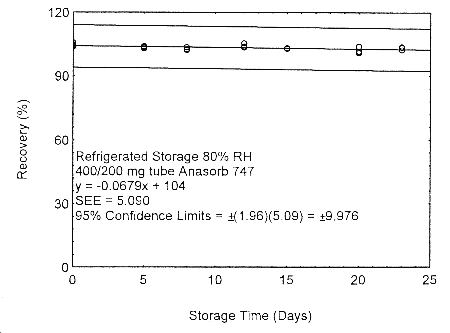
Figure 4.7.2.4. Refrigerated storage test of 400/200 mg Anasorb® 747 tube generated with 80%
RH air..
These migration studies indicate that isopropyl alcohol migrates upon storage, especially at
ambient temperature, but overall storage is stable at ambient and refrigerated temperatures. To
avoid migration after sampling, isopropyl alcohol samples should be collected on two Anasorb®
747 tubes in series, a 400 mg and a 200 mg. The tubes are separated, capped, and sealed
immediately after sampling.
4.8 Reproducibility
Six samples were collected from a test atmosphere using two Anasorb® 747 tubes, 400 mg and
200 mg, in series. The isopropyl alcohol concentration of the test atmosphere was 405 ppm. They
were sampled at 0.2 L/min for 90 minutes. The relative humidity was 80% at 22°. The
samples were submitted to an OSHA Salt Lake Technical Center service branch, along with a
draft copy of this method. The samples were analyzed after being stored for 3 days at 4°.
Sample results were corrected for desorption efficiency. No sample result for isopropyl alcohol
had a deviation greater than the precision of the overall procedure determined in Section 4.6.
Table 4.8
Reproducibility Data
|
| sample |
expected |
reported |
recovery |
deviation |
|
(mg) |
(mg) |
(%) |
(%) |
|
| 1 |
19.47 |
19.69 |
101.1 |
1.1 |
| 2 |
19.83 |
20.36 |
102.7 |
2.7 |
| 3 |
19.84 |
20.15 |
101.6 |
1.6 |
| 4 |
19.81 |
20.18 |
101.9 |
1.9 |
| 5 |
19.47 |
20.22 |
103.9 |
3.9 |
| 6 |
19.81 |
19.85 |
100.2 |
0.2 |
|
4.9 Sampler capacity
The sampling capacity of the 400-mg Anasorb® 747 sampling tube, Lot 645, was tested by
sampling from a dynamically generated test atmosphere of isopropyl alcohol of 808 ppm. The
samples were collected at 0.05 L/min and at 0.2 L/min. The relative humidities were
approximately 12% and 80% at 22°. A GC with a gas sampling valve was placed in-line
behind the 400-mg front test section. The valve was rotated to measure the amount of isopropyl
alcohol passing through the sampler at the time of rotation. The 5% breakthrough air volume at
84% RH and 22° was determined to be 22.6 L.
| Table 4.9 |
| Capacity Tests for Isopropyl Alcohol on 400-mg Anasorb® 747 Tubes |
|
| 13% RH at 0.05 L/min |
12% RH at 0.2 L/min |
84% RH at 0.2 L/min |
77% RH at 0.05 L/min |
| air volume |
(BT) |
air volume |
(BT) |
air volume |
(BT) |
air volume |
(BT) |
| (L) |
(%) |
(L) |
(%) |
(L) |
(%) |
(L) |
(%) |
|
| 32.11 |
0.0 |
29.01 |
0.0 |
19.78 |
0.0 |
27.39 |
0.0 |
| 34.32 |
0.44 |
29.37 |
2.15 |
22.08 |
2.85 |
28.83 |
4.23 |
| 36.57 |
1.23 |
29.76 |
2.98 |
22.13 |
0.58 |
28.94 |
4.70 |
| 37.18 |
5.24 |
29.78 |
4.13 |
24.61 |
19.51 |
29.53 |
11.31 |
| 38.80 |
5.44 |
30.20 |
6.21 |
24.74 |
6.63 |
30.07 |
10.40 |
| 39.38 |
14.83 |
30.55 |
8.03 |
26.47 |
19.84 |
30.29 |
21.82 |
| 41.26 |
18.29 |
30.65 |
13.47 |
|
|
31.01 |
10.60 |
|
|
31.14 |
22.08 |
|
| BT = breakthrough |
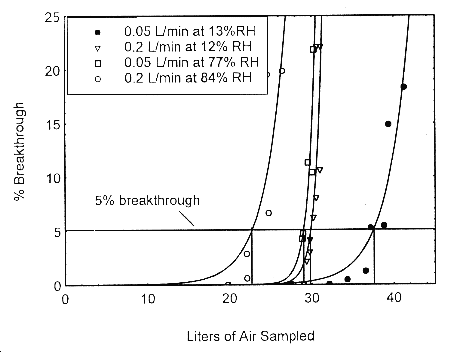
Figure 4.9. Five percent breakthrough air volume for
isopropyl alcohol on Anasorb® 747. The breakthrough
air volumes are: 22.6 L for 0.2 L/min at 84% RH, 29.0 L
for 0.05 L/min at 77% RH, 29.8 L for 0.2 L/min at 12%
RH, and 37.6 L for 0.05 L/min at 13% RH.
