|
BUTYL ACRYLATE
| Method number: |
PV2011 |
|
| Matrix: |
Air |
|
| Target Concentration: |
10 ppm (55 mg/m3) OSHA permissible exposure limit (PEL). |
|
| Procedure: |
Samples are collected by drawing known volumes of air through glass sampling tubes containing charcoal adsorbent which has been coated with 4-tert-butylcatechol (TBC). Samples are desorbed with carbon disulfide and analyzed by gas chromatography (GC) using a flame ionization detector (FID). |
|
Recommended air volume
and sampling rate: |
12 L at 0.05 L/min |
|
Detection limit of the overall
procedure (based on the
recommended air volume and
the analytical detection limit): |
0.25 ppm (1.4 mg/m3) |
|
| Status of method: |
Stopgap method. This method has been partially evaluated and is presented for information and trial use only. |
|
| Date: October 1991 (Final) |
Chemist: Ing-Fong Chan |
Organic Service Branch II
OSHA Technical Center
Salt Lake City, Utah
1. General Discussion
1.1. Background
1.1.1. History of procedure
This evaluation was undertaken to determine the effectiveness of the TBC-coated charcoal tube as a sampling device for butyl acrylate. TBC-coated charcoal tubes were initially used for 1,3-Butadiene in OSHA Method 56 (Ref. 5.1.).
1.1.2. Toxic effects (This section is for information only and should not be taken as the basis of OSHA policy.)
Butyl acrylate has an oral LD50 of 3.73 g/kg in rats (Ref. 5.2.). The dermal LD50 for rabbits was about 1800 mg/kg. Butyl acrylate was found to be moderately irritating to the skin, and as an eye irritant, produced corneal necrosis in the unwashed rabbit eye, similar to that produced by ethyl alcohol (Ref. 5.3.).
1.1.3. Potential workplace exposure
The monomer of butyl acrylate is used in the manufacture of acrylate polymers and resins for textile and leather finishes. It is also used in paint formulations (Ref. 5.2.).
1.1.4. Physical properties (Ref. 5.2. and 5.3.)
| CAS number: |
141-32-2 |
| IMIS number: |
0450 |
| Molecular weight: |
128.17 |
| Molecular formula: |
C7H12O2
|
| Density: |
0.8986 |
| Boiling point: |
145°C at 101.3 kPa (760 mmHg) |
| Solubility: |
in water at 20°C = 0.14 g/100 ml, at 40°C = 0.12 g/100 ml |
| Chemical name: |
Acrylic acid n-butyl ester; Butyl 2-propenoate |
| Appearance: |
monomer: colorless, flammable liquid
polymer: Elastic, tacky substance |
| Structure: |
CH2=CHCOOC4H9 |
1.2. Limit defining parameters
The detection limit of the analytical procedure, including a 7:1 split ratio, is 2.4 ng per injection. This is the amount of analyte which will give a peak whose height is approximately five times the baseline noise.
2. Sampling Procedure
2.1. Apparatus
2.1.1. Samples are collected by using a personal sampling pump that can be calibrated to within ± 5% of the recommended flow rate with the sampling device in line.
2.1.2. Samples are collected with 4-mm i.d. × 6-mm o.d. × 7.0 cm glass sampling tubes packed with two sections of charcoal that has been coated with 10% by weight of TBC. The front section contains 100-mg and the back section contains 50-mg of TBC-coated charcoal. The sections are held in place with glass wool plugs. Tubes were purchased from SKC, Inc. (catalog no. 226-73).
2.2. Reagents
No sampling reagents are required.
2.3. Sampling technique
2.3.1. Immediately before sampling, break off the ends of the TBC-coated charcoal tube. All tubes should be from the same lot.
2.3.2. Attach the sampling tube to the sampling pump with flexible tubing. Position the tube so that sampled air first passes through the 100-mg section.
2.3.3. Attach the tube vertically in the employee's breathing zone in such a manner that it does not impede work performance.
2.3.4. After sampling for the appropriate time, remove the sample and seal the tube with plastic caps.
2.3.5. Wrap each sample end-to-end with an OSHA seal (Form 21).
2.3.6. Record the air volume for each sample, and list any possible interferences.
2.3.7. Submit at least one blank for each set of samples. Handle the blank in the same manner as the samples, except no air is drawn through it.
2.3.8. Submit bulk samples for analysis in a separate container. Do not ship them with air samples.
2.4. Desorption efficiency
Sixteen vials, each containing 100-mg portion of TBC-coated charcoal, were divided into four groups of four vials each. They were liquid spiked with 67.4, 337, 674, and 1348 µg of butyl acrylate, respectively. These represent 0.1×, 0.5×, 1.0×, and 2.0× the target concentration. The vials were stored overnight in a freezer (-5°C), desorbed with 1.0 mL of the desorbing solution, shaken for 30 min, and analyzed as in section 3. The average desorption efficiency was 100.1%. The results are listed in Table 2.4.
