|
2-Hexanone
| Method number: |
PV2031 |
| |
| Matrix: |
Air |
| |
| Target concentration: |
100 ppm (410 mg/m3) (OSHA TWA PEL)
5 ppm (20 mg/m3) (ACGIH TWA TLV)(skin) |
| |
| Procedure: |
Samples are collected by drawing a known volume of air
through a charcoal tube. Samples are desorbed with 1 mL of 1:99
dimethyl formamide:carbon disulfide (DMF:CS2) for 30 minutes
with shaking and analyzed by gas chromatography using a flame ionization
detector (GC-FID). |
| |
Recommended air volume
and sampling rate: |
20 L at 0.2 L/min |
| |
| Reliable quantitation limit: |
0.018 ppm (0.076 mg/m3) |
| |
| Special requirements: |
Samples should be refrigerated after sampling as
soon as possible, and analyzed within one week. |
| |
| Status of method: |
Partially Evaluated Method. This method has been
subjected to established evaluation procedures, and is presented for
information and trial use. |
| |
| Date: September, 1995 |
Chemist: Mary E. Eide |
Organic Service Branch I
OSHA Salt Lake Technical Center
Salt Lake City, UT 84165-0200
1. General Discussion
1.1 Background
1.1.1 History
2-Hexanone (MBK) was a compound listed in OSHA method 7 Organic Vapors
(Ref. 5.1), but only desorption studies had been performed. There is
a proposal to lower the PEL of 100 ppm to the TLV level of 5 ppm. The
purpose of this study was to obtain data on retention and storage, and
to perform desorption studies over the two exposure levels. The
retention studies showed good recoveries when 20 liters of humid air
(80% RH at 22 °C) was drawn at 0.2 L/min. The storage studies showed
lower recoveries for those samples stored at ambient temperature, 23°C,
than the samples stored at refrigerated temperature, -10C. Humidity
affected the samples; those samples which had 20 liters of humid air
(80% RH at 21°C), drawn through them had lower recoveries. The ambient
humid air drawn samples had recoveries of 71.4% on day 7 and 45.4% on
day 14. The refrigerated humid air drawn samples had recoveries of
80.5% on day 7 and 64.5% on day 14. The dry samples stored at ambient
temperature had recoveries of 89.6% on day 7 and 82.5% on day 14. The
dry samples stored at refrigerated temperatures had recoveries of 93.3%
on day 7 and 91.9% on day 14. These storage studies indicate that MBK
samples should be refrigerated as soon as possible after sampling, and
should be analyzed within a week of sampling.
1.1.2 Toxic effects (This section is for information only and should not
be taken as the basis of OSHA policy.) (Ref. 5.2)
MBK is an irritant to eyes, skin and mucous membranes. Worker exposure
to 1000 ppm exhibited moderate irritation to eyes and mucous membranes.
In prolonged exposure to lower concentrations MBK can act as a
neurotoxin, which may cause permanent nerve damage. Workers exposed to
approximately 75 ppm over 9 months showed a decrease of sciatic-tibial
nerve conduction. Workers at another site showed a decrease in nerve
function after exposure to approximately 50 ppm over 6 months. The intake
of alcoholic beverages may enhance the toxic effects of MBK exposures.
1.1.3 Workplace exposure (Ref. 5.2 and 5.3)
MBK is used as a solvent in paints, lacquers, varnishes, nitrocellulose,
fats, and waxes.