4.10 Desorption efficiency and stability of desorbed samples
4.10.1 The desorption efficiency from dry Anasorb® 747
The desorption efficiencies (DE) of isopropyl alcohol were determined by liquid-spiking
400-mg portions of Anasorb® 747 with amounts equivalent to 0.05 to 2 times the target
concentration. These samples were stored overnight at ambient temperature and then
desorbed with 3-mL of 60/40 DMF/CS2 and analyzed by GC-FID. The average desorption
efficiency over the working range of 0.5 to 2 times the target concentration is 103.6%.
| Table 4.10.1.1 |
| Desorption Efficiency of Isopropyl Alcohol |
|
| × target concn |
0.05× |
0.1× |
0.2× |
0.5× |
1.0× |
2.0× |
| (mg/sample) |
0.942 |
1.884 |
3.768 |
9.421 |
18.84 |
37.68 |
|
| DE (%) |
99.2 |
102.4 |
102.9 |
102.5 |
103.6 |
103.8 |
|
| 101.0 |
102.2 |
104.9 |
104.4 |
103.2 |
104.1 |
|
| 99.9 |
104.2 |
102.7 |
102.4 |
104.3 |
102.6 |
|
| 99.2 |
102.0 |
103.3 |
102.8 |
103.8 |
104.2 |
|
| 100.6 |
102.8 |
104.9 |
104.8 |
103.6 |
103.3 |
|
| 101.0 |
103.6 |
102.8 |
102.1 |
103.7 |
104.6 |
|
 |
100.2 |
102.9 |
103.6 |
103.2 |
103.7 |
103.8 |
|
The stability of desorbed samples was investigated by reanalyzing the target concentration
samples 64 h after initial analysis. After the original analysis was performed, three vials
were recapped with new septa while the remaining three retained their punctured septa.
The samples were reanalyzed with fresh standards. The average percent change was
-0.2% for samples that were resealed with new septa and -0.3% for those that retained
their punctured septa.
| Table 4.10.1.2 |
| Stability of Desorbed Samples from Anasorb® 747 |
|
| punctured septa replaced |
punctured septa retained |
| initial |
DE after |
|
initial |
DE after |
|
| DE |
64 hours |
difference |
DE |
one day |
difference |
| (%) |
(%) |
|
(%) |
(%) |
|
|
| 103.6 |
103.6 |
0.0 |
103.8 |
103.5 |
-0.3 |
| 103.2 |
104.1 |
+0.9 |
103.6 |
102.6 |
-1.0 |
| 104.3 |
102.8 |
-1.5 |
103.7 |
104.2 |
+0.5 |
|
averages |
|
|
averages |
|
| 103.7 |
103.5 |
-0.2 |
103.7 |
103.4 |
-0.3 |
|
4.10.2 Desorption efficiency with wet Anasorb® 747
The possible interference of collected water vapor was studied by determining the
desorption efficiencies (DE) of isopropyl alcohol after drawing humid air through previously
spiked tubes. The desorption efficiencies were determined by liquid-spiking 400-mg
portions of Anasorb® 747 with amounts equivalent to 0.05 to 2 times the target
concentration. These samples were stored for 4 hours at ambient temperature and then
had 20 L of humid air (80% RH at 21°) drawn through them at 0.2 L/min. These samples
were immediately desorbed with 3-mL of 60/40 DMF/CS2
and analyzed by GC-FID. The
average desorption efficiency over the working range of 0.5 to 2 times the target
concentration is 105.1%. The presence of water appears to slightly increase the desorption
efficiency of isopropyl alcohol.
