 |
|
|
January 2006
|
 |
|
|
|
|

|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
 |
|
FREQUENTLY
ASKED QUESTIONS FROM GRANTSINFO
Q: If
an application receipt date falls on a weekend or a
holiday, when is the application due?
A: When a standard postmark/submission date
falls on a weekend or Federal holiday, the
submission deadline will be extended to the
next business day.
Q:
How can I get a copy of the NIH Guide for Grants and
Contracts?
A: You may subscribe to the NIH Guide
LISTSERV by sending an email to: listserv@list.nih.gov
with the following text in the message body (not the
Subject line): subscribe NIHTOC-L your name
(Example: subscribe NIHTOC-L Joe Smith). Your e-mail
address will be automatically obtained from the
e-mail message and add you to the LISTSERV.
Q: How can I find
grants awards by state?
A: You will find awards by state and foreign
site under the heading, "Award Trends" at
the Award
Data Web site.
Q: What do extramural
and intramural mean?
A: Extramural refers to research conducted at
universities and institutions outside of NIH;
intramural refers to research conducted at the NIH.
Q: What is the
definition of key personnel?
A: Individuals who contribute in a
substantive way to the scientific development or
execution of a project, whether or not they receive
compensation from the grant supporting that project.
Q: Where can I find
information about the current salary cap?
A: Visit the Salary
Cap Summary Web site.
GrantsInfo
is a communication service of the NIH Office of
Extramural Research. It provides general information
to the biomedical research community about NIH
extramural research and research training programs,
grant application procedures and process, how to
obtain the NIH
Guide and resources available on the NIH
Web site. In addition, GrantsInfo
is the contact point for organizations to request
application kits and forms.
 Back
to top Back
to top
|
|
|
|
 |
|
COMMUNICATE
WITH THE
NIH EXTRAMURAL NEXUS — WE WANT TO HEAR FROM
YOU
Feedback
(to the Editor) from recipients and
subscribers of the NIH Extramural Nexus is
vital. Your comments, questions, and suggestions for
topics will enable Nexus editorial staff to
deliver appropriate content to the extramural
community.
 Back
to top Back
to top
|
|
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
|
Printer
Friendly Version
(Adobe
Acrobat Reader Required) |
|
| INTRODUCING
THE NIH EXTRAMURAL NEXUS |
| The
National Institutes of Health Office of Extramural
Research (OER) welcomes you to your first update
on NIH extramural issues via the NIH Extramural
Nexus. The OER is the
hub
for grants policy and operations, grants
administration, and the coordination of NIH's
extramural programs and activities. Through the Nexus,
the OER will provide the external scientific
community with updates on NIH policies and
activities as well as an opportunity to gain a
better understanding of the operation of
extramural programs. We intend the Nexus to
be a two-way communications portal — we will be
listening to you and plan to modify the content of
the Nexus accordingly.
The Nexus appears at a time
of considerable change at the NIH. Among other
things, the President has signed the FY 2006
budget and we have developed financial operations
policies; we are moving quickly to electronic
submission of applications; we are considering new
policies to facilitate the careers of new
investigators; and, we are redesigning our grant
programs to recognize the contribution of multiple
members of research teams.
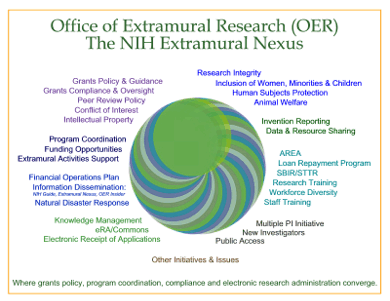
Click on
graphic to expand (opens in new window)
Each bimonthly issue of the NIH Extramural
Nexus will acquaint readers with topical
information about extramural policies and
initiatives as well as the various programs that
comprise the OER. In the articles below,
identified by the Nexus
Symbol you will be introduced to NIH
FY 2006 financial operations policies, the
status
of Electronic Submission, the Division
of Extramural Inventions & Technology
Resources, the Multiple
Principal Investigator and New
Investigator initiatives, and NIH's
Public Access Policy — all important
extramural issues and components integral to the
NIH mission. In future months, in conjunction with
providing information about policy, the Nexus
will highlight many other aspects of the NIH's
extramural program.