1.1.4 Physical properties and other descriptive information (Ref. 5.2,
5.3, and 5.4)
| Synonyms: |
N-Butyl methyl ketone; Methyl butyl ketone; Propyl acetone |
| CAS number: |
591-78-6 |
| IMIS: |
1690 |
| RTECS: |
MP1400000 |
| DOT: |
UN 1224 (flammable liquid) |
| Molecular weight: |
100.16 |
| Flash point: |
19°C (67 °F)(cc) |
| Boiling point: |
127°C |
| Melting point: |
-57°C |
| Odor: |
acetone-like, but more pungent |
| Color: |
clear liquid |
| Autoignition temperature: |
423°C (795 °F) |
| Density: |
0.830 (d2020) |
| Molecular formula: |
C6H12O |
| Structural formula: |
 |
The analyte air concentrations throughout this method are based on the
recommended sampling and analytical parameters. Air concentrations
listed in ppm are referenced to 25°C and 101.3 kPa (760 mmHg).
1.2 Limit defining parameters
1.2.1 Detection limit of the overall procedure (DLOP)
The detection limit of the overall procedure is 0.454 µg per sample
(0.00554 ppm or 0.227 mg/m3). This is the amount of analyte spiked
on the sampler that will give a response that is significantly different
from the background response of a sampler blank.
The DLOP is defined as the concentration of analyte that gives a response
(YDLOP) that is significantly different (three standard
deviations (SDBR)) from the background response
(YBR).
YDLOP - YBR = 3(SDBR)
The direct measurement of YBR and SDBR in
chromatographic methods is typically inconvenient, and difficult
because YBR is usually extremely low. inconvenient, and
difficult because YBR is usually extremely low. Estimates
of these parameters can be made with data obtained from the analysis
of a series of samples whose responses are in the vicinity of the
background response. The regression curve obtained for a plot of
instrument response versus concentration of analyte will usually be
linear. Assuming SDBR and the precision of data about the
curve are similar, the standard error of estimate (SEE) for the
regression curve can be substituted for SDBR in the above
equation. The following calculations derive a formula for the DLOP:

| Yobs |
= |
observed response |
| Yest |
= |
estimated response from regression curve |
| n |
= |
total no. of data points |
| k |
= |
2 for a linear regression curve |
At point YDLOP on the regression curve
| YDLOP = A(DLOP) +YBR |
A = analytical sensitivity (slope) |
therefore
Substituting 3(SEE) + YBR for YDLOP gives
The DLOP is measured as mass per sample and expressed as equivalent air
concentrations, based on the recommended sampling parameters. Ten
samplers were spiked with equal descending increments of analyte, such
that the highest sampler loading was 8.3 µg/sample. This is the amount,
when spiked on a sampler, that would produce a peak approximately 10
times the background response for the sample blank. These spiked samplers,
and the sample blank were analyzed with the recommended analytical
parameters, and the data obtained used to calculate the required
parameters A and SEE) for the calculation of the DLOP. Values of 123.68
and 18.24 were obtained or A and SEE respectively. DLOP was calculated
to be 0.454 µg/sample (0.00554 ppm or 0.0227 mg/m3).
Table 1.2.1
Detection Limit of the Overall Procedure
|
| mass per sample |
area counts |
| (µg) |
(µV-s) |
|
| 0 |
0 |
| 0.83 |
124 |
| 1.66 |
243 |
| 2.49 |
353 |
| 3.32 |
457 |
| 4.15 |
579 |
| 4.98 |
684 |
| 5.81 |
773 |
| 6.64 |
846 |
| 7.47 |
976 |
| 8.30 |
1045 |
|
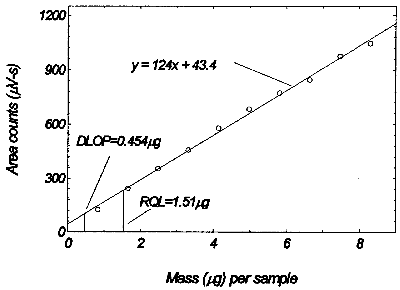
Figure 1.2.1. Plot of data to determine the DLOP/RQL.
1.2.2 Reliable quantitation limit (RQL)
The reliable quantitation limit is 1.51 µg per sample (0.018 ppm). This
is the amount of analyte spiked on a sampler that will give a signal that
is considered the lower limit for precise quantitative measurements.