| Table 4.10.2 |
| Desorption Efficiency of Isopropyl Alcohol with Wet Anasorb® 747 |
|
| × target concn |
0.05× |
0.1× |
0.2× |
0.5× |
1.0× |
2.0× |
| (mg/sample) |
0.942 |
1.88 |
3.77 |
9.42 |
18.8 |
37.7 |
|
| DE (%) |
101.4 |
102.3 |
103.0 |
104.2 |
104.7 |
107.2 |
|
102.5 |
103.2 |
102.5 |
104.0 |
103.3 |
106.1 |
|
103.2 |
104.5 |
103.8 |
104.7 |
104.3 |
107.3 |
|
103.2 |
104.1 |
104.4 |
106.1 |
104.4 |
106.1 |
|
103.4 |
102.9 |
102.8 |
104.4 |
105.2 |
106.1 |
|
101.1 |
102.5 |
103.9 |
105.4 |
105.3 |
103.8 |
|
 |
102.5 |
103.3 |
103.4 |
104.8 |
104.5 |
106.1 |
|
4.10.3 Desorption efficiency with 99/1 CS2/DMF
The desorption efficiencies (DE) of isopropyl alcohol were determined by liquid-spiking
400-mg portions of Anasorb® 747 with amounts equivalent to 0.05 to 2 times the target
concentration. These samples were stored overnight at ambient temperature. The
samples were desorbed with 3 mL of 99/1 CS2/DMF,
with 0.25 µL/mL p-cymene internal
standard, on a shaker for 1 hour, and analyzed by GC-FID. The average desorption
efficiency over the working range of 0.5 to 2 times the target concentration is 97.5%. The
samples in Table 4.10.3.2 were prepared by liquid spiking six tubes at each level, and then
drawing 18 L of 80% RH air at 22° through them at 0.2 L/min. They were immediately
desorbed with 3-mL of the 99/1 CS2/DMF,
with 0.25 µL/mL p-cymene internal standard, for
1 hour with occasional vigorous shaking, and analyzed by GC-FID. A layer of water was
observed floating on top of the 99/1 CS2/DMF, and upon analysis of this water layer a
significant amount of isopropyl alcohol was found. This indicates that 99/1 CS2/DMF
cannot be used to desorb isopropyl alcohol samples from Anasorb® 747 tubes.
| Table 4.10.3.1 |
| Desorption Efficiency of Dry Anasorb® 747
Desorbed with 99/1 CS2/DMF |
|
| × target concn |
0.05× |
0.1× |
0.2× |
0.5× |
1.0× |
2.0× |
| (mg/sample) |
0.942 |
1.88 |
3.77 |
9.42 |
18.8 |
37.7 |
|
| DE(%) |
92.1 |
93.9 |
94.0 |
95.2 |
98.6 |
98.6 |
|
92.0 |
92.0 |
93.9 |
97.2 |
99.2 |
99.5 |
|
91.5 |
93.7 |
94.9 |
96.6 |
97.3 |
98.6 |
|
92.0 |
94.3 |
94.3 |
95.7 |
97.1 |
100.0 |
|
92.9 |
95.4 |
96.3 |
94.7 |
96.3 |
99.0 |
|
93.4 |
94.2 |
95.6 |
95.0 |
96.5 |
99.8 |
|
 |
92.3 |
93.9 |
94.8 |
95.7 |
97.5 |
99.3 |
|
| Table 4.10.3.2 |
| Desorption Efficiency of Wet Anasorb® 747 (18 L 80% RH at
22° drawn through them) Desorbed with 99/1 CS2/DMF |
|
| × target concn |
0.5× |
1.0× |
2.0× |
| (mg/sample) |
9.42 |
18.84 |
37.68 |
|
| DE (%) |
85.4 |
87.6 |
90.2 |
|
86.4 |
88.5 |
89.7 |
|
87.5 |
89.7 |
88.1 |
|
85.7 |
90.4 |
87.3 |
|
86.1 |
88.1 |
90.4 |
|
84.9 |
90.3 |
89.9 |
|
 |
86.0 |
89.1 |
89.3 |
|
The apparent desorption efficiency of isopropyl alcohol with
99/1 CS2/DMF decreases in the
presence of water vapor, and should not be used to desorb field samples, as there will be
water vapor present when taking field samples.