Table 2.4.
Desorption Efficiency
|
Sample # |
Amount
Spiked, µg |
Amount
Found, µg |
%
Recovered |
|
D6
D7
D8
D9 |
67.4
67.4
67.4
67.4 |
69.4
67.8
69.3
68.9 |
103.0
100.6
102.8
102.2 |
|
Average of 0.1× PEL = 102.2% |
|
D10
D11
D12
D13 |
337
337
337
337 |
334
346
344
351 |
99.1
102.7
102.1
104.2 |
|
Average of 0.5× PEL = 102.0% |
|
D1
D2
D3
D4
D5 |
674
674
674
674
Blank |
617
655
668
681
0 |
91.5
97.2
99.1
101.0
Blank |
|
Average of 1× PEL = 97.2% |
|
DD1
DD2
DD3
DD4 |
1348
1348
1348
1348 |
1327
1310
1334
1354 |
98.4
97.2
99.0
100.4 |
|
Average of 2× PEL = 98.8% |
|
2.5. Retention efficiency
Five TBC-coated charcoal tubes were each liquid spiked with 3 µl (1× PEL) of a 225 µg/µl solution of butyl acrylate in carbon disulfide. These were allowed to dry for 2 hours and then 12 L of humid air (~80% relative humidity) were drawn through each tube at 0.05 L/min. The tubes were stored overnight in a freezer (-5°C), desorbed with 1.0 mL of desorbing solution, shaken for 30 min and then analyzed as in Section 3. The results are listed in Table 2.5.
Table 2.5.
Retention Efficiency
|
Sample # |
Amount
Spiked, µg |
Amount
Found, µg |
%
Recovered |
|
R1
R2
R3
R4
R5 |
674
674
674
674
674 |
638
635
654
637
649 |
94.7
94.2
97.0
94.5
96.3 |
|
|
Average = 95.3% |
|
2.6. Sample storage
Ten TBC-coated charcoal tubes were each liquid spiked with 3 µl
(1× PEL) of 225 µg/µl solution of butyl acrylate in carbon
disulfide. These were allowed to dry for 2 hours and then 12 L of
humid air (~80% relative humidity) were drawn through each tube
at 0.05 L/min. Half of the tubes were stored in a drawer at
ambient temperature, and the other half were stored in a freezer
(-5°C). After ten days they were extracted and analyzed as in
section 3. The results are given in Tables 2.6.1. and 2.6.2.
Table 2.6.1.
Ambient Storage
|
Days
Stored |
Amount
Spiked, µg |
Amount
Found, µg |
%
Recovered |
|
10
10
10
10
10 |
674
674
674
674
674 |
629
629
635
635
634 |
93.3
93.3
94.2
94.2
94.1 |
|
|
Average = 93.8% |
|
Table 2.6.2
Freezer Storage
|
Days
Stored |
Amount
Spiked, µg |
Amount
Found, µg |
%
Recovered |
|
10
10
10
10
10 |
674
674
674
674
674 |
633
652
649
654
649 |
93.9
96.7
96.3
97.0
96.3 |
|
|
Average = 96.0% |
|
2.7. Recommended air volume and sampling rate
2.7.1. The recommended air volume is 12 L.
2.7.2. The recommended flow rate is 0.05 L/min.
2.8. Interferences (sampling)
It is not known if any compounds will interfere with the
collection of butyl acrylate. Any suspected interferences should be
reported to the laboratory with submitted samples.
2.9. Safety precautions (sampling)
2.9.1. Attach the sampling equipment in such a manner that it
will not interfere with work performance or employee
safety.
2.9.2. Follow all safety practices that apply to the work area
being sampled.
3. Analytical Procedure
3.1. Apparatus
3.1.1. A GC equipped with a flame ionization detector (FID). A
Hewlett-Packard 5890 Gas Chromatograph equipped with a
7673A Autosampler and FID was used in this evaluation.
3.1.2. A GC column capable of separating butyl acrylate and the
internal standard from any interferences. A 60 m × 0.32
mm i.d. (0.25 µm film) DX-4 capillary column was used in
this evaluation.
3.1.3. An electronic integrator or some other suitable means to
measure detector response. A Waters 860 Networking
Computer System was used in this evaluation.
3.1.4. Volumetric flasks, pipets, and syringes for preparing
standards, making dilutions and performing injections.
3.1.5. Vials, 2-mL, and 4-mL, with PTFE-lined caps.
3.2. Reagents
3.2.1. Butyl acrylate. Reagent grade or better should be used.
Eastman Chemical Co. product was used in this evaluation.