The RQL is considered the lower limit for precise quantitative
measurements. It is determined from the regression line data obtained
for the calculation of the DLOP (Section 1.2.1), providing at least 75%
of the analyte is recovered. The RQL is defined as the concentration
of analyte that gives a response (YRQL) such that
YRQL - YBR = 10(SDBR)
therefore
RQL= 1.51µg per sample (0.018 ppm)

Figure 1.2.2. Plot of data to determine the RQL.
Table 1.2.2
Reliable Quantitation Limit
|
| mass per sample |
mass recovered |
recovery |
| (µg) |
(µg) |
(%) |
|
| 0.83 |
0.803 |
96.7 |
| 1.66 |
1.52 |
91.6 |
| 2.49 |
2.43 |
97.6 |
| 3.32 |
3.20 |
96.4 |
| 4.15 |
3.93 |
94.7 |
| 4.98 |
4.84 |
97.2 |
| 5.81 |
5.60 |
96.4 |
| 6.64 |
6.45 |
97.1 |
| 7.47 |
7.16 |
95.9 |
| 8.30 |
8.06 |
97.1 |
|

Figure 1.2.3. Chromatogram of the RQL.
2. Sampling Procedure
2.1 Apparatus
2.1.1 Samples are collected using a personal sampling pump calibrated,
with the sampling device attached, to within ±5% of the recommended flow
rate.
2.1.2 Samples are collected with tubes 7 cm x 4 mm i.d. x 6 mm o.d.
glass sampling tubes packed with two sections of charcoal, lot 120. The
front section contains 100 mg and the back section contains 50 mg of
charcoal, lot 120. The sections are held in place with glass wool plugs
and are separated by a urethane foam plug. For this evaluation,
commercially prepared sampling tubes were purchased from SKC Inc.,
(Eighty Four PA) catalog No. 226-01, Lot 120.
2.2 Technique
2.2.1 Immediately before sampling, break off the ends of the sampling
tube. All tubes should be from the same lot.
2.2.2 Attach the sampling tube to the pump with flexible tubing. It is
desirable to utilize sampling tube holders which have a protective
cover to shield the employee from the sharp, jagged end of the sampling
tube. Position the tube so that sampled air passes through the front
section of the tube first.
2.2.3 Air being sampled should not pass through any hose or tubing before
entering the sampling tube.
2.2.4 Attach the sampler vertically with the front section pointing
downward, in the worker's breathing zone, and positioned so it does not
impede work performance or safety.
2.2.5 After sampling for the appropriate time, remove the sample and seal
the tube with plastic end caps. Wrap each sample end-to-end with a Form
OSHA-21 seal.
2.2.6 Submit at least one blank sample with each set of samples. Handle
the blank sample in the same manner as the other samples except draw no
air through it.
2.2.7 Record sample volumes (in liters of air) for each sample, along
with any potential interferences.
2.2.8 Ship any bulk samples separate from the air samples.
2.2.9 Submit the samples to the laboratory for analysis as soon as
possible after sampling. If delay is unavoidable, store the samples in
a refrigerator.
2.3 Desorption efficiency
2.3.1 The desorption efficiencies of MBK were determined by liquid-spiking
the charcoal tubes with the analytes at 0.05 to 2 times the target
concentration. The loadings on the tubes were 91.3, 457, 913, 1826,
and 3652 µg of MBK. These samples were stored overnight at ambient
temperature and then desorbed with 1 mL of 1:99 DMF:CS2 with
0.25 µL/mL p-cymene internal standard, and analyzed by GC-FID.
The average desorption efficiency over the studied range was 96.6%.
The solution of 1:99 DMF:CS2 with 0.25 µL/mL p-cymene
internal standard was used to desorb the samples generated for the
retention, storage, and detection limit studies in this method.