— Norka Ruiz Bravo, PhD - Director,
OER and NIH Deputy Director for Extramural
Research
 Back
to top Back
to top
|
| NIH
FISCAL OPERATIONS PLAN FOR FY 2006 |
|
As many of you know, the President signed the
Department of Health and Human Services
appropriations bill on December 30, 2005. The NIH
appropriation included a 1 percent
across-the-board cut for all non-emergency
discretionary programs; the NIH share is $286
million. This has forced NIH to make some very
tough choices. Paramount in making these choices
is two principles: we must continue to support the
very best biomedical research in the world; and we
must ensure that the next generation of scientists
continues to have the opportunity to compete
successfully for grants and transition to
independent investigator status.
Guided by these principles, NIH will reduce all
non-competing research project grant (RPG) awards
by 2.35 percent (see
definition of RPGs); restore funds to all
previously issued FY 2006 awards to 97.65 percent
of the amount indicated in the FY 2005 Notice of
Grant Award; and maintain the average cost of
competing RPGs at FY 2005 levels. By undertaking
these difficult measures, we expect to achieve an
overall success rate of approximately 19.5 percent
compared to 22.3 percent in FY 2005. We also
expect to fund more than 38,300 competing and
non-competing RPGs. When compared to FY 2005, this
represents a decrease of about 570 RPGs or 1.5
percent (see the NIH
Fiscal Operations Plan).
As difficult as these decreases will be to
absorb, by tightening our belts in this fashion,
we will be able to continue to fund the very best
biomedical research in the world. We remain
concerned, however, about the impact of falling
success rates on the next generation of
investigators. Therefore, we expect to announce
programs targeting new investigators soon — look
for these in the NIH
Guide to Grants and Contracts.
We fully recognize that the difficult budget
situation will make the competition for funds even
tougher than in the past, and we stand ready to
assist you in any way that we can. I encourage you
to contact your program director or grants
management specialist if you have questions
specific to your grant. In addition, please feel
free to contact
me with questions or comments about
NIH's fiscal operation plan or other issues that
you may wish to discuss.
— Norka Ruiz Bravo, PhD - Director,
OER and NIH Deputy Director for Extramural
Research
 Back
to top Back
to top
|
 NIH
SUCCESSFULLY RECEIVES GRANT APPLICATIONS
ELECTRONICALLY THROUGH GRANTS.GOV NIH
SUCCESSFULLY RECEIVES GRANT APPLICATIONS
ELECTRONICALLY THROUGH GRANTS.GOV |
| The
first round of electronic grant submissions
through the Grants.gov
Federal Web portal involved applications for the
Small Business Innovative Technology (SBIR) and
Small Business Technology Transfer (STTR) grant
program and was followed by applications for the
R13 Grant Program (Support for Conference and
Scientific Meetings) for the December 15, 2005,
submission deadline.
 The
change to electronic applications is very complex.
It involves not only a change from paper to
electronic submission, it also involves a new
application form (from the PHS 398 to the new
SF424 Research and Related (R&R) form). As
expected with any change of this magnitude, we
have encountered a number of hurdles. Many
applicants for the initial submission dates were
not fully aware of the registration requirements
or the business validations associated with the
new electronic process. The use of automated
validations caught many application errors that
might not have been noticed in the paper process.
Ultimately, these validations will lead to
improved accuracy and fairness in the process, but
requires careful adherence to instructions. Many
applicants had to submit multiple corrected
applications in order to pass NIH validations.
Despite a doubling of the size of the NIH Help
Desk, applicants found it difficult to reach
support staff during peak times. Educating
applicants about registration requirements and
explaining the NIH validation process were among
the main issues that NIH encountered. Clearly,
educating applicants and addressing the related
issue of Help Desk support will be high priorities
as we move ahead with electronic submissions. The
change to electronic applications is very complex.
It involves not only a change from paper to
electronic submission, it also involves a new
application form (from the PHS 398 to the new
SF424 Research and Related (R&R) form). As
expected with any change of this magnitude, we
have encountered a number of hurdles. Many
applicants for the initial submission dates were
not fully aware of the registration requirements
or the business validations associated with the
new electronic process. The use of automated
validations caught many application errors that
might not have been noticed in the paper process.
Ultimately, these validations will lead to
improved accuracy and fairness in the process, but
requires careful adherence to instructions. Many
applicants had to submit multiple corrected
applications in order to pass NIH validations.
Despite a doubling of the size of the NIH Help
Desk, applicants found it difficult to reach
support staff during peak times. Educating
applicants about registration requirements and
explaining the NIH validation process were among
the main issues that NIH encountered. Clearly,
educating applicants and addressing the related
issue of Help Desk support will be high priorities
as we move ahead with electronic submissions.