4.11 Qualitative analysis
The mass spectrum for isopropyl alcohol was obtained from an HP5988A Mass Spec
interfaced to a Hewlett-Packard 5890 Series II GC.
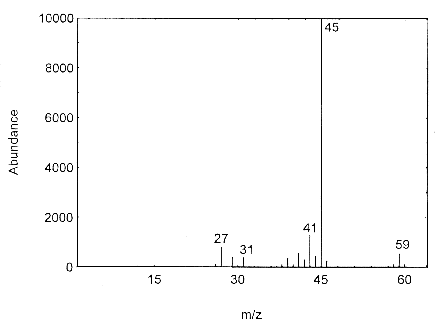
Figure 4.11. Mass spectra of isopropyl alcohol.
4.12 Capacity studies for ethyl and methyl alcohol at 0.2 L/min
The sampling capacity of Anasorb® 747 tubes at 0.2 L/min for methyl alcohol and ethyl alcohol
was tested to determine if the higher flow rate could be used to sample for these analytes in
conjunction with isopropyl alcohol. The sampling capacity of the 400-mg Anasorb® 747 sampling
tube, Lot 645, was tested by sampling from a dynamically generated test atmosphere of either
ethyl alcohol at 1987 ppm, or methyl alcohol at 402 ppm. The samples were collected at 0.2
L/min. The relative humidity in the ethyl alcohol tests was 13% and 80% at 23°. The relative
humidity in the methyl alcohol tests was 11% and 80% at 22°. A GC with a gas sampling
valve was placed in-line behind the 400-mg tube. The valve was rotated to measure the amount
of compound, either ethyl alcohol or methyl alcohol, passing through the sampler at the time of
rotation. The 5% breakthrough air volume for ethyl alcohol was 14.1 L at 13% RH and 15.7 L
for 80% RH. The 5% breakthrough air volume for methyl alcohol was 2.38 L at 11% RH and
2.84 L at 80% RH. These breakthrough determinations indicate that methyl alcohol should not be
sampled at 0.2 L/min.
| Table 4.12.1 |
| Capacity Tests for Ethyl Alcohol on 400-mg Anasorb® 747 Tubes at 0.2 L/min |
|
| 13% RH at 0.2 L/min |
80% RH at 0.2 L/min |
| air volume (L) |
breakthrough (%) |
air volume (L) |
breakthrounh (%) |
|
| 10.60 |
0.00 |
10.16 |
0.00 |
| 12.06 |
0.49 |
10.76 |
0.00 |
| 13.51 |
0.81 |
11.88 |
0.12 |
| 14.22 |
0.00 |
12.19 |
0.38 |
| 14.97 |
2.38 |
13.30 |
0.45 |
| 15.64 |
1.37 |
13.61 |
1.97 |
| 16.42 |
8.33 |
14.68 |
3.89 |
| 17.16 |
7.18 |
15.03 |
10.42 |
| 17.88 |
18.64 |
16.15 |
22.34 |
| 19.33 |
34.19 |
16.45 |
40.30 |
| 20.01 |
40.46 |
17.57 |
77.04 |
|
| Table 4.12.2 |
| Capacity Tests for Methyl Alcohol on 400-mg Anasorb® 747 Tubes at 0.2 L/min |
|
| 11% RH at 0.2 L/min |
80% RH at 0.2 L/min |
| air volume (L) |
breakthrough (%) |
air volume (L) |
breakthrough (%) |
|
| 1.66 |
0 |
1.61 |
0 |