Table 2.3.1
Desorption Efficiency of MBK
|
|
% Recovered |
|
|
0.05 X |
0.25 X |
0.5 X |
1.0 X |
2.0 X |
| Tube # |
91.3µg |
457µg |
913µg |
1826µg |
3652µg |
|
| 1 |
96.7 |
95.3 |
96.0 |
96.7 |
98.2 |
| 2 |
95.8 |
96.8 |
96.6 |
96.6 |
97.6 |
| 3 |
96.7 |
96.2 |
96.5 |
97.0 |
97.2 |
| 4 |
96.8 |
96.3 |
96.7 |
96.0 |
96.9 |
| 5 |
96.4 |
96.8 |
96.7 |
95.5 |
96.9 |
| 6 |
96.6 |
97.7 |
96.6 |
96.7 |
97.2 |
| average |
96.5 |
96.5 |
96.5 |
96.4 |
97.3 |
| overall average |
96.7 |
|
standard
deviation |
±0.60 |
|
|
2.3.2 The desorption efficiencies of MBK, using the other desorbing
solvent mentioned in OSHA method 7 Organic Vapors, carbon disulfide,
were determined by liquid-spiking the charcoal tubes with the analytes
at 0.05 to 2 times the target concentration. The loadings on the tubes
were 91.3, 457, 913, 1826, and 3652 µg of MBK. These samples were
stored overnight at ambient temperature and then desorbed with 1 mL of
CS2 with 0.25 µL/mL p-cymene internal standard, and
analyzed by GC-FID. The desorption efficiency over the range studied
was non-linear, with lower recoveries at the lower loadings. This
desorbing solvent was not used for the rest of this study due to this
non-linear desorption efficiency.
Table 2.3.2
Desorption Efficiency of MBK
|
|
% Recovered |
|
|
0.05 X |
0.25 X |
0.5 X |
1.0 X |
2.0 X |
| Tube # |
91.3µg |
457µg |
913µg |
1826µg |
3652µg |
|
| 1 |
77.2 |
82.5 |
85.8 |
89.9 |
91.0 |
| 2 |
74.7 |
82.0 |
85.7 |
88.0 |
92.3 |
| 3 |
74.2 |
81.6 |
85.3 |
89.7 |
90.2 |
| 4 |
75.7 |
82.1 |
83.4 |
86.8 |
91.0 |
| 5 |
75.7 |
82.0 |
86.1 |
87.5 |
91.2 |
| 6 |
73.8 |
81.1 |
84.3 |
88.0 |
91.7 |
| average |
75.2 |
81.9 |
85.1 |
88.2 |
91.2 |
|
2.4 Retention efficiency
The sampling tubes were spiked with 1826 µg (22.3 ppm) MBK, allowed to
equilibrate for 4 hours at room temperature, and then had 20 L humid
air (80% RH at 21°C) pulled through them at 0.2 L/min. They were opened,
desorbed, and analyzed by GC-FID. The retention efficiency averaged
93.5%. There was no MBK found on the back sections of the tubes.
Table 2.4
Retention Efficiency of MBK
|
| Tube # |
|
% Recovered |
|
|
Front section |
Back section |
Total |
|
| 1 |
94.6 |
0 |
94.6 |
| 2 |
93.4 |
0 |
93.4 |
| 3 |
92.6 |
0 |
92.6 |
| 4 |
94.2 |
0 |
94.2 |
| 5 |
94.9 |
0 |
94.9 |
| 6 |
91.0 |
0 |
91.0 |
|
average |
93.5 |
|
2.5 Sample storage
The front sections of twelve sampling tubes were each spiked with 913 µg
(11.1 ppm) of MBK, then six tubes were stored in the refrigerator (-10°C),
and six tubes were stored at room temperature 23°C. Twelve more tubes
were spiked with 913 µg (22.3 ppm) of MBK, and then had 20 liters of
humid air (80% RH at 21°C) drawn through them; afterwards six tubes
were stored in the refrigerator (-10°C), and six tubes were stored at
room temperature 23°C. Three of each type of samples were analyzed after
7 days and the remaining three samples of each type after 14 days.