"We are very pleased with the results of
our first electronic submissions through
Grants.gov and feel that we have a solid
foundation from which to build upon for future
submissions," Dr. Norka Ruiz Bravo, NIH
deputy director for Extramural Research, said.
"We've learned a great deal from our initial
submissions and we are already developing an
action plan to address identified issues."
Working hand-in-hand with Grants.gov through
this initial round, NIH has cemented a strong
working relationship with its Federal partner.
Grants.gov and the NIH worked together to lay the
groundwork for this initiative in advance,
ensuring sufficient capacity to handle the
additional load of NIH grant applications, making
sure that the NIH retrieval of applications from
Grants.gov went smoothly.
NIH is now rolling up its sleeves to prepare
for the October 1, 2006 transition of its largest
grant program, the R01 or Research Project Grant
Program, which has been known to receive 10,000
applications for a single receipt cycle.
NIH will continue to transition grant programs
to electronic submission using the new form set
through the end of May 2007. For additional
details, see NIH's
electronic submission timeline.
Having received a great deal of feedback from
applicants that took part in the initial
submissions, NIH offers the following advice to
applicants:
 |
Be informed. For
up-to-date, general information on
electronic submission, the SF424
(R&R), and Service Providers, visit
the NIH Electronic
Submission of Grant Applications Web site.
|
 |
Be prepared, register
early. One-time registrations for both
Grants.gov
and eRA
Commons systems must be completed
before application submission. These are
two distinct systems with separate
registration requirements.
Registration Tips:
 |
The registration process can
take several weeks, especially
when close to submission dates
when registration volume peaks.
Start early to avoid delays! NIH
recommends starting the
registration process now, or at a
minimum two-four weeks ahead of
your target submission date. New
organizations may need to start
the Grants.gov registration
process even earlier to obtain the
necessary DUNS numbers and CCR
registrations required for
Grants.gov registration.
|
 |
Applicant organizations must
register both in Grants.gov and
eRA Commons, but Principal
Investigators (PIs) need only
register in eRA Commons.
|
 |
The PI must hold a PI account
and be affiliated with the
applicant organization in the eRA
Commons.
|
 |
Both the Principal Investigator
(PI) and Signing Official (SO)
need separate accounts in eRA
Commons since both need to verify
the application. If you are the SO
for your organization as well as a
PI of the grant, you will need two
separate accounts with different
user names - one with SO authority
and one with PI authority.
|
 |
Organizations must include the
DUNS number in their eRA Commons
profile that matches the DUNS
number to be used on the submitted
applications.
|
 |
Grants.gov and eRA Commons
registration processes may be done
concurrently. |
|
| |
|
 |
Follow all instructions
carefully. Information found in the
funding opportunity announcement and the
application guide that accompanies each
application package is critical to
submitting a complete and accurate
application to NIH. Many applicants spent
a great deal of time correcting errors and
resubmitting applications. Much of this
effort can be avoided by reading and
following the instructions up-front. Sample
versions of the application guide and
packages are available, as are the most
common application errors. |
 Back
to top Back
to top
|
| ECONOMIC
IMPACT OF FEDERAL HEALTH CARE REGULATIONS:
REQUEST FOR PUBLIC COMMENT AND SCHEDULE OF
PUBLIC MEETINGS IN CALENDAR YEAR 2006 |
|
The NIH is helping the Department of
Health and Human Services (HHS)
disseminate information about an
opportunity to provide input on reducing
regulatory burden. HHS requests public
comments estimating the economic impact of
health care regulations, guidance
documents, or paperwork requirements, and
on methods they use to calculate the
economic impact of the regulations. This
activity is being driven by House Report
language requesting government review of
federal regulations governing the health
care industry in order to reduce costs and
to improve translation of biomedical
research into medical practice.
The government is holding several
public meetings to allow the public to
provide their input on the economic impact
of federal regulations. Those who are
unable to attend the public meetings may
submit written comments, including a
description of their proposed methods for
quantifying the economic impact of health
care regulations. For additional
background, more information about
submitting public comments, and a schedule
of Town Hall meetings, visit the HHS Action
to Reduce Regulatory Burden Web site.
Comments will be accepted through
February 9, 2006.
 Back
to top Back
to top
|
|
|
|
 MULTIPLE
PRINCIPAL INVESTIGATOR INITIATIVE LAUNCHED MULTIPLE
PRINCIPAL INVESTIGATOR INITIATIVE LAUNCHED
|
| As
scientific problems have become more complex, so
have the ways that we must address them. Many
scientists are moving from reductionist to systems
approaches, and biomedical science increasingly
involves teams of investigators, who have been
trained in different disciplines. NIH recognized
the importance of interdisciplinary approaches and
team science by launching a comprehensive Roadmap
initiative designed specifically to encourage
this kind of research.