3.2.2. Desorbing solution. Reagent grade or better should be
used. Fisher Scientific carbon disulfide was used in
this evaluation. p-Cymene (0.25 µL p-Cymene/mL carbon
disulfide) was added for use as an internal standard.
p-Cymene was purchased from Aldrich Chemical Company,
Inc.
3.3. Standard preparation
Prepare standards by diluting known volumes of butyl acrylate
with desorbing solution. Standards must be used the day they are
prepared.
3.4. Sample preparation
3.4.1. Transfer the 100-mg section of the sampling tube to a
2-mL vial. Place the 50-mg backup section in a separate
2-mL vial.
3.4.2. Add 1.0 mL of desorbing solution to each vial and seal
with a Teflon-lined cap.
3.4.3. Shake the vials for 30 minutes on a mechanical shaker.
3.5. Analysis
3.5.1. Instrument conditions
| Column: |
DX-4, 60 m × 0.32 mm i.d., 0.25 µm film |
| Injector temperature: |
200°C |
| Detector temperature: |
220°C |
| Column temperature: |
70°C (initial temp) |
| Temperature program: |
hold initial temp 3 min, increase temp at 10°C/min to 220°C, hold temp 5 min |
| Gas flow rates: |
|
column:
septum purge:
FID:
FID:
FID: |
1.5 mL/min (hydrogen)
1.2 mL/min (hydrogen)
19 mL/min (hydrogen)
10 mL/min (nitrogen)
480 mL/min (air) |
| Injection volume: |
1 µL |
| Split ratio: |
7:1 |
| Retention time: |
1.97 min (Butyl acrylate)
4.38 min (p-Cymene) |
3.5.2. An extended chromatographic run time was used to remove
the late eluting TBC from the analytical column.
3.5.3. Chromatogram (Figure 1.)
3.5.4. Measure detector response using a suitable method such as
electronic integration.
3.5.5. An internal standard (ISTD) calibration method is used.
A calibration curve may be constructed by plotting
concentration of analyte per sample versus ISTD-corrected
response of standard injections. Bracket the samples
with freshly prepared analytical standards over a range
of concentrations.
3.6. Interferences (analytical)
3.6.1. Any collected compound which produces an FID response and
has a similar retention time as butyl acrylate or the
internal standard is a potential interference.
3.6.2. GC conditions may generally be varied to circumvent interferences.
3.6.3. Retention time on a single column is not proof of
chemical identity. Analysis by an alternate GC column, high
performance liquid chromatography (HPLC) and confirmation
by mass spectrometry are additional means of identification.
3.7. Calculations
3.7.1. A calibration curve may be constructed by plotting
detector response versus ISTD-corrected response of
standard concentration (µg/mL) of butyl acrylate.
3.7.2. Determine the µg/mL of butyl acrylate in both sections of
each sample and blank from the calibration curve. If
butyl acrylate is found on the backup section, it is
added to the amount found on the front section. Blank
corrections should be performed before adding the results together.
3.7.3. Determine the air concentration by using the following formula.
| mg/m3 = |
(µg/mL, blank corrected) × (desorption volume, mL)
(air volume, L) × (desorption efficiency, decimal) |
| ppm = |
(mg/m3)(24.46)
(128.17) |
| where |
24.46
128.17 |
=
= |
molar volume (liters) at 101.3 kPa (760 mmHg) and 25°C
molecular weight of butyl acrylate |
3.8. Safety precautions (analytical)
3.8.1. Avoid skin contact and air exposure to butyl acrylate.
3.8.2. Avoid skin contact with all solvents.
3.8.3. Wear safety glasses at all times.
4. Recommendation for Further Study
This method should be fully validated.
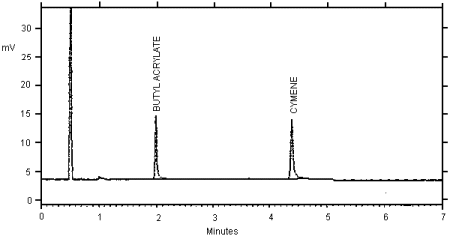
Figure 1. Chromatogram of butyl acrylate
5. References
5.1. "OSHA Analytical Methods Manual", 2nd ed., U.S. Department of
Labor, Occupational Safety and Health Administration; OSHA
Analytical Laboratory; Salt Lake City, UT, 1990; Method 56 -
1,3-Butadiene; American Conference of Government Industrial
Hygienists (ACGIH); Cincinnati, OH, Publ. No. 4542.
5.2. Merck Index, 10th ed.; Windholz, Martha ED.; Merck: Rahway,
N.J., 1983; p1512.
5.3. Documentation of the Threshold Limit Values and Biological
Exposure Indices, American Conference of Governmental Industrial
Hygienist INC., fifth edition, 1986; p 75.
|