The amounts recovered indicate that humidity and temperature affect
the ability of charcoal to retain intact the MBK. The recoveries
decreased with time and/or added humidity, with the lowest recovery on
day 14 day storage with humidity.
Table 2.5
Storage Test for MBK
|
| Time |
%Recovery |
%Recovery |
%Recovery |
%Recovery |
| (days) |
Humid |
Humid |
Dry |
Dry |
|
Ambient |
Refrigerated |
Ambient |
Refrigerated |
|
| 7 |
67.7 |
80.5 |
89.7 |
95.4 |
| 7 |
70.5 |
80.6 |
92.2 |
90.8 |
| 7 |
75.9 |
80.2 |
87.0 |
93.7 |
| average |
71.4 |
80.5 |
89.6 |
93.3 |
| 14 |
45.5 |
66.4 |
80.4 |
92.9 |
| 14 |
46.5 |
63.0 |
84.2 |
91.3 |
| 14 |
44.1 |
64.0 |
83.0 |
91.4 |
| average |
45.4 |
64.5 |
82.5 |
91.9 |
|
2.6 Recommended air volume and sampling rate.
Based on the data collected in this evaluation, 20 L air samples should
be collected at a sampling rate of 0.1 L/min, with a maximum rate of
0.2 L/min.
2.7 Interferences (sampling)
2.7.1 It is not known if any compounds will severely interfere with the
collection of MBK on the sampling tubes. In general, the presence of
other contaminant vapors in the air will reduce the capacity of the
charcoal tube to collect MBK.
2.7.2 Suspected interferences should be reported to the laboratory with
submitted samples.
2.8 Safety precautions (sampling)
2.8.1 Attach the sampling equipment to the worker in such a manner that
it will not interfere with work performance or safety.
2.8.2 Follow all safety practices that apply to the work area being
sampled.
2.8.3 Wear eye protection when breaking the ends of the glass sampling
tubes.
3. Analytical Procedure
3.1 Apparatus
3.1.1 The instrument used in this study was a gas chromatograph equipped
with a flame ionization detector, specifically a Hewlett Packard model
5890.
3.1.2 A GC column capable of separating the analyte from any
interferences. The column used in this study was a 60 meter capillary
column with a 0.5 µ coating of DB-WAX, with an I.D. of 0.32 mm.
3.1.3 An electronic integrator or some suitable method of measuring peak
areas.
3.1.4 Two milliliter vials with TeflonTM-lined caps.
3.1.5 A 10µL syringe or other convenient size for sample injection.
3.1.6 Pipets for dispensing the desorbing solution. A Repipet®
dispenser was used in this study.
3.1.7 Volumetric flasks - 5 or 10 mL and other convenient sizes for
preparing standards.
3.2 Reagents
3.2.1 GC grade nitrogen, hydrogen, and air.
3.2.2 2-Hexanone(MBK), Reagent grade
3.2.3 Carbon disulfide (CS2), Reagent grade
3.2.4 Dimethyl formamide (DMF), Reagent grade
3.2.5 p-Cymene, Reagent grade (internal standard)
3.2.6 Desorbing solution was 1:99 DMF:CS2 with 0.25 µL/mL
p-cymene internal standard.
3.3 Standard preparation
3.3.1 At least two separate stock standards are prepared by diluting a
known quantity of MBK with the desorbing solution of 1:99
DMF:CS2 with 0.25 µL/mL p-cymene internal standard.
The concentration of these stock standards was 1 µL/mL or 830 µg/mL.
3.3.2 A third standard at a higher concentration was prepared to check
the linearity of the calibration. For this study, two analytical
standards were prepared at a concentration of 1 µL/mL (830 µg/mL), and one
at 4 µL/mL (3320 µg/mL) MBK in the desorbing solution.