There are, however, systemic barriers to team
research. These barriers include the fact that NIH
recognizes only a single Principal Investigator
(PI) on research grants. That means that
frequently, major contributors to NIH-funded
research projects do not receive appropriate
credit for their intellectual contributions, and
this serves as a deterrent to team science. To
reduce this barrier and encourage and facilitate a
broader use of team approaches, the NIH plans to
start recognizing more than one PI on research
grants and contracts.
Beginning in May 2006, the NIH expects to start
receiving applications that name more than one PI.
Initially, this capacity will be restricted to a
small number of pre-announced Requests for
Applications (RFAs) and Program Announcements
(PAs). A phased approach will allow us to test
this very complex adaptation of our administrative
systems in order to ensure that everything is
working correctly before we open it up to all
applicants. A timeline for the complete roll-out
will be announced in the near future.
Applications identifying more than one PI will
supplement rather than replace the traditional
single-PI model. Each PI listed will be designated
as such by the grantee institution and will be
expected to share responsibility for directing the
grant-supported project or activity. Reviewers
will continue to examine qualifications of named
PIs as they relate to the proposed project. And if
an award is made, each PI will be named on the
notice of grant award and will be responsible and
accountable to the grantee institution and the NIH
for the proper conduct of the project or activity.
The NIH along with the Office
of Science and Technology Policy recently
issued Requests for Information (RFIs) in order to
gauge the level of support within the scientific
community for the overall multiple-PI concept. The
RFI issued by the NIH explored specific issues of
interest to the health-related research community,
including the desirability of resource allocation
to the individual PIs and the use of linked awards
when PIs are located at different institutions.
Responses revealed overall support for the
multiple-PI initiative, as well as for the idea of
budget allocation to individual PIs and the
development of ways to use linked awards to
support projects that span more than one
institution. The findings of the RFI can be found
on the Multiple
Principal Investigator Web site. As
implementation plans are approved, they will
appear on this website and will be described in
future issues of the Nexus.
 Back
to top Back
to top
|
 INtellectual
Property Issues? DEITR is Here to Help INtellectual
Property Issues? DEITR is Here to Help |
|
Increasingly, intellectual property (IP)
and issues related to the Bayh-Dole
Act are emerging in NIH funding
agreements. OER's Division of Extramural
Inventions & Technology Resources
(DEITR) within the Office of Policy for
Extramural Research Administration
supports government agencies, businesses,
universities, scientific researchers, and
others by providing policy and compliance
guidance, technical assistance, and
practical advice on extramural inventions
and related IP issues. DEITR is well
versed in domestic and international
patent prosecution and other intellectual
property protections, international trade,
resource sharing and technology transfer
policies, and the development and
negotiation of technology transfer
agreements.
DEITR also:
 |
Supports various other aspects
of the Bayh-Dole Act, such as
handling of waivers and evaluating
Determination of Exceptional
Circumstances for which Bayh-Dole
patent rights may flow to other
parties.
|
 |
Is responsible for the Interagency
Edison (iEdison) system, the
lead electronic means of
monitoring and adhering to the
invention reporting requirements
under the Bayh-Dole Act used
across the U.S. Government by more
than 25 federal agency
components.
|
 |
Provides ongoing guidance and
educational seminars for NIH
funding recipients on a variety of
topics, including Bayh-Dole
reporting for the research
community. |
NIH funding recipients who have
intellectual property concerns or seek IP
guidance on extramural invention/patent
matters are encouraged to contact DEITR.
 Back
to top Back
to top
|
|
 COMMITTEE
ADDRESSES ATTRACTING NEW INVESTIGATORS COMMITTEE
ADDRESSES ATTRACTING NEW INVESTIGATORS |
|
Entry of new investigators into the ranks
of independent, NIH-funded researchers is
essential to the health of this country's
biomedical research enterprise. In today's
challenging budget environment, supporting
a healthy cohort of NIH-supported new
investigators is the number one priority
of NIH Director Elias A. Zerhouni, M.D.
Dr. Zerhouni formed the NIH New
Investigator Committee to develop a list
of recommended action items that will
facilitate an investigator's ability to
receive his/her first independent R01
award earlier in their research career.