| 2.34 |
0 |
2.34 |
0 |
| 3.12 |
14.56 |
2.98 |
3.994 |
| 3.72 |
13.87 |
3.96 |
12.22 |
| 4.68 |
49.40 |
4.47 |
16.50 |
| 5.18 |
44.77 |
5.59 |
23.00 |
| 6.24 |
76.64 |
5.93 |
30.36 |
| 6.56 |
69.97 |
7.01 |
39.35 |
|
|
7.38 |
39.31 |
|
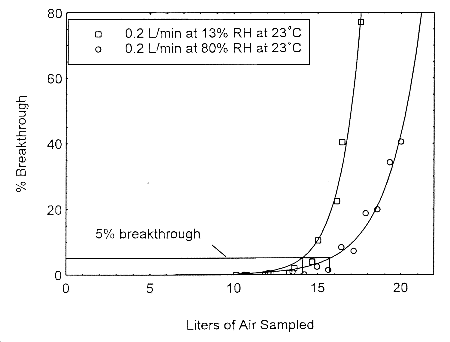
Figure 4.12.1. Five percent breakthrough air volume for
ethyl alcohol on Anasorb® 747. The breakthrough air
volumes at 0.2 L/min were 14.1 L and 15.7 L for 13% and
80% RH respectively.
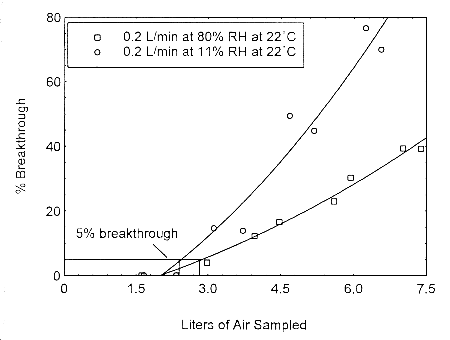
Figure 4.12.2. Five percent breakthrough air volume for
methyl alcohol on Anasorb® 747. The breakthrough air
volumes at 0.2 L/min were 2.38 L and 2.84 L for 11% and
80% RH respectively.
5. References
5.1 NIOSH Manual of Analytical Methods, Eller, P.M., Ed, 4th ed., U.S. Department of Health
and Human Services, Public Health Service, Centers for Disease Control, National Institute for
Occupational Safety and Health, Division of Physical Sciences and Engineering, Cincinnati, OH,
DHHS (NIOSH) Publication No. 94-l 13, 1994; Method 1400.
5.2 Eide, M., A Study of the Desorption, Retention, and Storage Efficiencies of Isopropyl
Alcohol from Charcoal tubes Lot 120, OSHA Salt Lake Technical Center, unpublished, Salt
Lake City, UT 84165, October 1996.
5.3 OSHA Analytical Methods Manual, 2nd ed., U.S. Department of Labor, Occupational Safety
and Health Administration, Directorate for Technical Support, OSHA Salt Lake Technical
Center, Salt Lake City, UT 84165, October 1993, Part 1, Volume 4, Method 91, ACGIH
publication #4542.
5.4 OSHA Analytical Methods Manual, 2nd ed., U.S. Department of Labor, Occupational Safety
and Health Administration, Directorate for Technical Support, OSHA Salt Lake Technical
Center, Salt Lake City, UT 84165, October 1993, Part 1, Volume 4, Method 100, ACGIH
publication #4542.
5.5 Documentation of the Threshold Limit Values and Biological Exposure Indices, 6th ed.;
American Conference of Governmental Industrial Hygienists, Inc.: Cincinnati, OH, 1991, Vol. II,
p. 828.
5.6 Chem. Eng. News, 1995, 73(26), p. 40.
5.7 Merck Index, Budavari, S., Ed., 12th ed., Merck & Co., Whitehouse Station NJ, 1996, p.
889.
5.8 Hawley's Condensed Chemical Dictionary, Lewis, R., Ed., 12th ed., Van Nostrand Reinhold
Co., New York, 1993, p. 660.
|