3.4 Sample preparation
3.4.1 Sample tubes are opened and the front and back section of each tube
are placed in separate 2 mL vials.
3.4.2 Each section is desorbed with 1 mL of the desorbing solution of
1:99 DMF:carbon disulfide with 0.25 µL/mL p-cymene internal
standard.
3.4.3 The vials are sealed immediately and allowed to desorb for 30
minutes with constant shaking.
3.5 Analysis
3.5.1 Gas chromatograph conditions.
| Injection size: |
1 µL |
|
| Flow rates (mL/min) |
|
| Nitrogen (make-up): |
30 |
|
| Hydrogen(carrier): |
2 |
|
| Hydrogen(detector): |
40 |
|
| Air: |
420 |
|
| Temperatures (°C) |
|
| Injector: |
200 |
|
| Detector: |
220 |
|
| Column: |
50° for 2 min then 10°/min to 160° for 4 min |
|

Figure 3.5.1 Chromatogram of an analytical standard at the target
concentration. Peak identification; (1) carbon disulfide, (2) benzene,
(3) MBK, (4) p-cymene, (5) DMF.
3.5.2 Peak areas are measured by an integrator or other suitable means.
3.6 Interferences (analytical)
3.6.1 Any compound that produces a response and has a similar retention
time as the analyte is a potential interference. If any potential
interferences were reported, they should be considered before samples are
desorbed. Generally, chromatographic conditions can be altered to
separate an interference from the analyte.

Figure 3.6.1 A mass spectra of 2-hexanone (MBK).
3.6.2 When necessary, the identity or purity of an analyte peak may be
confirmed by GC-mass spectrometer or by another analytical procedure.
3.7 Calculations
3.7.1 The instrument was calibrated with a standard of 830 µg/mL MBK in
the desorbing solution. The linearity of the calibration was checked
with a standard of 3320 µg/mL.
3.7.2 If the calibration is non-linear, two or more standard at different
concentrations must be analyzed, bracketing the samples, so a calibration
curve can be plotted and sample values obtained.
3.7.3 To calculate the concentration of analyte in the air sample the
following formulas are used:
(µg/m)(desorption volume)
(desorption efficiency) |
= mass of analyte in sample |
(mass of analyte in sample)
molecular weight |
= number of moles of analyte |
| (number of moles of analyte) |
(molar volume at 25°C & 760mm) |
= |
volume the analyte will occupy at 25°C & 760mm |
(volume analyte occupies)(106)
(air volume) |
= ppm |
* All units must cancel.
3.7.4 The above equations can be consolidated to the following formula.
(µg/mL)(DV)(24.46)(106)(g)(mg)
(20 L)(DE)(MW)(1000 mg)(1000 µg) |
= ppm |
3.7.5 This calculation is done for each section of the sampling tube and
the results added together.
3.8 Safety precautions (analytical)
3.8.1 Avoid skin contact and inhalation of all chemicals.
3.8.2 Wear safety glasses, gloves and a lab coat at all times while in
the laboratory areas.
4. Recommendations for Further Study
Collection studies need to be performed from a dynamically generated
test atmosphere. Other sampling medias should be explored to find one
that will provide better storage stability.
5. References
5.1 Shulsky, M., Method 7 "Organic Vapors", Organic Methods Evaluation
Branch, OSHA Salt Lake Technical Center, 1989.
5.2 "Documentation of the Threshold Limit Values and Biological Exposure
Indices", Fifth Edition, American Conference of Governmental Industrial
Hygienists Inc., Cincinnati, OH, 1986, p.378.
5.3 Windholz, M., "The Merck Index", Eleventh Edition, Merck & Co.,
Rahway N.J., 1989, p. 951.
5.4 Lewis, R., "Hawley's Condensed Chemical Dictionary", Twelfth edition,
Van Nostrand Reinhold Co., 1993, p.760.
|