The Committee, chaired by Dr. Norka Ruiz
Bravo, Director, OER, and Dr. Story
Landis, Director, NINDS, will soon
announce a comprehensive, NIH-wide program
designed to foster new investigator
independence.
 The
committee's central recommendation is to
develop a career transition award program
to accelerate the transition to research
independence and R01 support. The career
transition program will provide five years
of support; one to two years of support
for postdoctoral fellows, and three years
of independent research support, provided
the grantee secures a tenure-track or
equivalent position. Extramural and NIH
intramural postdocs will be eligible for
these grants, with the first application
deadline expected to be this spring. The
committee's central recommendation is to
develop a career transition award program
to accelerate the transition to research
independence and R01 support. The career
transition program will provide five years
of support; one to two years of support
for postdoctoral fellows, and three years
of independent research support, provided
the grantee secures a tenure-track or
equivalent position. Extramural and NIH
intramural postdocs will be eligible for
these grants, with the first application
deadline expected to be this spring.
Information on current NIH practices
cultivating the success of new
investigators is available at the Resources
for New Investigators Web page.
 Back
to top Back
to top
|
|
|
 NIH
PUBLIC ACCESS — WHAT IT MEANS FOR YOU NIH
PUBLIC ACCESS — WHAT IT MEANS FOR YOU
|
|
On May 2, 2005, the NIH's
Enhancing Public Access to Archived Publications
Resulting from NIH-Funded Research (Public Access
Policy) became effective. The Public Access Policy
requests that investigators funded by the NIH
submit an electronic version of their final,
peer-reviewed manuscripts upon acceptance for
publication to the NIH National Library of
Medicine's PubMed Central (PMC). The Policy has
three goals: 1) to create a central archive of
NIH-funded research publications; 2) to advance
science and enable NIH to better manage its
research portfolio; and 3) to provide electronic
access to the public to NIH-funded research
publications.
 PMC
is a digital repository of full-text,
peer-reviewed biomedical, behavioral, and clinical
research journals. It is publicly accessible,
permanent and searchable. For authors and
investigators, participating in Public Access by
submitting manuscripts to PMC provides: PMC
is a digital repository of full-text,
peer-reviewed biomedical, behavioral, and clinical
research journals. It is publicly accessible,
permanent and searchable. For authors and
investigators, participating in Public Access by
submitting manuscripts to PMC provides:
 |
Higher Visibility: Investigators will be
able to ensure timely public access to
their research manuscripts.
|
 |
Time Saving Links: Investigators and
institutions can use the manuscript
submission process to fulfill the existing
requirement to provide publications as
part of progress reports.
|
 |
Cutting-edge Tools: Investigators will
benefit from the modern information
technology tools and services integrated
with the PMC system, including PubMed,
GenBank (DNA sequences), Complete Genome
Maps, Protein Sequences and Structures,
Taxonomy, PubChem (Small Molecules),
MedlinePlus, and Clinical Trials. |
There has been steady growth in
the use of PMC as the number of articles it
contains increases. From May to December 2005, the
contents of the PMC database grew from 371,000 to
515,000 articles overall, and 6.8 million unique
users viewed some 32 million copies of these
articles. Despite the outstanding visibility of
PMC publications, participation with the Public
Access Policy is low — less than 4 percent (1,636) of the
articles eligible for submission under the Public
Access policy have been added to the PMC archive.
NIH has implemented an efficient system for submitting articles. Authors
may submit in a variety of electronic formats. The median submission time to PMC is 8.5 minutes; after a few submissions and experience with the process, most authors are able to submit articles faster. NIH is also working to enable publishers to submit articles to PMC on behalf of their authors. Look for further information on the progress of publisher-submitted manuscripts later this year.
For additional information,
including Public Access Policy documents,
submission instructions, FAQs and statistics,
visit the NIH
Public Access Policy Web page.
 Back
to top Back
to top
|
|
|
|
|
|

|
|
|
The NIH Extramural Nexus is a bimonthly update
from the NIH Office of Extramural Research. Articles,
comments, questions and suggestions may be addressed to
the Editor.
The NIH Extramural Nexus reserves the right to
select and edit items submitted for inclusion.
To subscribe to the NIH Extramural Nexus, send a
plain text email to Listserv@list.nih.gov
including only the words Subscribe EXTRAMURALNEXUS
in the body of the message. To unsubscribe, follow the
same procedure, using the words Unsubscribe
EXTRAMURALNEXUS in the message body.
|
|
|
NIH Extramural
Nexus Web Page |
|
|
